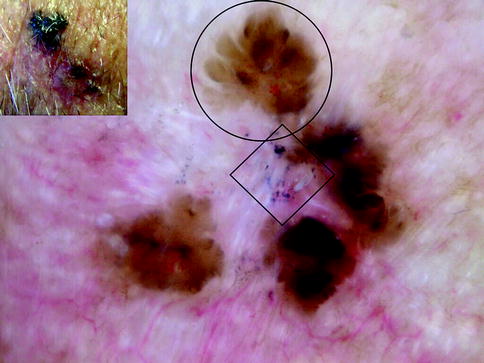
Fig. 13.1
Clinical image (left upper corner) and dermoscopy of a nevus showing symmetry in the distribution of colors and structures and predominantly one color (brown). The predominant pattern is reticular with presence of some globules regularly distributed
Dermoscopy Features
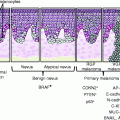
Several dermoscopy structures with specific histopathological correlates have been described and used for the description of a given lesion. Some authors use more descriptive/geometrical terms, and others use the classical metaphorical description initiated with pattern analyses, but both describe the same structures with the same histopathological meanings. In this chapter the metaphoric terms will be used (see the definition, histopathological correlation, and diagnostic association of each feature in Table 13.1) [4, 10, 11]. The dermoscopy features associated with the presence of melanin in cutaneous structures are the pigment network, dots and globules, streaks, negative of pigment network, peppering, blue-white veil, blotches, and blue-gray-brown structures. The features associated to keratin are milia-like cyst, comedo-like opening, fissures, and crypts. The criteria associated to dermal component, mainly fibrosis, are crystalline structures or white shiny streaks, central white patch, and scar-like depigmentation. Features associated to the presence of hemoglobin are vessels (comma-like, dotted, polymorphous, corkscrew, arborizing, fine horizontal in focus, hairpin, crown, and glomerular), red-blue lagoons, and milky-red areas. Finally ulceration may also be seen.
Table 13.1
Definition of dermoscopy structures and their histopathological correlation
Structure | Definition | Histopathological correlation |
|---|---|---|
Pigment network | Weblike structure consisting of brown or black lines and hypopigmented holes | Melanocytic pigmentation at the rete ridges |
Typical network | Uniform, regular lines and holes, even color, fades in the periphery | Regular melanocytic pigmentation at the rete ridges in nevus |
Atypical network | Nonuniform, darker, and/or broadened lines; heterogeneous holes in areas or shapes; abruptly ends at the perimeter | Disarranged melanocytic pigmentation at the rete ridges in atypical melanocytic lesions and melanoma |
Pseudo-network | Because the face has absent or poorly developed rete ridges, diffuse pigmentation interrupted by the surface openings of the adnexal structures | Pigment at the epidermis or dermis interrupted by follicular openings of the face |
Structureless (homogeneous) areas | Regions devoid of structures without signs of regression | |
Dots | Black, brown, or blue-gray, small spherical structures less than 0.1 mm diameter | Aggregates of melanocytes or melanin granules at the dermis, epidermis, or stratum corneum |
Globules | Brown, black, or red spherical or ovoid structures with diameters usually greater than 0.1 mm | Brown globules are nests of melanocytes at the upper dermis or dermoepidermal junction. Red globules are vessels |
Cobblestone globules | Polygonal globules crowded together causing their deformation resulting in a cobblestone pattern | Large dermal nevus nests |
Radial streaming | Linear extensions at the edge of the lesion | Aggregates of tumoral cells running parallel to the epidermis (Spitz/Reed nevi or radial growth phase of melanoma) |
Pseudopods | Brown-black, fingerlike projections from the perimeter of the lesion. Variously shaped knobs are present at the termini of the projections | Aggregates of tumoral cells running parallel to the epidermis (Spitz/Reed nevi or radial growth phase of melanoma) |
Streaks | Alternate term for radial streaming and/or pseudopods | Aggregates of tumoral cells running parallel to the epidermis (Spitz/Reed nevi or radial growth phase of melanoma) |
When symmetrically arranged around the entire edge of the lesion, the appellation “starburst pattern” is used | ||
Blotches | Black, usually homogeneous, areas of pigment obscuring underlying structures | Aggregates of melanin in stratum corneum, epidermis, and upper dermis |
Regression areas | White, scar-like depigmentation often combined with blue-gray peripheral zone and/or peppering (speckled blue-gray granules) | Regression with melanophages, fibrosis, and neo-angiogenesis |
Blue-white veil | Irregularly marginated, confluent blue pigmentation with overlying, white, ground-glass haze | Compact aggregation of heavily pigmented tumor cells in the superficial dermis in combination with compact orthokeratosis, acanthosis, and more or less pronounced hypergranulosis |
Vascular structures | The primary structure of hemangiomas and vascular malformations are clusters of blood vessels called “lacunae” or “saccules” | Tumor vessels |
Various telangiectasias of multiple shapes and sizes including comma, pinpoint, arborizing, wreath-like, hairpin-like, irregular | ||
Maple leaflike areas | Brown to blue-gray, discrete structures resembling leaflike patterns | Nodules of pigmented epithelial cells located in the upper dermis |
Spoke-wheel-like structures | Brown to gray-blue radial projections meeting at a darker brown or black central “hub” | Nests of basal cell tumor in the upper dermis |
Large blue-gray ovoid nests | Circumscribed, blue-gray or brown, ovoid structures larger than globules | Nests of basal cell tumor in the upper dermis |
Multiple blue-gray globules | Spherical, well-circumscribed structures which, in the absence of a pigment network, suggest basal cell carcinoma | Nests of basal cell tumor in the upper dermis |
Milia-like cysts | Small, white, or yellow cystic structures resembling milia which often shine brightly (“stars in the sky”) when viewed with the dermoscope. Pigmented milia-like cysts can resemble brown globules | Intraepidermal keratin cysts |
Comedo-like openings | Blackhead-like plugs due to keratin-filled invaginations on the skin surface | Comedo-like openings containing keratin |
Ridges and fissures | Cerebriform surface resulting in gyri and sulci. The latter can be filled with keratin producing irregular linear, pigmented bands | Tumoral masses with papillomatosis and acanthosis characteristics of seborrheic keratosis and papillomatous nevi |
Fingerprint-like structures | Thin brown lines resulting in patterns that resemble fingerprints | Hyperkeratosis and acanthosis characteristics of seborrheic keratosis |
Patterns
Pattern Analysis for the Diagnosis of Nevus
The presence of uniform and regularly distributed reticular, globular, starburst, and homogeneous blue patterns identifies a given lesion as a melanocytic nevus (i.e., per definition absence of melanoma-specific patterns) [4]. Each pattern corresponds to a specific underlying histopathologic correlate.
Based on the most common dermoscopic patterns associated with melanocytic nevi, a new classification has been recently proposed that includes four main categories: globular, reticular, starburst, and homogenous blue nevi [22, 23]. In this classification system, small congenital nevi, compound nevi, and dermal nevi are lumped together in the globular category based on their common dermoscopic-histopathologic features (globules correspond to predominantly dermal nests of melanocytes). The reticular pattern typically corresponds to junctional or lentiginous nevi, with the exception of congenital nevi in the lower extremities [24, 25]. The starburst category includes both pigmented Spitz nevi and Reed nevi based on their striking dermoscopic features [26] (regular peripheral streaks). Homogenous structureless blue pigmentation without additional dermoscopic features is the pattern commonly observed in blue nevi [17]. Nevi with a homogeneous structureless brown pattern are not considered within the group of blue nevi because they reveal histopathologic correlates similar to reticular nevi [27, 28].
Based on the work of Kittler et al. [29, 30] in digital follow-up, growing nevi can be easily recognized by their striking dermoscopic hallmark of a peripheral rim of small brown globules frequent in adolescents and young adults (Fig. 13.2). The same pattern in individuals older than 60 years should raise suspicion and preferably be excised and studied to rule out malignancy.
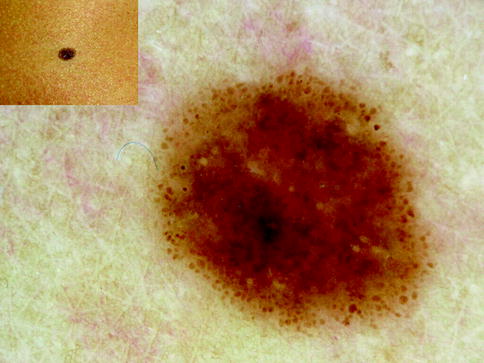

Fig. 13.2
Clinical image (left upper corner) and dermoscopy of a nevus showing symmetry in the distribution of colors and structures, with two colors (brown and gray) and globular-homogeneous pattern. The lesion presents a rim of globules characteristic of growth, in this case a nevus in young adult
Spitz and Reed nevi reveal different patterns, depending on the growth phase of the lesion [31–33]. After an initial globular pattern, Spitz and Reed nevi tend to show the classic starburst pattern, which represents an intermediate pattern of their evolution, showing finally a reticular or homogeneous pattern at the end. Since no single criterion allows differentiating such spitzoid-appearing melanomas from Spitz or Reed nevi with sufficient accuracy, excision of all spitzoid lesions, in adults, is always recommended [34, 35].
Conversely, homogeneous blue nevi seem to be highly stable lesions. This stability of blue nevi is an important, although subjective, clue for the diagnosis because blue color alone is a highly unspecific feature that may occur also in nodular melanoma, melanoma metastases, or pigmented basal cell carcinoma (4–17–23). Therefore, the diagnosis of blue nevi should always be based on the combination of the dermoscopic pattern and a convincing subjective history of no changes. When a reliable history is difficult to obtain, excision, not monitoring, must be performed.
Pattern Analyses for the Diagnosis of Melanoma
On the contrary, melanoma tends to show multiple colors and structures. A given melanocytic lesion with multiple colors, asymmetry, and multicomponent pattern (three or more structures) is highly suggestive of melanoma. For those lesions that we do not recognize at a first glance as melanoma with global pattern analyses, various dermoscopic algorithms may be applied to differentiate nevi from melanoma [4, 36–38]. Pattern analyses consider the presence of some structures associated to melanoma as additional ten clues for the diagnosis of melanoma: the presence of atypical pigment network, atypical dots and globules, regression structures, crystalline structures, negative pigment network, irregular blotches, irregular pseudopods or streaks, blue-white veil, milky red areas, or polymorphous/dotted vessels (Figs. 13.3, 13.4, 13.5, 13.6, and 13.7).
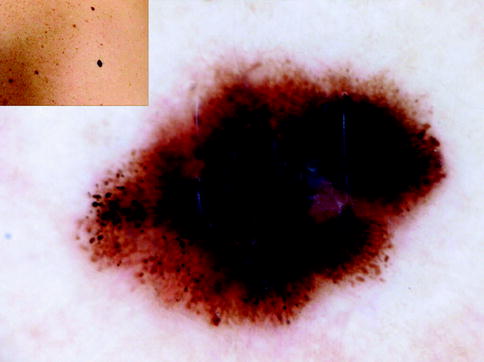
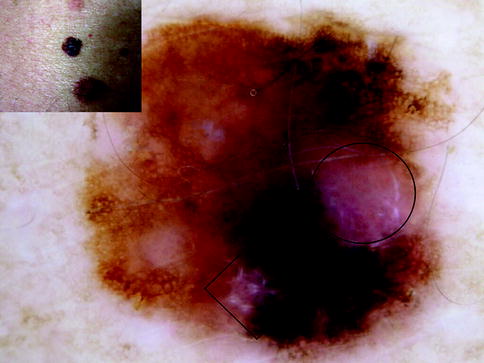
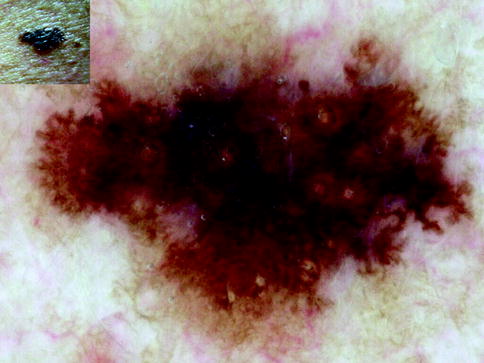
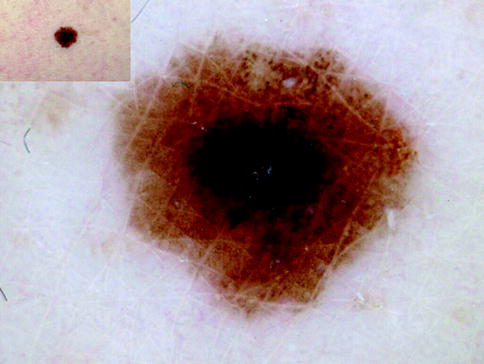
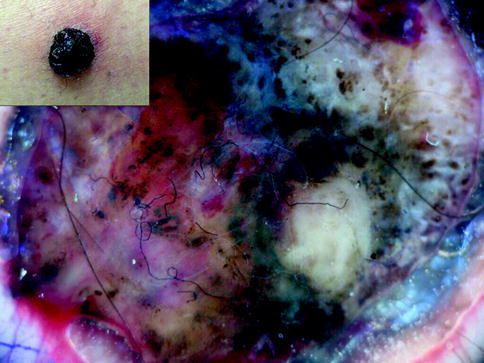

Fig. 13.3
Clinical image (left upper corner) and dermoscopy of a superficial spreading melanoma showing asymmetry in the distribution of colors and structures, multiple colors (black, light brown, dark brown, and blue), and multicomponent pattern. The lesion shows atypical network, atypical dots and globules, blotch and blue whitish veil

Fig. 13.4
Clinical image (left upper corner) and dermoscopy of a superficial spreading melanoma showing asymmetry in the distribution of colors and structures, multiple colors (black, light brown, dark brown, red, white, and blue), and multicomponent pattern. The lesion shows atypical network, atypical dots and globules, blue whitish veil, dotted vessels in milky red areas (circle), and crystalline white lines (chrysalides) (square)

Fig. 13.5
Clinical image (left upper corner) and dermoscopy of a superficial spreading melanoma showing asymmetry in the distribution of colors and structures, two colors (dark brown and gray), and reticular pattern. The lesion shows atypical network, atypical dots and globules, and peppering (gray dots) characteristic of melanophages in regression. It is a second melanoma diagnosed during follow-up of a patient carrier of the mutation G101W in CDKN2A

Fig. 13.6
Clinical image (left upper corner) and dermoscopy of an in situ melanoma showing asymmetry in the distribution of structures, three colors (black, gray, and dark brown), and reticular-homogeneous pattern. The lesion shows atypical network (upper part), atypical dots and globules, and a central black blotch

Fig. 13.7
Clinical image (left upper corner) and dermoscopy of a nodular melanoma showing asymmetry in the distribution of colors and structures, multiple colors (black, light brown, dark brown, red, white, and blue), and unspecific pattern. The lesion shows atypical dots and globules, blue whitish veil, atypical vessels, milky red areas, and ulceration with the sign of sticky fibers (textile fibers stick to the serum or blood in ulcerated tumors). In a nodular tumor the combination of black and blue and red and white is suggestive of malignancy
According to the results of the Consensus Net Meeting on Dermoscopy, the sensitivity for melanoma of these algorithms (pattern analysis, the ABCD rule, 7-point checklist, or Menzies scoring method) was similar, but pattern analysis demonstrated a better specificity [4].
However, dermoscopy is not 100 % accurate, and a certain percentage of suspicious but benign lesions have to be excised in order not miss melanoma. It is worth highlighting again that malignant cutaneous melanoma may sometimes mimic benign melanocytic and non-melanocytic lesions. For this reason, it is important that a good clinical-dermoscopic correlation exists, and the presence of conflicting clinical- dermoscopic features should lead the clinician to perform a biopsy.
Pattern Analyses for the Diagnosis of Melanoma in Special Locations
On the contrary of what happen in other body sites, the application of dermoscopy in facial lesions increases the number of biopsies and in consequence the number of early melanomas identified.
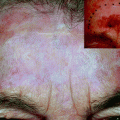
Lesions on the face show specific dermoscopic patterns due to the anatomy of the region, being the classical pattern of melanocytic lesions the pseudo-network.
Early lentigo maligna melanomas (LMMs) may show annular granular pattern (with gray dots around hair follicles), irregular perifollicular pigmentation, short lines, and rhomboidal structures. When lentigo maligna progresses, occlusion of follicular openings and specific criteria of melanoma as blue whitish veil may appear [39].
The histopathological correlation of each dermoscopy criteria of LMM is also well known: the annular granular pattern corresponds to melanophages in papillary dermis and around hair follicles; perifollicular pigmentation corresponds to the proliferation/invasion of atypical melanocytes in the hair follicle; short lines and rhomboidal structures correspond to the proliferation of atypical melanocytes in the dermoepidermal junction; and finally, the occlusion of follicular openings corresponds to the progression of the melanoma and complete invasion of the hair follicle.
Multiple gray dots (granularity) may also be seen in pigmented actinic keratosis and in liken planus-like keratosis (LPLK). Sometimes, in LPLK the granularity is coarser, but in other occasions the diagnosis is just performed after a biopsy. A difficult differential diagnosis with dermoscopy is between early LMM and pigmented actinic keratosis because both may show multiple gray dots, and again the diagnosis is performed by histopathology. Dermoscopy is useful not only to increase the index of suspicion in lesions on the face but also to select the best area to be biopsied to obtain a diagnosis.
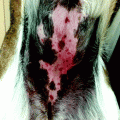
For the diagnosis of melanoma in acral sites, the recognition of parallel ridge pattern [40] (Fig. 13.8) is highly specific, while benign patterns are also well characterized being the more frequent benign pattern the parallel furrow pattern [41]. Dermoscopy is very useful in the recognition of benign patterns and permits to avoid unnecessary excision.


Fig. 13.8
Dermoscopy of a sole showing parallel ridge pattern in a lentiginous acral melanoma
Comparative Recognition for the Diagnosis of Melanoma
In clinical practice, physicians are dealing with patients with multiple lesions and not with isolated lesions. In these situations comparative recognition process is used. Most individuals have a predominant lesion’s pattern; therefore, the examination of all lesions is an essential step in the recognition process because it allows the identification of lesions deviating from the individual’s prevailing benign pattern (concept of the “ugly duckling sign”) [42–45] (Fig. 13.9).
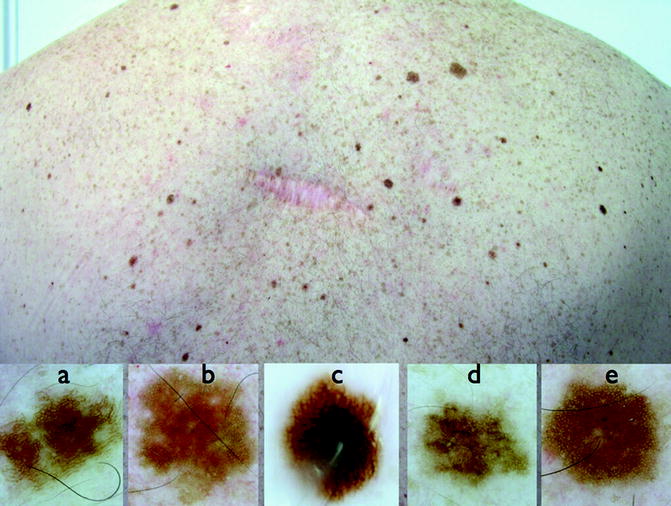

Fig. 13.9
Clinical image (up) of a patient with a previous melanoma. The “ugly duckling sign” of dermoscopy allows the identification of a second melanoma (c) compared with the predominant nevus pattern of the patient (a, b, d, e)
Digital Dermoscopy Follow-Up for the Diagnosis of Melanoma in High-Risk Patients
The importance of surveillance by digital dermoscopy follow-up programs to identify melanoma at an early stage in high-risk populations such as patients with hundreds of nevi, atypical mole syndrome (AMS), personal or family history of melanoma, and carriers of genetic mutations as in CDKN2A has been clearly demonstrated [14, 46] (Fig. 13.10). In a recent study comparing melanomas identified in patients included in digital dermoscopy follow up (DFU) program with melanomas diagnosed in patients referred to a melanoma unit, the multicomponent pattern (defined by the combination of three or more distinctive dermoscopic structures within a given lesion) was noted in 60 % of referred melanomas but in only 16 % of those identified in the DFU surveillance program. By contrast, the reticular pattern was the most frequent pattern observed in melanomas that appeared in patients included in the digital dermoscopic follow-up program [46]. This reticular pattern is the characteristic pattern of slow-growing melanomas described by Argenziano et al. [47] and that may be slow growing in a radial phase for years or decades being diagnosed when this pigment network is altered and a homogeneous structure appears [48] associated with a disruption of the dermoepidermal architecture or the occurrence of regression. The inclusion of high-risk population in follow-up programs allows the detection of melanomas in early stages, with good prognosis (mainly in situ, if not AJCC stage IA), even in the absence of clinical and dermoscopic features of melanoma. In a selected study population, with 90 % of the patients displaying AMS and almost 45 % having had a previous melanoma, one of eight developed melanoma during surveillance, which is more than 1,500 times higher than expected in the general population. According to these results, MM can be diagnosed at any time once a high-risk patient is included in the DFU program. Thus, maintained surveillance is required in individuals at high risk, and the “two-step method of digital follow-up” allows detection of melanomas with few dermatoscopic criteria by digital dermoscopy follow-up (60 % of the new melanomas diagnosed in DFU) and also the detection of melanoma that present as a new lesion or arising from nevi not monitored by dermatoscopy (40 % of melanomas detected in digital follow-up) [14].
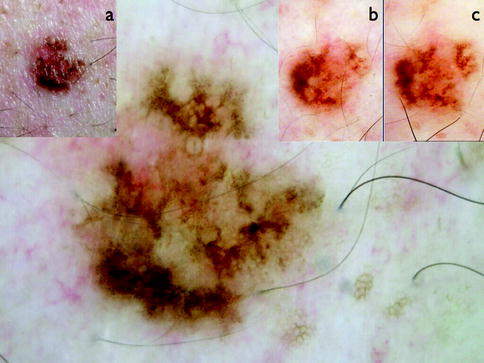

Fig. 13.10
Clinical image (a) of a melanoma diagnosed during digital follow-up (sequential dermoscopy images at 6 months, b and c) showing a focal change in the structure. High-resolution dermoscopy shows a lesion with atypical pigment network and peppering in less than 10 % of the lesion
Pattern Analyses in the Diagnosis of Basal Cell Carcinoma
Classic Pattern
The dermatoscopic model for the diagnosis of the pigmented variant of BCC, initially described by Menzies and co-workers [49], is based on the absence of a pigmented network and the presence of at least one of six positive morphological features including ulceration (not associated with a recent history of trauma), multiple blue-gray globules, maple leaflike areas, large blue-gray ovoid nests, spoke-wheel areas, and arborizing (treelike) telangiectasia [49, 50] (Figs. 13.11 and 13.12). Several publications confirm the reproducibility and reliability of this method [50–52]. Arborizing vessels may also be seen in other conditions such as xanthogranuloma but in this case with a yellowish background [19].