Introduction
- ■
Cancer immunoediting is the process by which malignant cells evade the immune system—first by incomplete elimination of tumor cells during immunosurveillance, followed by an equilibrium phase, and finally by an immune escape phase. The immune escape phenomenon is mediated by a variety of interactions within the tumor microenvironment, among which immune checkpoint pathways play a prominent role. Immune checkpoint axes such as cytotoxic T-lymphocyte–associated protein 4 (CTLA-4)–B7-1/B7-2 and programmed cell death 1 transmembrane protein (PD-1)–PD-L1/PD-L2 are physiological negative regulators of activated T cells that lead to immune tolerance of normal tissues and therefore prevent autoimmunity. Because tumor cells upregulate these pathways to dampen immunosurveillance, inhibiting these axes by utilizing monoclonal antibodies has proved to be a successful therapeutic strategy for a wide range of malignancies.
- ■
The downside to the unbridled immune activation with immune checkpoint inhibitors (ICIs) is that it can also lead to loss of immune homeostasis and result in a broad spectrum of immune-related adverse effects (irAEs). These irAEs can affect a variety of organ systems and bear a resemblance to autoimmune diseases in their clinical presentation. Dermatological irAEs are the most common irAEs reported and can be seen in 37% to 70% (all-grade) of patients treated with ipilimumab and in 17% to 37% of those receiving PD-1 inhibitors. Grade 3 or higher toxicity is seen only in 1% to 3% of these patients. The incidence of cutaneous toxicities also varies according to the underlying malignancy—it appears to be more common in patients with melanoma or renal cell carcinoma in comparison to patients with non–small-cell lung cancer.
There are emerging data to suggest that the development of irAEs is associated with objective tumor responses and improved survival outcomes. , Objective response rates as high as 69% to 75% in patients who developed cutaneous irAEs with ICIs have been reported in the literature. In a meta-analysis of 27 studies of patients with advanced melanoma treated with immunotherapy, the development of vitiligo as an irAE was associated with improved progression-free survival (hazard ratio [HR] 0.51; 95% confidence interval [CI] 0.32–0.82, P < .005) and overall survival (HR 0.25; 95% CI 0.10–0.61; P < .003). It is therefore imperative to treat cutaneous irAEs effectively while minimizing interruption of therapy with ICIs because patients who develop irAEs appear to be more likely to derive benefit from continued treatment.
- ■
In this chapter we describe the various clinical manifestations of cutaneous toxicities of ICIs, their pathophysiology, and approaches for their management and treatment.
Description
- ■
Clinical manifestations: The time to onset of dermatological toxicities with immunotherapeutic agents is quite variable. Whereas some patients may develop cutaneous adverse events within 2 weeks of initiating therapy, others may manifest toxicities several months into their treatment. Despite the broad spectrum of dermatological reactions noted with immunotherapeutic agents, these can be classified into broad categories based on common clinicopathological features. The most commonly observed rashes are usually inflammatory in nature. This category includes maculopapular rashes along with a variety of other less frequent rashes such as lichenoid, acneiform, exfoliative, dermal hypersensitivity-like, psoriasiform, or photosensitivity rashes. The common cutaneous toxicity noted with ICIs is a pruritic maculopapular rash. Maculopapular rashes tend to have an early onset into the treatment course and occur earlier with a combination of CTLA-4/PD-1 blockade than with either agent alone. On examination, characteristic erythematous macules or papules are usually noted on the trunk but usually spare the hands, feet and face. Occasionally, the rash may be more prominent in sun-exposed areas. Rarely, maculopapular rashes may precede more severe cutaneous toxicities, which are described later. Lichenoid rashes are characteristically noted with PD-1/PD-L1 inhibitors rather than with CTLA-4 blockers and tend to present later. Physical examination in such cases shows flat-topped papules with visible striae which may involve the palms and soles and rarely present with mucosal lesions in the oral or genital regions. Psoriasiform dermatological reactions resemble psoriasis in their manifestations and include the typical erythematous plaques covered by silvery white scales, which are preferentially located over the knees, elbows, scalp, or the intertriginous areas. Other less common forms of psoriasis, such as pustular or guttate psoriasis, or even the development of de-novo psoriatic arthritis with immunotherapy have been described. ,
- ■
Inflammatory rashes: Inflammatory rashes also include the potentially life threatening severe cutaneous adverse reactions (SCARs) such as drug reaction with eosinophilia and systemic symptoms (DRESS), Stevens-Johnson Syndrome/Toxic Epidermal Necrolysis (SJS/TEN), and acute generalized exanthematous pustulosis (AGEP).
- ■
DRESS: DRESS is a clinical syndrome in which patients develop a skin eruption accompanied by significant eosinophilia and organ involvement after exposure to a culprit drug. Patients often present with a morbilliform rash which progresses to a confluent erythematous rash with follicular accentuation, typically involving greater than 50% of the body surface area (BSA) and often with facial involvement. Patients have other systemic symptoms such as fever, malaise, lymphadenopathy, and other symptoms related to underlying organ dysfunction. Laboratory workup usually reveals significant eosinophilia, atypical lymphocytosis, and abnormal liver or renal function tests. Some patients may also have pulmonary involvement in the form of interstitial pneumonitis with or without pleural effusions.
- ■
AGEP: AGEP is a drug reaction characterized by the development of diffuse sterile pustules usually on top of an erythematous rash and is accompanied by systemic symptoms such as fever. The rash usually starts in the face or intertriginous areas and spreads to the trunk or limbs. Mucosal surfaces are usually spared. Patients may have facial edema and fever at presentations. Laboratory evaluations are remarkable for leukocytosis with a neutrophilic predominance.
- ■
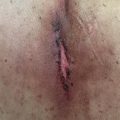
SJS and TEN: SJS and TEN are severe cutaneous adverse effects which are a consequence of extensive keratinocyte necrosis in reaction to immunotherapy, leading to detachment of the epidermis. Based on the percentage of BSA involved, the reaction is either categorized as SJS (<10% of BSA) or TEN (>30% BSA). Patients with 10% to 30% BSA involvement have an SJS/TEN-overlap syndrome. Patients may present with a prodrome consisting of fever, flu-like symptoms, photophobia, conjunctival itching, erythematous rash, myalgias, or arthralgias. On examination, patients usually have a painful erythematous rash in the involved areas which may evolve into vesicles or bullae with time. The palms and soles tend to be spared in this condition. A positive Nikolsky sign, in which superficial sloughing of the skin can be provoked by applying lateral pressure to the surface of apparently uninvolved skin, is classically positive. Mucosal involvement is also typical, manifesting as painful erosions in the buccal mucosa and, occasionally, with severe conjunctivitis, odynophagia, or genital erosions. Due to massive fluid and electrolyte losses and the catabolic state associated with this disorder, electrolyte imbalances as well as hypoalbuminemia are commonly noted upon laboratory evaluation. These patients are at risk of bacterial superinfection of their skin lesions and must be carefully monitored and treated for the same.
- ■
- ■
Rashes resembling autoimmune conditions: Another broad category of cutaneous reactions includes rashes that resemble autoimmune dermatological conditions. Bullous pemphigoid-like reactions have been reported with PD-1/PD-L1 inhibitors but have not been reported with CTLA-4 inhibitors. The onset of blistering may be heralded by pruritis or a mild erythematous rash. On physical examination, patients often have a variable number of tense bullae which can rupture leading to moist erosions. These blisters are more commonly noted on the trunk or intertriginous areas and rarely involve mucosal surfaces. These lesions may also persist even after discontinuation of the offending immunotherapeutic agent. Other reactions that resemble autoimmune conditions include dermatomyositis (serological workup may be negative for anti-Jo1 antibodies), vasculitis, and Sjogren’s or sicca syndrome.
- ■
Vitiligo: Other rare cutaneous toxicities as a result of alteration of melanocytes include vitiligo, regression of melanocytic nevi, and tumoral melanosis. The higher incidence of vitiligo in patients with melanoma is likely a consequence of shared antigens among melanoma cells and melanocytes. Vitiligo associated with immunotherapeutic agents is usually symmetric in distribution and tends to be irreversible.
- ■
Other dermatological toxicities: Other infrequently observed toxicities include alopecia, re-pigmentation of hair, and dystrophic nails. ,
Diagnostic Workup
- ■
Diagnostic workup for all patients should include thorough physical examination, including close attention to examination of mucosal surfaces. In addition, history of concomitant medications, travel, or other environmental exposures which could point towards an alternative etiology must be obtained. Routine laboratory evaluation, including a complete blood count and comprehensive metabolic panel, may be obtained for patients with higher grade toxicities. Peripheral eosinophilia has been noted in some patients with cutaneous immune related adverse effects.
- ■
Dermatological toxicity of ICIs is a clinical diagnosis; however, a biopsy may aide diagnosis in cases where the etiology is not clear or in cases that do not respond to initial therapy. A majority of patients with cutaneous adverse effects of ICIs will have a lichenoid or interface pattern on histology. This is characterized by a dense lymphocytic infiltrate that abuts or obscures the dermoepidermal junction. In patients with maculopapular rashes with ICIs, a perivascular T cell infiltrate, along with varying degrees of eosinophilic infiltrate, may be observed. In patients with bullous pemphigoid, direct immunofluorescence shows immunoglobulin (Ig) G and complement 3 deposits at the basal membrane. Serum enzyme-linked immunosorbent assay (ELISA) for the pathognomonic antibodies to BP180 and BP230 can be performed to confirm the diagnosis. In patients where an autoimmune condition is suspected, workup for autoantibodies including antinuclear antibody, SS-A/Anti-Ro, SS-B/Anti-La (if predominantly photodistributed/photosensitivity), antihistone, and double-stranded DNA antibodies, is indicated.
Agents and Mechanisms of Action
- ■
Immune checkpoint inhibitors: Dermatological toxicities are the most commonly reported adverse effect with ICIs. These are a consequence of the dysregulation of immune tolerance mediated by physiological immune checkpoints resulting in unrestrained infiltration of activated T cells in affected organs. Although cutaneous toxicities have been observed with blockade of both the PD-1/PD-L1 pathway and the CTLA-4 pathway, there are some subtle differences in some of the clinical manifestations with agents belonging to each of those classes. For example, bullous pemphigoid-like reactions have been reported with PD-1/PD-L1 inhibitors but not with CTLA-4 inhibitors. Cutaneous toxicities are observed more frequently with CTLA-4 blockade and with combination therapy of a PD-1 and CTLA-4 inhibitor than with a PD-1 inhibitor alone.
- ■
Cytokines: A self-limiting pruritic erythematous macular rash is seen in most patients who receive high-dose interleukin-2 for the treatment of melanoma or renal cell carcinoma. However, in a subset of patients this reaction may be severe and may resemble one of the SCARs, such as TEN.
- ■
Chimeric antigen receptor (CAR) T-cells: A skin rash may be observed in 8% to 16% of patients treated with CAR T cells for refractory hematological malignancies.
- ■
Oncolytic viruses: Rare side effects observed with talimogene laherparepvec (T-VEC), which is the most commonly used oncolytic virus, include rash, cellulitis, vitiligo, and exacerbation of psoriatic lesions.
Pharmacological Approaches for Management and Treatment The severity of dermatological toxicity should be measured in accordance with the National Cancer Institute’s Common Terminology Criteria for Adverse Events (NCI-CTCAE) which takes into account both the extent of the lesions and the impact on activities of daily living. Although most cutaneous adverse effects resolve with conservative management or interruption of therapy, close attention must be paid to the physical examination for all patients because innocuous clinical presentations such as pruritis or maculopapular rash may precede other potentially life-threatening SCARs.
Withholding immune checkpoint inhibitors For most cutaneous adverse effects that are grade 3 or higher, ICIs must be placed on hold at least until the toxicity is reduced to grade 1 or less. It is prudent to involve a dermatologist in the decision-making prior to rechallenging patients with higher grade cutaneous toxicities. For patients with grade 1 inflammatory rashes, ICIs may be continued; however, for those with grade 2 toxicity, consideration should be given to withholding ICIs until the severity is less than grade 1. On the other hand, for any patient with bullous dermatoses and greater than 10% BSA involvement, ICIs should be withheld, and may be permanently discontinued for involvement of greater than 30% BSA associated with fluid or electrolyte abnormalities. Similarly, for any patient with suspected SCAR, ICIs should be withheld immediately. A SCAR is usually considered to be a contraindication for rechallenging patients with ICIs.
■
Immunosuppression:
Immunosuppression and other supportive measures form the backbone of treatment for immune-mediated cutaneous toxicities. Supportive care includes antihistamines and emollients for symptomat relief. For lower grade (1 and 2) inflammatory rashes, patients should first be treated with mild and moderate potency topical corticosteroids. If there is no improvement with this regimen, high-potency topical steroids should first be started along with oral prednisone (1 mg/kg/day), which should be tapered gradually over 4 weeks. For higher grade (3 or 4) toxicity, intravenous methylprednisolone (1–2 mg/kg/day) may be initiated and followed by a slow steroid taper upon clinical improvement. Steroid-sparing agents such as infliximab, mycophenolate mofetil, or cyclophosphamide may be used for steroid-dependent or refractory cases.
- ■
For patients with grade 2 bullous dermatoses (blisters affecting 10%–30% of BSA), high- potency topical steroids should be initiated. If there is no improvement, early initiation of 0.5–1 mg/kg/day of prednisone should be considered. For grade 3 or higher bullous dermatoses, IV methylprednisolone (1–2 mg/kg) should be initiated with a taper over at least 4 weeks. Rituximab has been used successfully for the management of bullous dermatoses and may allow for shorter courses of steroids.
- ■
For patients with severe cutaneous adverse reactions, topical treatments such as emollients, medium- to high-potency topical corticosteroids, and oral antihistamines should be initiated. Any patient with skin sloughing should be considered to have at least grade 3 toxicity and should be treated with 0.5–1 mg/kg/day of methylprednisolone. For grade 4 SCARs (sloughing/blisters involving >30% BSA), higher doses of methylprednisolone (1–2 mg/kg/day) should be considered. Intravenous immunoglobulin or cyclosporine may be used in severe or refractory cases.
Nonpharmacological Approaches for Management and Treatment
- ■
For any patient with an immune-mediated cutaneous adverse effect, skin care including topical emollients and the use of sunscreens and protective clothing to avoid sun exposure is advised. For patients with denuding cutaneous reactions (bullous dermatoses or SJS/TEN), local wound care is crucial in avoiding complications such as bacterial superinfections. For bullous dermatoses, petroleum ointment or bandages may be used over open erosions. Any patient with a grade 3 or higher SCAR should be admitted to a burn unit for intensive monitoring and treatment of associated fluid and electrolyte imbalances. In addition, aggressive wound care and prompt recognition and treatment of bacterial infections is an integral part of the management of these patients. Pain management and involvement of other specialties such as ophthalmology, urology, etc., depending on the mucosal surfaces involved, may be warranted.
- ■
In conclusion, although most cutaneous adverse effects of immunotherapeutic agents are easily managed with a conservative approach and brief interruption of therapy, careful attention to the history and physical examination must be paid to identify patients with more severe forms of reactions that could result in significant morbidity and mortality.
References
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree