15317
Colorectal Cancer
Armin Shahrokni
INTRODUCTION
Every year about 140,000 patients are diagnosed with colorectal cancer (1). Older cancer patients are disproportionally affected by it. About 29% and 43% of deaths related to colorectal cancer occur in men and women older than age 80, respectively (2). Although individuals older than age 65 are minority compared to the whole population, 60% of colorectal cancer cases are diagnosed in this age group (2). Although the incidence and mortality from colorectal cancer have decreased in the aging population, due to colorectal cancer screening and surgical treatment of localized cancer, the number of older colorectal cancer patients requiring treatment still remains very high (2).
Treatment of older colorectal cancer patients presents different challenges than treatment of younger patients. In general, as patients become older, they develop more comorbid conditions, take more medications, lose their independence, and become more socially isolated. As a result, applying standard of care without addressing these issues may cause more harm rather than achieving benefit.
In this chapter, we briefly discuss the concept of frailty as it impacts chemotherapy decision making, and review some of the most common challenges that may be faced by medical oncologists taking care of older colorectal cancer patients.
INTERSECTION OF FRAILTY AND AGING-RELATED PHYSIOLOGICAL CHANGES (FIGURE 17.1)
■ Geriatric assessment (GA) is a multidimensional assessment of older patients that captures their functional activity, psychosocial well-being, cognitive function, polypharmacy, and nutritional status.
■ Patients with more deficits based on GA are considered to be frail.
■ Less time-consuming screening versions of GA are available (3).
■ Cancer treatment decision making will be based on GA and organ function of older colorectal cancer patients.
ADJUVANT TREATMENT FOR LOCALIZED COLORECTAL CANCER (FIGURE 17.2)
1. Assessment of frailty/fitness: Various definitions and screening tools for fitness/frailty assessment exist. In a broad definition, a fit patient is a patient with no or just one 154comorbid condition who is independent in basic and instrumental activities of daily living (see Chapters 3 and 13).
2. Assessment of patient’s life expectancy: Older patients are more likely to die from noncancer causes than younger patients (4). Consider administering adjuvant chemotherapy in patients with life expectancy of greater than 5 years or in those with high lymph node involvement. A comorbidity-adjusted life expectancy table (5) is useful in this assessment. Other useful methods for calculating life expectancy are the Schonberg mortality index (6), and e-prognosis (7).
3. Chemotherapy toxicity calculators for older adults with cancer: At least two chemotherapy toxicity prediction models have been validated among older cancer patients. In the Cancer and Aging Research Group (CARG) calculator (8), the toxicity is assessed by aging-related deficits, hemoglobin and kidney function, and the number of chemotherapy agents. In the Chemotherapy Risk Assessment Scale for High-Age Patients (CRASH) calculator (9), the risk is assessed by aging-related deficits, diastolic blood pressure, lactate dehydrogenase (LDH), and the toxicity risk associated with certain chemotherapy agents and regimens. The CRASH calculator estimates hematological and nonhematological toxicity risk, whereas the CARG calculator provides only one risk score.
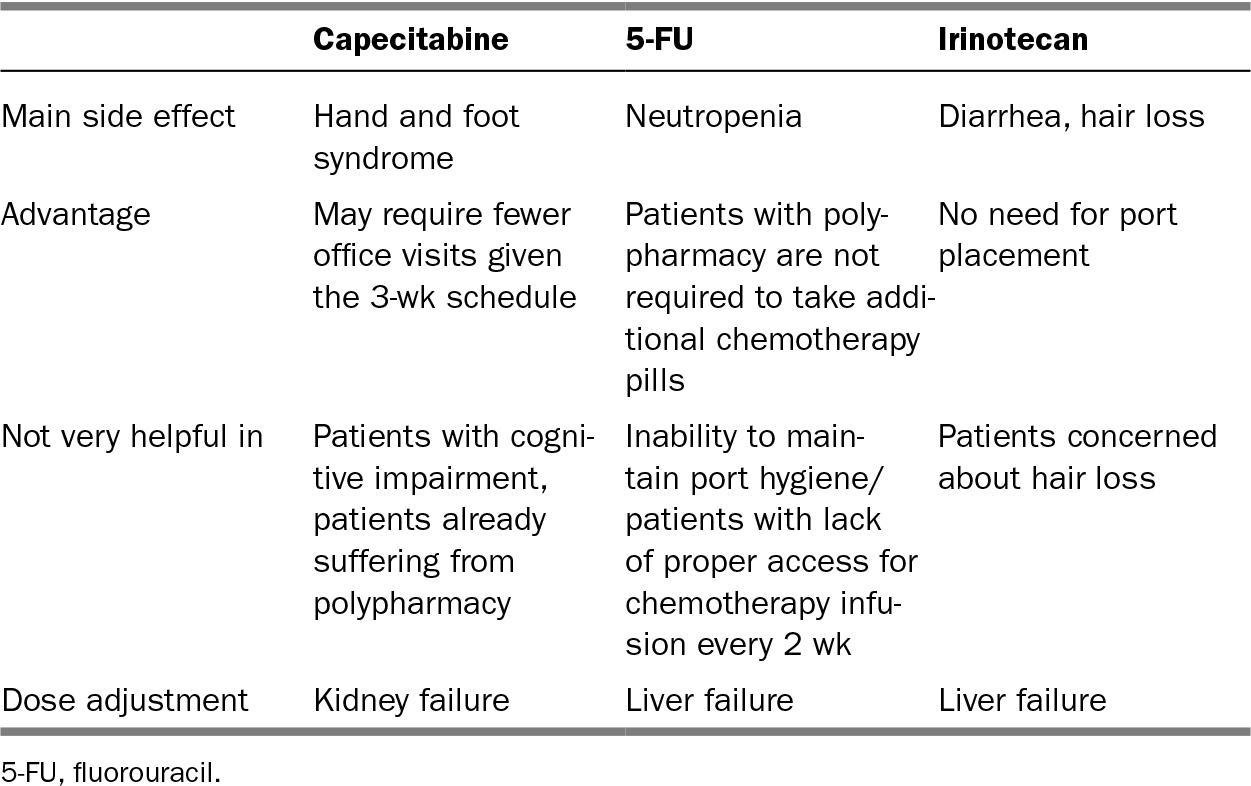
4. Capecitabine versus fluorouracil (5-FU): A—5-FU and capecitabine have similar efficacy in improving patient outcomes (10). B—The decision for choosing 5-FU versus capecitabine should be based on their toxicity profiles and ease of use based on the patient’s overall status. C—About 30% of patients only receive 1 to 4 months 155of adjuvant chemotherapy (11). These patients are two times more likely to die compared to those who complete the adjuvant chemotherapy. This emphasizes the importance of the proper selection of older colon cancer patients for adjuvant chemotherapy, proper decision making on single versus doublet chemotherapy and 5-FU versus capecitabine, and providing support for older colon cancer patients.
5. Use of oxaliplatin: A—oxaliplatin has limited benefits in improving outcomes of older patients with colon cancer. The probability of 5-year survival was 75.8% for patients who received 5-FU and oxaliplatin compared to 76.1% for patients who only received 5-FU/Leucovorin (LV) (12). B—Despite lack of solid benefit, in the community, 50% of patients age 70 to 79, 33% of patients age 80 to 84, and 13% of patients older than age 85 have received oxaliplatin as a part of their adjuvant treatment (13). C—Strongly consider omitting oxaliplatin in patients with diabetes and/or diabetic neuropathy, with history of multiple falls, or with other sensory deficits (e.g., poor vision).
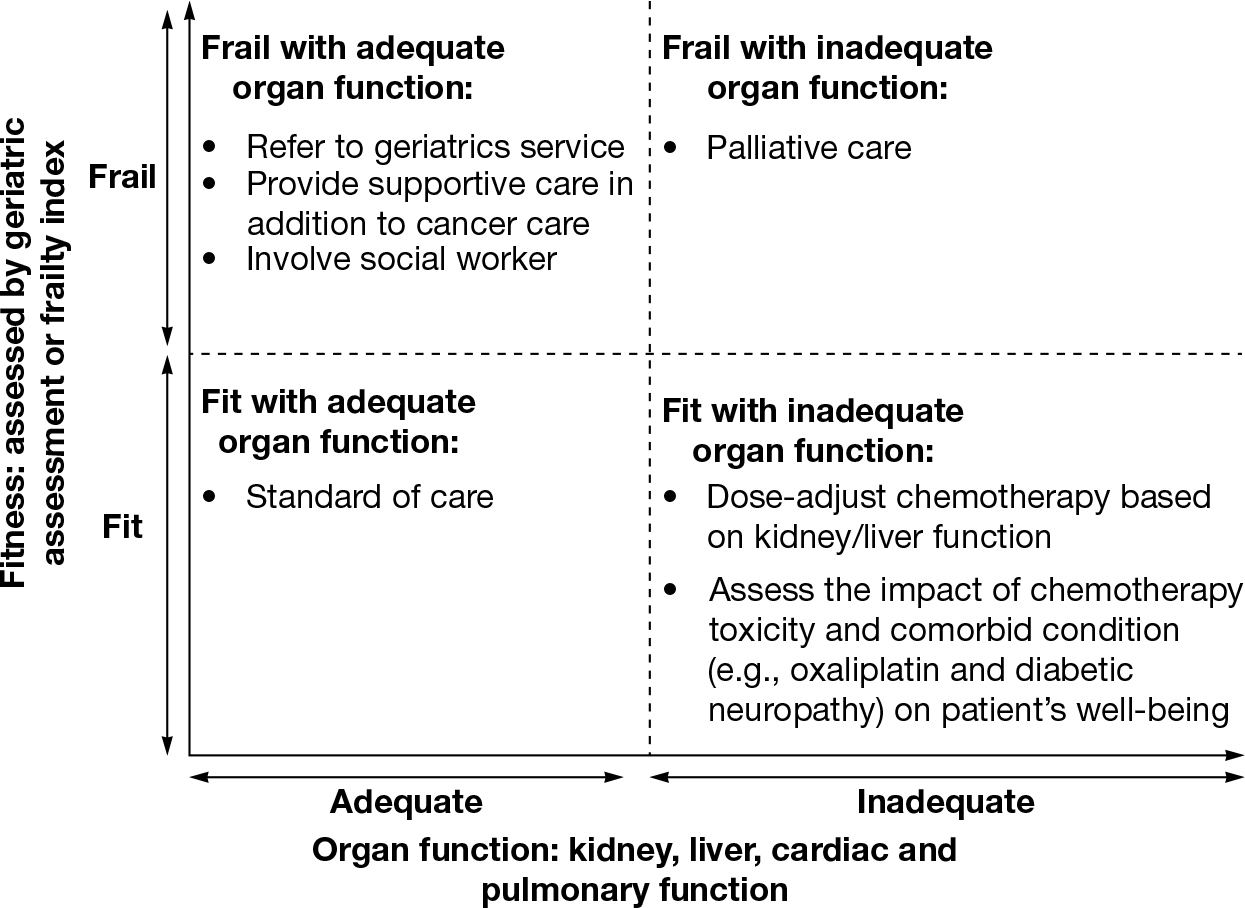
FIGURE 17.1 The impact of frailty and organ dysfunction on treatment decision making.
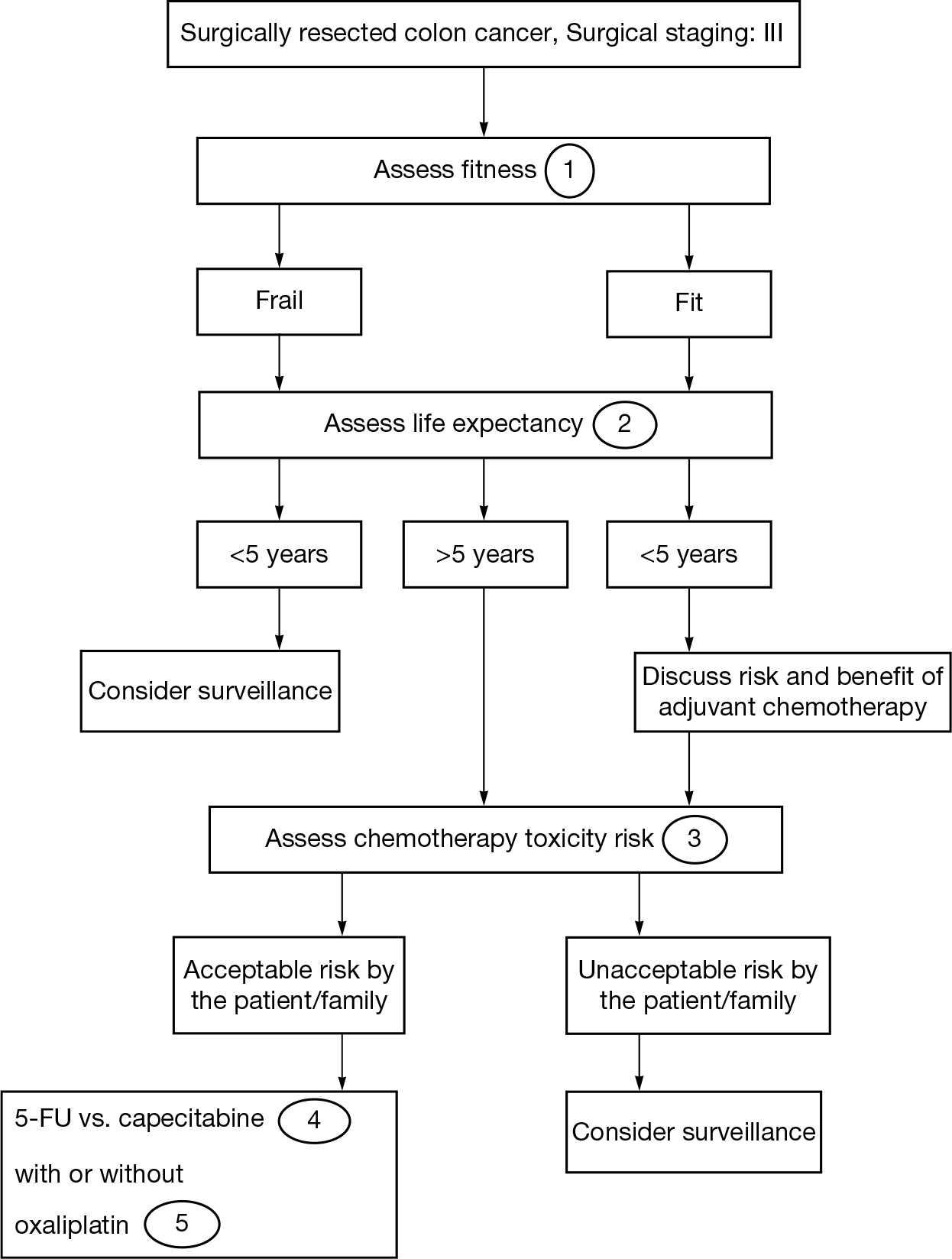
FIGURE 17.2 Treatment decision making for older cancer patients with localized colorectal cancer.
5-FU, fluorouracil.
156METASTATIC COLORECTAL CANCER; TREATMENT DECISION MAKING (FIGURE 17.3)
1. Refer to chemotherapy toxicity calculators for older adults with cancer in the previous section.
2. For selecting between single-agent 5-FU versus capecitabine or irinotecan, refer to Table 17.1.
3. For making decisions on administering oxaliplatin, refer to point 5 of the previous section.
4. One study showed that among patients unfit for standard-dose chemotherapy, starting with 80% dose-reduced chemotherapy is an acceptable option leading to less toxicity and no significant change in overall survival (14).
1575. Two studies showed that stopping treatment after 12 weeks of treatment, and restarting the treatment after the disease progressed, does not impact patients’ outcome and may lead to better quality of life (15,16).
6. A study on older patients with metastatic colon and rectal cancer who received treatment between the years 2000 and 2009 showed that the addition of three or more lines of treatment led only to an additional month of survival for patients older than age 75. For those 65 to 74 years of age, the increased survival was 6 months for colon cancer patients and 10 months for rectal cancer patients (17).
7. Adding bevacizumab to the first line of treatment leads to an increase in progression-free survival of approximately 3 months in older patients with metastatic colorectal cancer. Patients receiving bevacizumab are at higher risk for arterial thromboembolic events, gastrointestinal perforation, slower wound healing, and increase in blood pressure (18). As a result, it is important to evaluate comorbid conditions of older patients with metastatic colorectal cancer very carefully. Cetuximab and panitumumab are antibodies toward epidermal growth factor receptors. Patients with metastatic colorectal cancer with wild-type V-Ki-ras2 Kristen rat sarcoma (KRAS) status benefit from addition of cetuximab or panitumumab to combination chemotherapy (19,20). For those who progress on previous lines of chemotherapy, cetuximab or panitumumab can be used as single agent if patients have not received them before. In this setting, their benefit is about prolonging life by about 1.5 months (21) or not at all, partially explained by a crossover study (22). These agents can cause skin rash, diarrhea, and fatigue (23). In our experience, we use triplet treatment as the first line of treatment for metastatic colorectal cancer patients if they are fit and have low volume of metastatic disease; surgery is considered curative following chemotherapy.
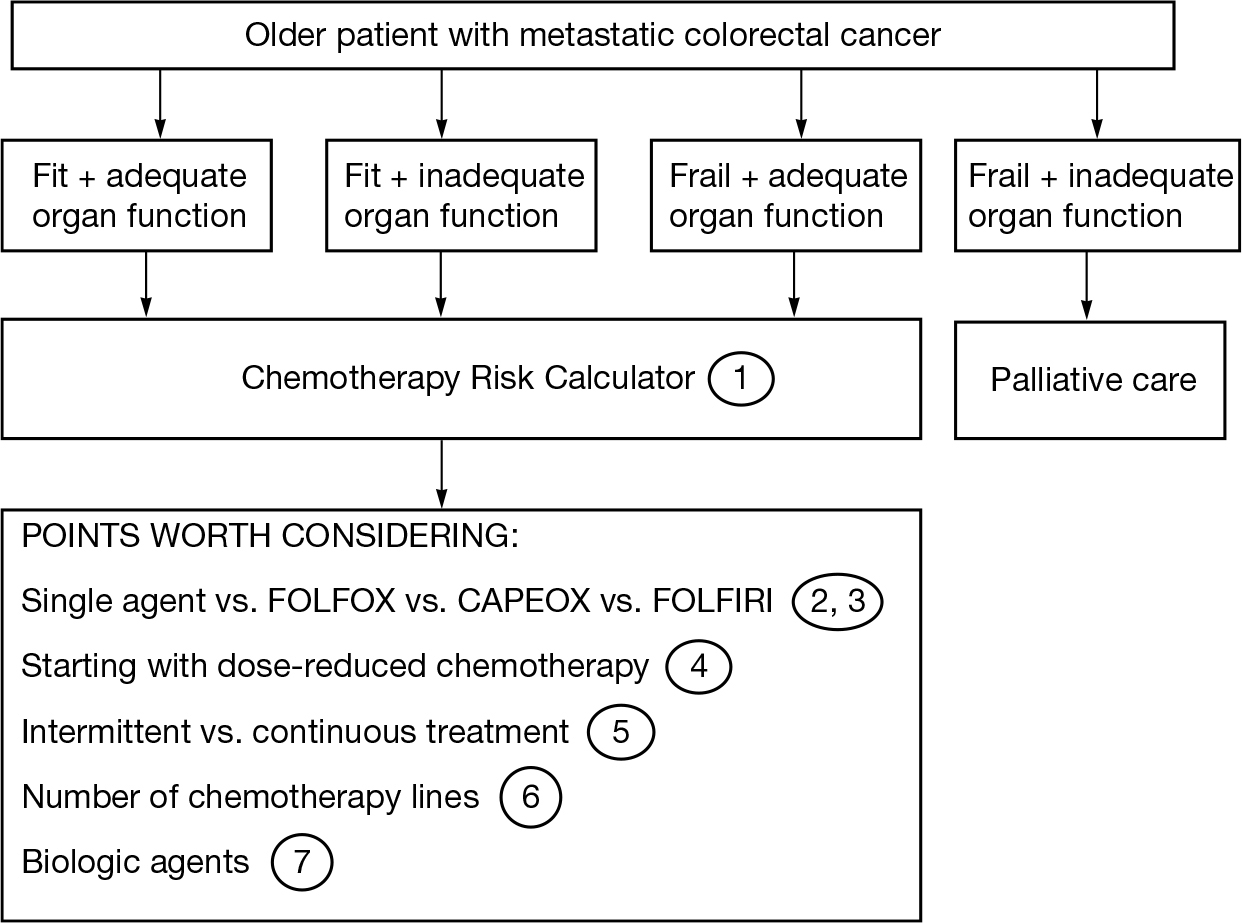
FIGURE 17.3 Treatment decision making for older patients with metastatic colorectal cancer.
TABLE 17.1 Barriers and Benefits of Capecitabine, 5-FU, and Irinotecan
TAKE HOME POINTS
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree