Colon Cancer
EPIDEMIOLOGY
 STATISTICS
STATISTICS
In the United States, 102,480 new cases of colon cancer were expected in 2013 (men 50,090; women 52,390) (1), and 40,340 new cases of rectal cancer were expected in 2013 (men 23,590; women 16,750). Colorectal cancer is the second leading cause of cancer-related death in the United States with 50,830 deaths annually. Age is a major risk factor in developing colon cancer. The lifetime risk of developing colorectal cancer is approximately 5% with the vast majority of cancers occurring after age 50 years. The overall incidence has been falling perhaps due to screening.
 EPIDEMIOLOGIC ASSOCIATIONS
EPIDEMIOLOGIC ASSOCIATIONS
The vast majority of colorectal cancers are sporadic and not familial. Epidemiologic studies demonstrate an increased risk of colorectal cancer with the following conditions/characteristics:
• Family history of colorectal cancer is associated with an increased risk of developing colorectal cancer. If one first-degree family member had colorectal cancer, the risk increases 1.7-fold
• Western/urbanized societies
• Diet high in red or processed meat
• Increased bowel anaerobic flora
• Diabetes mellitus/insulin resistance: the risk of colon cancer may be 30% higher in diabetics compared with nondiabetics
• Inflammatory bowel disease. Increased incidence is seen with both Crohn’s disease and ulcerative colitis and is associated with the severity, extent, and duration of disease affecting the colon. The risk of colon cancer in ulcerative colitis is approximately 10% at 10-year duration, 20% at 20-year duration, and >35% at 30-year duration. Total colectomy eliminates the risk of colon cancer
• Cigarette smoking
• Alcohol consumption
• Ureterosigmoidostomy
• Streptococcus bovis bacteremia
• Prior pelvic radiation
 INHERITED SYNDROMES
INHERITED SYNDROMES
Fewer than 10% of colon cancers are known to be associated with an inherited predisposition to colon cancer. The most common inherited syndromes are FAP and HNPCC. The MYH gene mutations are associated with an inherited predisposition to colon cancer as well (2).
Familial Adenomatous Polyposis (FAP)
Most cases of FAP are due to mutations in the APC gene on chromosome 5q21. These mutations are inherited in an autosomal dominant fashion.
APC is a tumor suppressor gene whose product interacts with critical cell proliferation genes in part by its interaction with transcription factor, beta catenin.
FAP is associated with hundreds to thousands of polyps throughout the colon. Fewer polyps and a later onset of colorectal cancer characterize an attenuated form of FAP. The use of COX-2 inhibitors can result in regression of some polyps.
By age 10 years, 15% of carriers will have adenomas; by age 20 years, 75% will have adenomas; and by age 30 years more than 90% will have adenomas. Screening of first-degree relatives should be done by age 10 years. Treatment is a total proctocolectomy.
FAP accounts for <1% of colon cancers and is associated with congenital hypertrophy of the retinal pigment, desmoid tumors (Gardner’s syndrome), and brain tumors (Turcot’s syndrome).
Hereditary Nonpolyposis Colon Cancer (HNPCC)
HNPCC is due to mutation in mismatch repair genes (e.g., MLH1, MSH2), which leads to microsatellite instability and errors in DNA replication. Inherited in an autosomal dominant fashion, HNPCC may account for up to 6% of all colon cancers. The median age for development of colon cancer is less than 50 years. Right-sided tumors are much more common than left-sided tumors. HNPCC is associated with endometrial cancer, ovarian cancer, upper gastrointestinal cancers, and transitional cell cancers of the renal pelvis/ureter.
An individual is likely to belong to a family with HNPCC and require genetic testing if: (1) three or more relatives have had colon cancer (or another cancer associated with HNPCC such as uterine, small bowel, urethral, or renal pelvic cancer) and at least one of the relatives is a first-degree relative, (2) two or more generations of the family have colon cancer, or (3) one or more relatives were diagnosed with colon cancer before age 50 years. Screening should begin by age 21 years in affected patients and be done at least every 5 years thereafter.
These criteria for identifying HNPCC are referred to as the Amsterdam II Criteria. The Bethesda criteria modify the Amsterdam II Criteria to include in the evaluation those patients who have had family members with adenomatous colonic polyps in addition to colon cancers.
MYH
MYH is a base excision repair gene located on the short arm of chromosome 1. Homozygous mutations in MYH have been associated with a syndrome manifesting itself as multiple colonic polyps and colorectal cancer. It is inherited in an autosomal recessive fashion. MYH mutations are thought to account for less than 1% of colorectal cancers.
PRIMARY PREVENTION AND SCREENING
 PREVENTION STRATEGIES
PREVENTION STRATEGIES
Nonsteroidal anti-inflammatory drugs (NSAIDs), calcium, folate, and estrogens prevent the development of polyps, but there is no clear prevention of cancer. Their role is unknown in the patient who is getting adequately screened (3).
Antioxidants do not prevent colon cancer and conflicting data exist for the preventive ability of calcium, vitamin D, and statins. Diets high in fiber do not prevent colon cancer and diets high in red/processed meat and low in fish have been associated with an increased risk of colon cancer. However, physical activity may have a protective effect.
 SCREENING
SCREENING
Most colorectal cancers arise from adenomatous polyps. The progression of adenomatous polyps from small polyps, to larger polyps, to dyspastic polyps, and finally to cancer occurs over at least a 10-year period. Villous adenomas have a higher rate of progression to colon cancer than tubular or hyperplastic polyps. This progression is due to a series of acquired mutations as outlined by the work of Bert Vogelstein. It is generally felt that only 1% of polyps will progress through this sequence to form a cancer. This progression is due to a series of acquired mutations and is often referred to as the Vogelgram model after Bert Vogelstein, who initially described these events (Figure 47-1). The purpose of screening is to detect polyps before they turn into cancer. Guidelines for screening take into account the effectiveness, sensitivity, specificity, cost, and morbidity of the test (4).
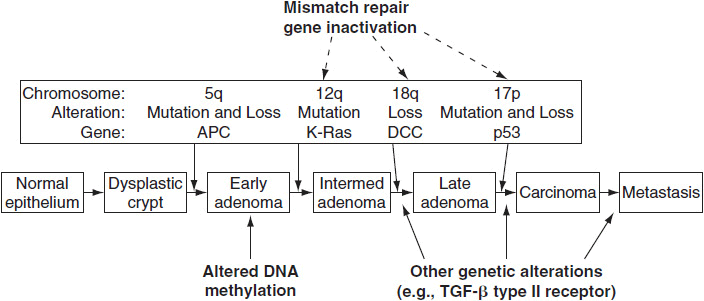
FIGURE 47-1 Vogelgram model of colon carcinogenesis.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree