Chapter Outline
Common presentations of haematological diseases 497
Initial screening tests 497
Specific tests for common haematological disorders 503
Classification of haematological neoplasms 505
Classification of acute myeloid leukaemia and related neoplasms 505
Classification of the myelodysplastic syndromes 506
Classification of acute lymphoblastic leukaemia 507
Classification of myeloproliferative neoplasms and related conditions 508
Common presentations of haematological diseases
An abnormal blood count or blood cell morphology does not necessarily indicate a primary haematological problem because it may reflect an underlying nonhaematological condition or may be the result of therapeutic interventions. Anaemia occurs in many conditions, but a primary blood disease should be considered when a patient has splenomegaly, lymphadenopathy, a bleeding tendency or thrombosis and/or nonspecific symptoms characteristic of leukaemias and lymphomas such as malaise, sweats or weight loss.
As with any clinical problem, the first steps in determining the diagnosis include obtaining a careful clinical, travel and drug history and a thorough physical examination. The result of these, in combination with the patient’s age, sex, ethnic origin, social and family history and knowledge of the locally prevalent diseases, will determine subsequent laboratory investigations.
Initial screening tests
Although the range of haematological tests available to support clinical and public health services is broad, it is often the simplest investigations that are most useful in indicating the diagnosis. Even poorly resourced laboratories are usually able to provide an initial panel of tests such as haemoglobin concentration (Hb), white blood cell count (WBC) and platelet count ( Chapter 26 ) and examination of a peripheral blood film for a differential leucocyte count ( Chapter 3 ) and cellular morphology ( Chapter 5 ). These screening tests will often enable the underlying pathological processes to be suspected promptly and point to a few key diagnostic tests.
Interpretation of screening tests
Results of laboratory screening tests should always be interpreted with an understanding of the limitations of the tests and the physiological variations that occur with sex, age, ethnic group and conditions such as pregnancy and exercise. Physiological variations in cell counts are detailed in Chapter 2 . Abnormalities of red cells, white cells or platelets may be quantitative (increased or reduced numbers) or qualitative (abnormal appearance and/or function).
Quantitative abnormalities of blood cells
Increased numbers of cells
Increases affecting more than one cell line
A simultaneous increase in the cells of more than one cell line suggests overproduction of cells originating in an early precursor cell. This occurs in myeloproliferative neoplasms in which one cell type may predominate, e.g. platelets in essential thrombocythaemia and red cells in polycythaemia vera.
Erythrocytosis
Patients with a persistently (e.g. > 2 months) raised venous haematocrit (Hct) (> 0.52 males, > 0.48 females) should be assessed to determine the cause. Erythrocytosis can be relative or absolute and, if absolute, primary or secondary.
Relative: normal total red cell volume with reduced plasma volume (e.g. dehydration).
Absolute: males and females with Hct values above 0.60 and 0.56, respectively, can be assumed to have an absolute erythrocytosis and do not require confirmatory studies. However, the reason for the elevation of the Hct must still be elucidated.
Primary: this is usually polycythaemia vera (PV), part of the spectrum of myeloproliferative neoplasms. The mutation JAK2 V617F is present in approximately 95% of patients with PV and mutations affecting exon 12 occur in many of those who lack the V617F mutation.
Secondary: to chronic hypoxia (e.g. chronic lung disease, congenital heart disease, high-affinity haemoglobins) or aberrant erythropoietin production. Secondary polycythaemia can generally be excluded by the clinical history and examination, assessment of serum erythropoietin concentration and arterial oxygen saturation, haemoglobin electrophoresis or high performance liquid chromatography (HPLC) plus an oxygen dissociation curve and abdominal ultrasound examination. If initial screening tests are negative for a JAK2 mutation and there is no obvious secondary cause for the high Hct, then red cell mass studies are indicated ( Chapter 17 ).
Leucocytosis
Neutrophilia
Neutrophils are commonly increased during pregnancy and in acute infections, inflammation, alcohol intoxication, corticosteroid therapy and acute blood loss or red cell destruction. Additional findings on the full blood count can be helpful to identify the cause of neutrophilia. The combination of anaemia and neutrophilia occurs in chronic infection or inflammation, and also in malignant conditions; a high Hct with neutrophilia suggests polycythaemia vera. Neutrophilia with an increased platelet count occurs in infectious or inflammatory processes or malignant conditions and during marrow recovery. Neutrophilia with thrombocytopenia is classically seen in sepsis and occasionally in microangiopathic haemolytic anaemia. Examination of the peripheral blood film also provides additional clues to confirm or exclude a particular diagnosis. For example, neutrophilia with the neutrophils showing heavy cytoplasmic granulation (‘toxic’ granulation) is a common finding in severe bacterial infections. In the absence of any underlying cause, a high neutrophil count with immature myeloid cells suggests chronic myelogenous leukaemia (CML), and cytogenetic and molecular studies to look for t(9;22) and the BCR–ABL1 fusion gene are indicated ( Chapter 8 ).
Lymphocytosis
Lymphocytosis is a feature of certain infections, particularly infections in children. It may be especially marked in pertussis, infectious mononucleosis, cytomegalovirus infection, infectious hepatitis, tuberculosis and brucellosis ( Table 23-1 ). Elderly patients with lymphoproliferative disorders, including chronic lymphocytic leukaemia and lymphomas, often present with lymphadenopathy and a lymphocytosis. Morphology and immunophenotyping of the cells combined with histological examination of a bone marrow trephine biopsy specimen (and if necessary other tissue biopsy) are used to classify these disorders and to give an indication of management and prognosis. If lymph nodes are enlarged, a lymph node biopsy for histology and immunohistochemistry may be helpful in diagnosis. It is occasionally difficult to differentiate between a reactive and a neoplastic lymphocytosis. In this situation, immunophenotyping, to provide evidence of light chain restriction and polymerase chain reaction for immunoglobulin or T-cell receptor gene rearrangements, may indicate the presence of a monoclonal population of lymphocytes, thereby supporting a diagnosis of neoplastic, rather than reactive, lymphoproliferation.
| Infections |
|
| Stress and Postsplenectomy |
| Smoking |
| Hypersensitivity Reactions |
| Autoimmune Disorders |
| Thymoma |
| Clonal |
|
Monocytosis
A slight to moderate monocytosis may be associated with some protozoal, rickettsial and bacterial infections including malaria, typhus and tuberculosis. Monocytosis associated with neutrophilia is suggestive of chronic myelomonocytic leukaemia. High levels of monocytes (monocyte count > 1 × 10 9 /l) in an elderly patient suggest chronic myelomonocytic leukaemia or sometimes, atypical chronic myeloid leukaemia. These conditions fall into the myelodysplastic/myeloproliferative neoplasm group of disorders, so the diagnosis is supported by finding splenomegaly, quantitative and qualitative abnormalities in other cell lines or a clonal cytogenetic abnormality.
Eosinophilia
Eosinophilia is typically associated with parasitic infections, skin diseases and allergic disorders. Eosinophils have a tendency to infiltrate and damage tissues such as the heart, lungs and gut, so in patients with eosinophilia, assessment of these organs is necessary. In most cases, the cause of the eosinophilia is indicated by the clinical history, which should include details of all medications and foreign travel, and by examination of the stool and urine for parasites, cysts and ova. Other causes of eosinophilia are given in Table 23-2 .
| Parasitic Infections – Especially with Helminths |
| Neoplastic Diseases |
|
| Allergic Disorders |
|
| Immunodeficiency Disorders |
|
| Connective tissue/rheumatology disorders |
Basophilia
Basophilia as an isolated finding is unusual. However, it is a common feature of myeloproliferative neoplasms, and basophils may be particularly prominent in CML. In this condition, an increasing basophil count may be the first indication of accelerated phase disease. Endocrinopathies such as myxoedema and oestrogen abnormalities, infections and allergic diseases – and rarely, other haematological malignancies – can also cause basophilia.
Thrombocytosis
Thrombocytosis can be primary or secondary (reactive) to surgery, infectious and inflammatory conditions, hyposplenism, blood loss and malignancy, and can occur as a rebound phenomenon following recovery from marrow suppression. Spurious thrombocytosis can also occur in severe burns and cryoglobulinaemia because the size of the red cell fragments or cryoglobulin particles is similar to that of platelets. A moderately increased platelet count (e.g. 450–800 × 10 9 /l) often does not indicate a primary haematological disorder. When there is isolated persistent thrombocytosis in a myeloproliferative neoplasm the diagnosis is essential thrombocythaemia (providing that the presence of a BCR – ABL1 fusion gene has been excluded). Thrombotic or haemorrhagic complications can occur with thrombocytosis but often the diagnosis is made incidentally. Individuals with essential thrombocythaemia have been noted to have JAK2 V617F (50%), MPL (10%) or CALR mutations, with the JAK2 mutation being associated with an increased risk of thrombosis.
Reduced numbers of cells
Reductions in more than one cell line
A reduction in cell numbers occurs because of increased destruction, reduced production or increased pooling in the spleen or other organs. Reduced production of cells may be the result of aplastic anaemia, a lack of haematinics such as folate or cobalamin or interference with normal haemopoiesis by infiltration (e.g. leukaemia), infection (e.g. human immunodeficiency virus (HIV) infection, tuberculosis, leishmaniasis) or exposure to toxins (e.g. alcohol) or myelosuppressive drugs (e.g. hydroxycarbamide or methotrexate). Certain myeloid neoplasms, e.g. primary myelofibrosis and myelodysplastic syndromes (MDS), are characterised by cytopenias, which are at least in part the result of ineffective haemopoiesis. Cytopenia is also sometimes a feature of acute myeloid leukaemia (AML), when it is due both to ineffective haemopoiesis and to replacement of normal haemopoietic stem cells by leukaemic cells. A relatively common cause of a global reduction in circulating cells is pooling of the cells in a markedly enlarged spleen (hypersplenism), which may be secondary to conditions such as primary myelofibrosis and portal hypertension. Examination of a bone marrow aspirate and trephine biopsy specimen is often helpful in determining the cause of cytopenias for which no obvious cause is apparent.
Anaemia
The mechanisms which result in anaemia are decreased production, reduced red cell lifespan, blood loss and splenic pooling. Anaemia is broadly divided into three types: microcytic (low mean cell volume (MCV)), macrocytic (high MCV) and normocytic (normal MCV). The choice of investigations is guided by the MCV and red cell morphology in addition to clinical features. Figures 23-1 to 23-3 are flow charts that provide an orderly sequence of investigations for the different types of anaemia on the basis of these indices. Examination of a blood film will usually suggest the quickest route to the diagnosis, though confirmation may require more specific tests. The presence of basophilic stippling with microcytic red cells suggests thalassaemia trait or, much less often, lead poisoning. A dimorphic blood film is typical of congenital sideroblastic anaemia but is more often the result of iron deficiency responding to treatment. Pappenheimer bodies suggest that a microcytic anaemia is the result of sideroblastic erythropoiesis.
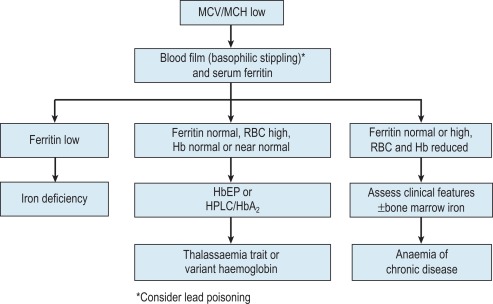
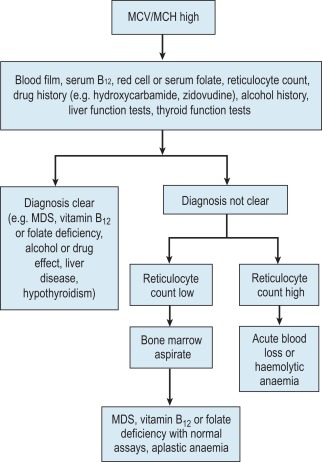
Microcytic anaemia
The most common cause of anaemia worldwide is iron deficiency, which can be suspected from a low MCV ( Fig. 23-1 ) and the presence of hypochromic, microcytic red cells. Laboratory confirmation of iron deficiency can be based on measurement of (1) serum ferritin or (2) serum iron plus either total iron-binding capacity or transferrin or (3) red cell protoporphyrin or (4) staining of bone marrow aspirates for iron (see Chapter 4 ). A diagnosis of iron deficiency must be followed by a search for the cause. This should include specific questions relating to blood loss and dietary insufficiency and may require stool examination for parasites and occult blood, endoscopic examination of the gastrointestinal tract to exclude occult malignancy and tests for coeliac disease. The differential diagnosis of iron deficiency anaemia includes anaemia of chronic disease (also known as anaemia of inflammation). Clinical and laboratory features of inflammation or chronic infection may suggest this diagnosis, which is confirmed by demonstration of normal or high serum ferritin and reduced serum iron, transferrin and iron-binding capacity. Serum soluble transferrin receptors may be helpful in distinguishing between iron deficiency anaemia and anaemia of chronic disease when interpretation of ferritin levels is difficult, though additional research is needed to define the overall diagnostic accuracy of these tests.
The thalassaemias also cause microcytosis, but both α and β thalassaemia trait are usually associated with an increased red blood cell count (RBC) and a normal or near-normal Hb despite a considerable reduction of the MCV and mean cell haemoglobin (MCH). In contrast, in iron deficiency the MCV and MCH do not fall until the Hb is significantly reduced. Further investigations, such as HPLC or haemoglobin electrophoresis supplemented by measurement of haemoglobin A 2 and haemoglobin F usually confirm the diagnosis of β thalassaemia trait. The diagnosis of α thalassaemia trait is more difficult; detection of infrequent haemoglobin H inclusions is usually possible in α 0 thalassaemia trait, but definitive diagnosis requires deoxyribonucleic acid (DNA) analysis (see Chapter 8 ). A diagnosis of α 0 thalassaemia heterozygosity can be clinically important for prediction of haemoglobin Bart’s hydrops fetalis.
Macrocytic anaemia
A high MCV ( Fig. 23-2 ) with oval macrocytes and hypersegmented neutrophils suggests folate or cobalamin deficiency and is an indication for assays of these vitamins (see Chapter 10 ). Plasma methylmalonic acid assays may be a useful second-line test to help clarify uncertainties of underlying biochemical or functional cobalamin deficiencies. Serum folate is the first-line test to assess folate status and has equivalent diagnostic capability to red cell folate. Subsequent investigations could include malabsorption studies, tests for coeliac disease and screening for intrinsic factor antibodies. In patients with these blood film findings and normal folate and cobalamin assays, haematinic deficiency cannot be completely excluded and further investigations are indicated (see Chapter 10 ). As there is no definitive test to define cobalamin deficiency, treatment should be started if there is a strong clinical suspicion of deficiency irrespective of the test results to avoid neurological impairment. In the absence of intrinsic factor antibodies, the diagnosis of pernicious anaemia may be presumptive. Pernicious anaemia is commonly associated with autoimmune thyroid disease and other autoimmune disorders, such as diabetes mellitus.
A high MCV may also be the result of alcohol excess and liver disease or the use of drugs such as hydroxycarbamide. Macrocytosis resulting from chronic haemolysis is associated with increased numbers of immature red cells, which appear slightly larger and bluer than normal red cells (polychromatic macrocytes) on a Romanowsky-stained peripheral blood film. An automated reticulocyte count or supravital staining of blood films (see p. 27) can be used to confirm reticulocytosis. Untreated anaemia associated with polychromasia is likely to indicate blood loss or haemolysis. The combination of red cell fragments, thrombocytopenia and polychromasia indicates a microangiopathic haemolytic anaemia. This is a medical emergency because these may be features of thrombotic thrombocytopenic purpura, which requires immediate treatment, usually by plasma exchange. These features should therefore trigger further tests such as a platelet count, coagulation studies, assessment of renal function, measurement of ADAMTS13 concentration and a search for infection or neoplastic disease.
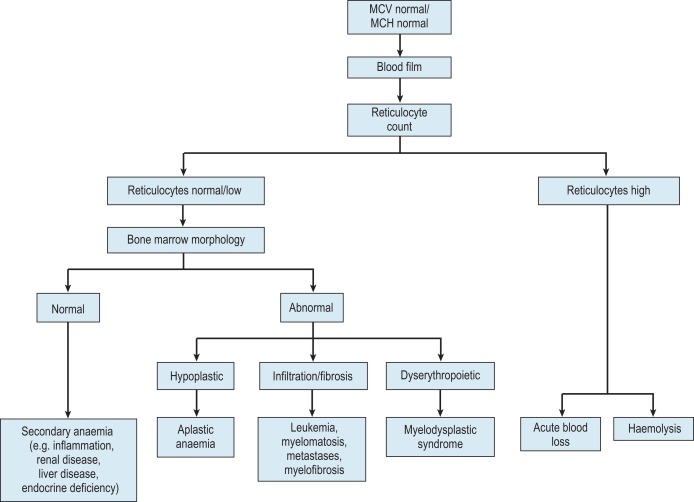
Normocytic anaemia
Normochromic, normocytic anaemia ( Fig. 23-3 ) is frequently the result of an underlying chronic, nonhaematological disease. Investigations should include screening for renal insufficiency, subclinical infections, autoimmune diseases and neoplasia. In the presence of anaemia, a lack of polychromasia, confirmed by reticulocytopenia, points toward a primary failure of erythropoiesis or lack of compensatory increased red cell production in blood loss or haemolysis. Examination of the bone marrow may be helpful in demonstrating haematological causes for a normochromic, normocytic anaemia such as MDS or aplastic anaemia. Staining for iron may also show that there is a block in iron metabolism suggestive of anaemia associated with chronic inflammatory disease.
Leucopenia
Neutropenia
Once physiological variation, ethnicity and familial or cyclic neutropenia have been excluded (see p. 15), the nonhaematological causes of isolated neutropenia to be considered include overwhelming infection, autoimmune disorders such as systemic lupus erythematosus, irradiation, drugs (particularly anticancer agents) and large granular lymphocytic leukaemia. Bone marrow examination may assist in determining whether the problem is the result of peripheral destruction (increased marrow myeloid precursors) or stem cell failure (lack of marrow myeloid precursors). Typical marrow appearances occur in drug-induced neutropenia, in which there is a relative paucity of mature neutrophils and in infant genetic agranulocytosis (Kostmann syndrome) in which there is maturation arrest at the promyelocytic stage.
Reduced numbers of lymphocytes, monocytes, eosinophils and basophils
Lymphocytes, eosinophils and basophils may all be reduced by physical stress such as surgery, trauma and infection. Lymphopenia with neutrophilia is a common combination of haematological abnormalities in severe acute respiratory syndrome and in many other patients with acute illness or trauma. Lymphopenia, especially affecting the CD4 cells, may occur in HIV infection and renal failure. Monocytopenia (monocyte count < 0.2 × 10 9 /1) is typically found in hairy cell leukaemia, which is also associated with pancytopenia, typical bone marrow histology and lymphocytes with a characteristic cytology and immunophenotype.
Thrombocytopenia
Thrombocytopenia is a common isolated finding, and it is important to ensure that the laboratory result reflects a true reduction in platelet count before embarking on further diagnostic tests. Frequent causes of spurious thrombocytopenia include blood clots in the sample, platelet clumping and platelet satellitism. Platelet clumping, which is seen on the blood film, can occur in vitro as the result of a temperature- dependent or anticoagulant-dependent autoantibody or on slides that have been made directly from a finger prick sample. True thrombocytopenia is most frequently the result of autoantibodies (i.e. immune thrombocytopenia), HIV infection, anticancer chemotherapy, other drugs (such as thiazide diuretics), alcohol excess, hypersplenism and MDS. Heparin-induced thrombocytopenia and thrombosis is a particularly important syndrome to recognise (see Chapter 20 ).
The first step in the assessment of patients with thrombocytopenia is the examination of a blood film. The clinical circumstances, together with blood film and bone marrow examination, usually enable the various causes of thrombocytopenia to be differentiated. An association with thrombosis, disturbed renal or hepatic function and haemolytic anaemia should prompt investigations for other diseases, such as thrombotic thrombocytopenic purpura and, in a pregnant woman, the HELLP (haemolysis, elevated liver enzymes, low platelet count) syndrome. The presence of thrombocytopenia with atypical features on the blood film may prompt a bone marrow examination to exclude conditions such as acute leukaemia, especially in children.
Pancytopenia
Pancytopenia means a reduction in the WBC, Hb and platelet count and is most often the result of anticancer chemotherapy, HIV infection, hypersplenism and bone marrow infiltration or failure. Reduction of two cell lineages is referred to as bicytopenia and has similar causes to pancytopenia. Careful examination of a blood film is important if the reason for the cytopenia is not apparent from the clinical history. If this does not reveal the cause, bone marrow aspiration and trephine biopsy may be needed.
Qualitative abnormalities of blood cells
In health, only the most mature forms of cells appear in the peripheral blood. Cells at various stages of immaturity, such as nucleated red blood cells, polychromatic red cells, myelocytes and metamyelocytes, may be released from the bone marrow in conditions where the bone marrow is overactive (e.g. acute haemolytic states or recovery from suppression) or functionally abnormal. Their presence in the peripheral blood indicates that active haemopoiesis is taking place.
Abnormalities of all cell lines
The combination of anisopoikilocytosis, mild macrocytosis, hypogranular neutrophils with abnormal nuclear morphology and platelet anisocytosis, often with quantitative abnormalities, is virtually pathognomonic of MDS. These features are reflected in the bone marrow with disturbance of the normal developmental pathway and sometimes nuclear:cytoplasmic asynchrony. Cytogenetic studies are helpful for confirming the diagnosis, especially when cytological abnormalities are minor, and can also assist in determining the prognosis.
Abnormalities of individual cell lines
Red cells
Congenital abnormalities of the red cell affecting the structure (e.g. spherocytosis, elliptocytosis) and content (e.g. haemoglobinopathies, enzymopathies) often produce typical morphological changes (see Chapter 5 ). The type of changes will guide further investigations such as analysis of structural proteins, haemoglobin electrophoresis or HPLC, or enzyme assays. The type of red cell abnormality may also help to indicate underlying pathology. For example, target cells may prompt investigation of liver function, whereas increased rouleaux formation may indicate the need for investigations for multiple myeloma or inflammatory conditions.
White cells
Congenital abnormalities of neutrophils are unusual, but similar morphological abnormalities (e.g. pseudo-Pelger–Huët cells) may be seen in acquired conditions such as MDS (see Figs. 5-83 and 5-84 ). Reactive changes in lymphocytes, including increased size, irregular shape and basophilic cytoplasm, are typically seen in infectious mononucleosis (see Fig. 5-90 ), which can be diagnosed using an appropriate serological screening test or, if this is negative, by demonstration of immunoglobulin M (IgM) antibodies to the Epstein–Barr virus (EBV). These atypical lymphocytes can sometimes be difficult to differentiate from circulating lymphoma cells. Immunophenotyping studies and determination of lymphocyte clonality, by demonstration of light chain restriction or by gene rearrangement studies, may be needed to reach a firm conclusion.
Platelets
Platelets that function poorly may not necessarily appear morphologically abnormal, although sometimes they are hypogranular or larger than normal. A normal platelet count with an abnormal in vitro platelet function test is characteristic of a disorder of platelet function, but some patients with abnormal platelet function also have thrombocytopenia. Hereditary disorders of platelet function are uncommon and usually present as a bleeding diathesis. When a qualitative disorder of platelets is suspected, platelets should be examined to assess size and to detect the cytological features of platelet alpha-granule deficiency (i.e. grey platelet syndrome). Neutrophils should also be examined for inclusions indicative of MYH9 -related disorders such as the May–Hegglin anomaly. Qualitative disorders of platelets can broadly be divided into two categories: abnormalities of the platelet membrane glycoproteins (e.g. Bernard–Soulier syndrome, Glanzmann thrombasthenia) and abnormalities of platelet secretory function (e.g. storage pool diseases).
Acquired disorders of platelet function are more common than inherited disorders. Haematological conditions associated with platelet dysfunction include myeloproliferative neoplasms, MDS and dysproteinaemias (in plasma cell neoplasms). Many widely prescribed drugs, including aspirin and nonsteroidal anti-inflammatory agents, interfere with platelet function. Systemic conditions, particularly chronic renal failure and cardiopulmonary bypass, are also associated with a bleeding tendency as a result of qualitative platelet defects. Most of these acquired functional defects are not associated with any abnormality in platelet appearance but in MDS and, to a lesser extent, in the myeloproliferative neoplasms, there may be hypogranular and giant platelets.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree