Ancillary Tools in Thyroid Diagnosis
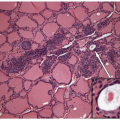
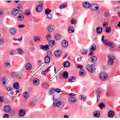
Many thyroid tumors can be readily diagnosed using cytologic and histopathologic criteria, which allow the pathologist to distinguish benign from malignant lesions and guarantee accurate classification of the majority of malignant tumors. However, in several situations, the pathologist is confronted with thyroid lesions in which the distinction between benign and malignant can be quite subtle, or the exact classification of malignancy is not evident on routine examination. The accuracy of diagnosis has clinical consequences and implies different modalities of treatment. On one hand, there is the need to avoid excessive treatment and psychological discomfort to the patient. On the other hand, patients with potentially aggressive disease need to be guaranteed effective management at the initial stages of disease when it is still curable. For this reason, the approach to these challenging situations should include the application of ancillary techniques, such as immunohistochemistry, immunophenotyping, and molecular profiling, that can improve the standard morphologic assessment both in surgical specimens [1] and in cytology samples obtained by fine needle aspiration (FNA) [2].
Genetic studies have identified a process of cumulative molecular events involved in thyroid tumor initiation and progression, resulting in genomic instability and the capacity for independent cellular growth, invasion, and metastasis [3]. It seems unrealistic to expect a single tool, in the form of a magic biomarker, to be able to effectively resolve the diagnostic dilemmas in thyroid pathology. Each marker differentially expressed in tumorous and nontumorous tissues represents a snapshot of the molecular events succeeding in the tissue environment. The amount of information a single marker offers is often insufficient to understand tumor biology or to render accurate diagnosis. The use of combined immunohistochemical markers as a panel seems to be an alternative to aid some of the diagnostic challenges in surgical pathology and cytopathology of thyroid specimens [1, 4, 5], and modern genomic and proteomic technological approaches are being developed to introduce molecular signatures capable of separating benign from malignant thyroid tumors, and in the last group, to distinguish tumors with indolent and aggressive behavior [6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19].
The availability and application of these ancillary tools vary from laboratory to laboratory. In this section, we will review briefly the technologies available. The application of individual tests will be discussed in the sections that follow, where the significance of their results will clarify differential diagnosis.
IMMUNOHISTOCHEMISTRY
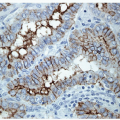
The application of immunohistochemistry is valuable to distinguish lesions of differing histogenesis [1]. This tool can readily determine if a lesion is epithelial or represents a lymphoma or sarcoma. Specific antibodies can identify the profile of a metastatic carcinoma or distinguish a medullary from a follicular carcinoma. There are several antibodies that have been proposed as markers of malignancy, especially for thyroid follicular lesions of an indeterminate nature. These are discussed in detail in the sections that follow.
Immunoperoxidase staining has been optimized for most antibodies for their use on formalin-fixed, paraffin-embedded tissue with or without epitope retrieval techniques. Therefore, when working with FNA samples, any immunoperoxidase staining should be performed on material that has been collected fresh and then undergone formalin fixation with paraffin embedding as a cell block. Occasionally, this may not be possible. For example, an FNA sample may have been deposited into a commercial liquid fixative for liquid-based preparation. In the absence of alternatives, it may be possible to use this material for immunoperoxidase staining. However, if this will be employed, it is important to ensure that validation studies have shown that the results of immunoperoxidase studies performed on material handled in the manner proposed are comparable to standard formalin-fixed tissue.
CELL SURFACE IMMUNOPHENOTYPING AND PLOIDY
The pathologic evaluation of lymphoid populations to establish the diagnosis of thyroid lymphoma [20] is greatly facilitated by the use of cell surface immunophenotyping by either flow cytometry or laser scanning cytometry [21]. With the use of these techniques, surface light chain restriction is demonstrable in many B-cell malignancies, and a number of antigens are detectable that are difficult or impossible to detect in formalin-fixed paraffinembedded tissue. One limitation of cell surface immunophenotyping is the requirement for a fresh cell suspension. In this arena, FNA excels at providing superb cell suspensions and, if the FNA has been performed adequately, yields tremendous cellularity. A cell suspension may also be obtained from a needle core biopsy following maceration and vigorous agitation, but the cellularity typically is far lower than that obtainable from an FNA.
Ploidy and cell cycle determinations are also possible with the use of either flow or laser scanning cytometry. Ploidy analysis was popular during the 1980s when it was thought that this gross method of DNA analysis
would be both diagnostic and predictive [22, 23, 24]. However, it became apparent that benign thyroid lesions could exhibit aneuploidy [23], so the diagnostic value was not proven. Nonetheless, there are data supporting the predictive value of aneuploidy as a marker of more aggressive behavior in documented malignancies of follicular epithelial derivation [25].
would be both diagnostic and predictive [22, 23, 24]. However, it became apparent that benign thyroid lesions could exhibit aneuploidy [23], so the diagnostic value was not proven. Nonetheless, there are data supporting the predictive value of aneuploidy as a marker of more aggressive behavior in documented malignancies of follicular epithelial derivation [25].
MOLECULAR ANALYSIS
The progress in molecular biology of cancer has resulted in highly specific and sensitive tests that can identify cancers, provide more accurate subtyping of malignancies, and determine the appropriate use of targeted therapies. While some of these alterations result in proteomic changes that can be identified by immunohistochemistry, many require identification of changes in RNA, DNA, or microRNAs of tumor cells [12, 14, 17, 18, 26, 27]. DNA is quite stable and often can be extracted from formalin-fixed paraffin-embedded tissue. While RNA can also be obtained from these sources, it is highly dependent on careful handling and the quality is often poor. It is therefore recommended that tissue be prepared specially for the ancillary techniques that require RNA analysis.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree