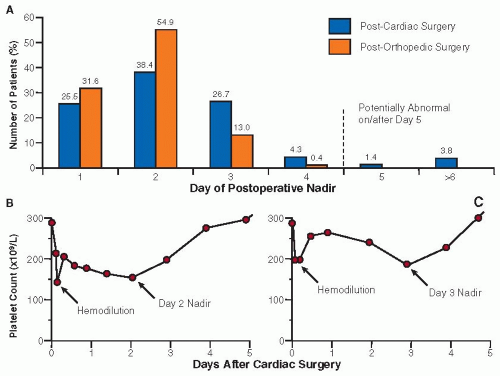
Thrombocytopenia often is caused by decreased platelet production, which can be divided into three categories: acquired, generalized marrow disorders; acquired, isolated megakaryocytic disorders (e.g., alcohol-associated thrombocytopenia); and hereditary thrombocytopenic disorders.
Acquired, Generalized Marrow Disorders
By far the most common cause of decreased platelet production is acquired, generalized bone marrow disease. The pathologic process usually perturbs all cell lines, and pancytopenia typically results. Occasionally, however, isolated thrombocytopenia is the first abnormality detected during the early stages of a panmyelopathy, for example, myelodysplasia (MDS).
Nonneoplastic Hypoproliferative Stem Cell Disorders— Thrombocytopenia accompanies aplastic anemia, chemotherapyinduced pancytopenia, and idiosyncratic drug-induced aplastic anemia. Sometimes, isolated thrombocytopenia is a precursor of aplastic anemia.
Paroxysmal Nocturnal Hemoglobinuria (PNH)—PNH is a stem cell defect resulting in the loss of phosphatidylinositol-anchored proteins, resulting in intravascular hemolysis due to increased susceptibility of red blood cells to complement-mediated lysis; PNH is also associated with thrombocytopenia.
13 The low platelet count is a composite of a dysplastic platelet production defect, enhanced platelet consumption due to the higher susceptibility of PNH platelets to thrombin, and enhanced platelet destruction due to increased generation of lytic C5b9 complexes on platelet membranes.
14 Patients are at increased risk for thrombotic complications, perhaps induced by the production of procoagulant microparticles from affected blood cells. Diagnosis is made by showing by flow cytometry a population of blood cells with decreased/absent phosphatidylinositol-anchored proteins. A treatment option in severe PNH is the monoclonal antibody, eculizumab.
15,16
Myelodysplasia—Isolated, refractory thrombocytopenia is the initial presentation of MDS in 1% to 3% of patients and can be confused with immune thrombocytopenic purpura (ITP).
17 Bleeding can result from thrombocytopenia or defective platelet function or from both. Findings that aid in differentiating MDS from ITP include red cell macrocytosis, a left-shifted white cell series, leukocyte morphologic abnormalities (e.g., hypogranular neutrophils and Pelger-Huët anomaly), and hypogranular, giant platelets. Marrow examination reveals dysmegakaryocytopoiesis in half of cases. Typically, the megakaryocytes are abnormally
small, but large mononuclear and abnormal segmented megakaryocytes are found in some patients.
18 Megakaryocyte numbers are usually normal, but increased megakaryocytes (resembling ITP) or amegakaryocytic thrombocytopenia
19 have also been observed.
Neoplastic Stem Cell Disorders—Thrombocytopenia is common with primary neoplastic disorders of the bone marrow. Usually the presence of pancytopenia and circulating malignant cells points to the diagnosis; bone marrow aspirate and biopsy are confirmatory. These disorders include acute myeloid and acute lymphoid leukemia, as well as many chronic lymphoproliferative disorders, such as chronic lymphoid leukemia, hairy cell leukemia, non-Hodgkin’s lymphoma, adult T-cell leukemia and lymphoma, multiple myeloma, and Waldenström’s macroglobulinemia.
Infiltrative Bone Marrow Disorders—Thrombocytopenia can be secondary to infiltration of the bone marrow by nonhematologic metastatic malignancies, infectious organisms, storagecell disorders, myelofibrosis, and osteopetrosis. When bone marrow replacement occurs from malignancies (e.g., breast cell carcinoma, prostate carcinoma), peripheral blood features include normoblasts (nucleated red cells), teardrop red cells, and leukopenia usually with leukocyte left shift. This constellation of findings is called a “myelophthisic” blood picture.
20
Megaloblastic Anemia—Megaloblastic anemia, caused by vitamin B
12 or folic acid deficiency, is often associated with mild thrombocytopenia. Vitamin deficiency-induced defective DNA synthesis results in megakaryocytes of low ploidy and small platelets.
21 Thus, the combination of large, heterogeneous red cells (oval macrocytes) together with small platelets (low mean platelet volume) is characteristic of megaloblastic anemia.
Isolated Thrombocytopenia Due to Drugs
Alcohol-associated thrombocytopenia is associated with heavy and prolonged alcohol consumption and can cause an acute, self-limited thrombocytopenia.
22 The platelet count increases 2 to 3 days after cessation of alcohol consumption, with “rebound” thrombocytosis. Bone marrow aspirates early in the course of the thrombocytopenia reveal reduced megakaryocyte numbers and sometimes vacuolation of normoblasts and promyelocytes.
Interferon used for the treatment of hepatitis C causes a 10% to 50% drop in the platelet count in patients on combination therapy and 4% to 6% of patients require dose modification secondary to thrombocytopenia.
23 Interferon is thought to alter thrombopoietin production, thereby reducing platelet production. In most cases, thrombocytopenia is clinically insignificant, but reduction of the interferon dose is necessary when platelet counts decrease below 50 × 10
9/L and cessation of treatment is required if platelet counts decrease below 30 × 10
9/L.
Drugs targeting RUNX1, a transcription factor important in leukemia, have the potential to cause decreased platelet production,
24 as RUNX1 plays an important role in thrombopoiesis.
25
Iron repletion: Mild-to-moderate thrombocytopenia can be observed also approximately 1 week after beginning iron replacement therapy
26; this transient platelet count decline is most likely the result of a shift of stem cells to erythropoiesis.
Fatty acid-induced thrombocytopenia is most prominent in patients with adrenoleukodystrophy, who are treated with Lorenzo’s oil (a mixture of glycerol trioleate and trierucate).
27 These oils change the composition of the platelet membrane, causing membrane anisotropy the production of enlarged and giant platelets, as well as platelet function defects.
28
Miscellaneous Acquired Thrombocytopenia
Iron depletion—Mild-to-moderate thrombocytopenia can be observed at presentation of severe iron deficiency anemia.
32
Acquired amegakaryocytic thrombocytopenia—Acquired amegakaryocytic thrombocytopenia is a rare disorder characterized by isolated thrombocytopenia associated with a total or marked reduction in megakaryocytes.
33,34 Sometimes, progression to aplastic anemia or MDS occurs. In other patients, there are autoimmune features (e.g., association with systemic lupus erythematosus (SLE) or a positive direct Coombs’ test). Cyclic amegakaryocytic thrombocytopenia has also been observed.
35
The pathogenesis of acquired amegakaryocytic thrombocytopenia is heterogeneous and at least three different mechanisms have been implicated: an intrinsic stem cell defect (defective growth of megakaryocyte precursors), autoantibodies directed against megakaryocyte precursors, and T-cell-mediated suppression of megakaryocyte development. Thrombopoietin levels are usually much greater than seen in patients with ITP.
36Treatment of these disorders have included corticosteroids, antithymocyte globulin, cyclosporine, cyclophosphamide, immunoglobulins, splenectomy, or allogenic bone marrow transplantation (BMT).
33,34