The incidence of cervical cancer in 2010 in the United States is estimated to be 12,200.
Estimated deaths in the United States due to cervical cancer is 4,210 (1).
The 2007 global statistics from the American Cancer Society revealed that cervical cancer is the second most common malignancy in the world and the third leading cause of cancer death in women.
The uterus is located in the midplane of the true pelvis, in an anteverted position, behind the bladder and in front of the rectum. It is partially covered by peritoneum in its fundal portion. Posteriorly, its lateral surfaces are related to the parametria and the broad ligaments.
The two main regions are the corpus and cervix. The cervix is separated from the corpus by the isthmus and is divided into the supravaginal portion, which is above the ring containing the endocervical canal, and the vaginal portion, which projects into the vault.
The uterus is attached to the surrounding structures in the pelvis by the broad and the round ligaments. The broad ligament is a double layer of peritoneum extending from the lateral margin of the uterus to the lateral wall of the pelvis and the pelvic floor. It contains the fallopian tubes. The round ligament extends from its attachment in the anterolateral portion of the uterus to the lateral pelvic wall; it crosses the pelvic brim and reaches the abdominoinguinal ring, through which it traverses the inguinal canal and terminates in the superficial fascia.
The uterosacral ligaments extend from the uterus to the sacrum and run along the recto-uterine-peritoneal fields.
The cardinal ligaments, also called the transverse cervical ligaments (Mackenrodt’s), arise at the upper lateral margins of the cervix and insert into the pelvic diaphragm.
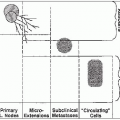
The uterus has a rich lymphatic network that drains principally into the paracervical lymph nodes; from there it goes to the external iliac (of which the obturator nodes are the innermost component) and the hypogastric lymph nodes (Fig. 35-1). The pelvic lymphatics drain into the common iliac and the periaortic lymph nodes.
Lymphatics from the fundus pass laterally across the broad ligament continuous with those of the ovary, ascending along the ovarian vessels into the periaortic lymph nodes. Some of the fundal lymphatics also drain into the external and internal common iliac lymph nodes.
Squamous cell carcinoma of the uterine cervix usually originates at the squamous columnar junction (transformation zone) of the endocervical canal and the portion of the cervix.
The malignant process breaks through the basement membrane of the epithelium and invades the cervical stroma. If the invasion is less than 3 mm (less than 7 mm on the surface), the lesion is classified as stage IA1; the probability of lymph node metastasis is 1% or less (8).
Invasion may progress: In a modification of the International Federation of Gynecology and Obstetrics (FIGO) staging schema (32), a tumor is classified as stage IA2 invasive carcinoma if it is not grossly visible and has a depth of penetration of less than 5 mm and breadth of 7 mm or less. The incidence of metastatic pelvic lymph nodes is related to the depth of invasion, with an overall incidence of 3% to 8%.
Growth of the lesion eventually may be manifested by superficial ulceration, exophytic tumor in the exocervix, or extensive infiltration of the endocervix.
The tumor may spread to the adjacent vaginal fornices or to the paracervical and parametrial tissues, with eventual direct invasion of the bladder, rectum, or both (if untreated).
Regional lymphatic or hematogenous spread frequently occurs, depending on the stage of the tumor. However, dissemination does not always follow an orderly sequence; occasionally, a small carcinoma may produce distant metastasis or infiltrate the pelvic lymph nodes, bladder, or rectum.
The incidence of pelvic node metastasis is approximately 15% in stage IB, 25% to 30% in stage IIB, and 50% in stage IIIB disease. The incidence of para-aortic node metastasis is approximately 5% in stage IB, 19% in stage IIB, and 30% in stage IIIB disease (48).
The most common metastatic sites are the lungs (21%), para-aortic nodes (11%), mediastinal and supraclavicular lymph nodes (7%), bones, and liver (18).
Intraepithelial or early invasive carcinoma of the cervix can be detected by cytologic Papanicolaou (Pap) smears, colposcopy and biopsies, and human papillomavirus (HPV) testing before it becomes symptomatic.
One common, early manifestation is postcoital spotting, which may increase to limited metrorrhagia (intermenstrual bleeding) or more prominent menstrual bleeding (menorrhagia). If chronic bleeding occurs, the patient may complain of fatigue or other symptoms of anemia.
Vaginal discharge is also common.
Pain, usually in the pelvis or hypogastrium, may be caused by tumor necrosis or associated pelvic inflammatory disease.
Pain in the lumbosacral area suggests periaortic lymph node involvement with extension into the lumbosacral roots or hydronephrosis.
Urinary and rectal symptoms (hematuria, rectal bleeding) may occur in advanced stages.
Initiating screening with a Pap smear should begin at age 21 or 3 years after the onset of sexual activity, whichever comes first (62). The American Cancer Society advocates annual screening for women under age 30 (biennial if using liquid-based testing), and reducing the frequency to every two to three years for women aged 30 and older who have had three consecutive normal Pap tests, or every three years if they also are tested for HPV DNA.
Average risk women, age 30 years and older, may be screened with a combination of Pap smear and HPV testing; combination testing should be performed no more frequently than every three years if both tests are negative.
Women with a negative cytology and a positive HPV test should have both tests repeated in 6 to 12 months. A positive result in either test warrants a colposcopy.
HPV 16 and 18, which are sexually transmitted diseases, have been well established as the etiologic agent in approximately two thirds of cervical cancer cases.
E6- and E7-transforming oncoproteins of high-risk HPV genotypes play a crucial role in both the transformation and the maintenance of the malignant phenotype. The interaction of E6 and E7 proteins with cell cycle-regulating proteins p53 and pRB leads to the disruption of normal growth regulatory mechanisms.
An HPV vaccine was approved by the FDA in 2006 for HPV types 6, 11, 16, and 18. The HPV vaccine has been found to be effective in clinical trials in significantly lowering the occurrence of high-grade cervical intraepithelial neoplasia related to HPV-16 or HPV-18 than did those in a placebo group (56).
The technique for obtaining the Pap smear is described in standard textbooks (66).
Every patient with invasive carcinoma of the cervix should be jointly evaluated by radiation and gynecologic oncologists.
After a general physical examination, with special attention to the supraclavicular (nodal) areas, abdomen, and liver, a careful pelvic examination should be carried out, including bimanual palpation of the pelvis.
Colposcopy may adequately evaluate the exocervix and a portion of the endocervix adjacent to the transition of the squamous and columnar epithelium (T zone).
Conization must be performed when no gross lesion of the cervix is noted, when an endocervi-cal tumor is suspected, when diagnosis of microinvasive carcinoma is made on biopsy, or if the patient is not reliable for continuous follow-up.
When a gross lesion of the cervix is present, multiple punch biopsies should be obtained from the margin of any suspicious area as well as in all four quadrants of the cervix and from any suspicious areas in the vagina.
Fractional curettage of the endocervical canal and the endometrium is recommended at the time of initial evaluation or, if the patient is treated with irradiation, during the first intracavitary brachytherapy insertion.
For invasive carcinoma, patients should have complete peripheral blood evaluation, including hemogram, white blood cell count, differential, and platelet count; Sequential Multiple Analysis-12-channel biochemical profile, with particular attention to blood urea nitrogen, creatinine, and uric acid; liver function values; and urinalysis.
Cystoscopy or rectosigmoidoscopy should be performed in all patients with stage IIB, III, and IVA disease, and possibly in those with earlier stages who have a history of urinary or lower gastrointestinal tract disturbances.
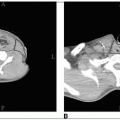
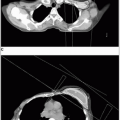
Chest radiograph and intravenous pyelogram (IVP) should be obtained in all patients for staging. Computed tomography (CT) scan with contrast frequently replaces IVP.
A barium enema study should be performed in patients with stage IIB, III, and IVA disease, as well as in those with earlier stages who have symptoms referable to the colon and rectum.
Magnetic resonance imaging (MRI) is being used more frequently for assessment of parametrial invasion (30).
Positron emission tomography scanning with fluorodeoxyglucose (FDG-PET) is increasingly being incorporated in initial staging, management, monitoring response to therapy, and follow up of cervical cancer (26, 29).
In general, FDG-PET is reported to be highly sensitive and specific for the detection of pelvic and para-aortic lymph nodes in patients with locally advanced disease (39).
The diagnostic procedures for carcinoma of the cervix are presented in Table 35-1.
It is imperative that the gynecologic and radiation oncologist jointly stage the tumor in every patient, with bimanual pelvic and rectal examination under general anesthesia.
Ideally, staging should be done before institution of therapy; however, final staging occasionally is postponed after initial evaluation (because of logistic and economic reasons) until the time of
a radical hysterectomy or the first intracavitary brachytherapy insertion, which should be done within 2 weeks from initiation of the external irradiation if the patient is treated with this modality.
The FIGO staging recommendations were revised in 2009. Deletion of stage 0: FIGO has decided to delete stage 0 from the staging of all tumors, since it is a preinvasive lesion.
Stage IIA: Several reports in the literature and data analyses from the FIGO Annual Report database consistently demonstrate that, in stage IIA patients, size, defined as the maximum tumor diameter, has an effect on prognosis similar to that observed in stage IB. Therefore, definitions of stage IIA substages have been included:
Stage IIA1: tumor size of <4 cm with involvement of less than the upper two thirds of the vagina.
Stage IIA2: tumor size of more than 4 cm with involvement of less than the upper two thirds of the vagina.
A parallel tumor-node-metastasis staging system has been proposed by the American Joint Committee on Cancer (3). The current criteria for the various stages are defined in Table 35-2.
All histologic types should be included.
When there is a disagreement regarding the staging, the earlier stage should be selected for statistical purposes.
The FIGO staging system is based on clinical evaluation (inspection, palpation, colposcopy), roent-genographic examination of the chest, kidneys, and skeleton, and endocervical curettage and biopsy.
Lymphangiogram, arteriogram, CT, PET scan, laparoscopy, or laparotomy findings should not be used for clinical staging.
Suspected invasion of the bladder or rectum should be confirmed by biopsy for staging purposes.
TABLE 35-1 Diagnostic Workup for Carcinoma of the Uterine Cervix | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
TABLE 35-2 TNM and FIGO Staging Categories for Cancer of the Cervix | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Some gynecologists have advocated the use of pretherapy laparotomy, particularly to evaluate the presence of para-aortic lymph nodes. Recently, sentinel node biopsy in cervical cancer patients has been used with encouraging results (15).
No significant impact of surgical staging on overall survival (OS) has been reported (44).
Over 90% of tumors of the cervix are squamous cell carcinomas. Approximately 7% to 10% are classified as adenocarcinoma, and 1% to 2% is clear cell, mesonephric type.
Verrucous carcinoma is a variant of a very well-differentiated squamous cell carcinoma that has a tendency to recur locally but not to metastasize.
Adenosquamous carcinoma is relatively rare (2% to 5% of all cervical carcinomas) and consists of intermingled epithelial cell cores and glandular structures.
Adenoid cystic carcinoma is a rare neoplasm of the cervix (less than 1% incidence) with an appearance similar to its counterparts in the salivary gland or bronchial tree.
Glassy cell carcinoma is considered a poorly differentiated adenosquamous tumor with a distinctive histologic appearance. Survival is poor after surgery or irradiation.
Small-cell carcinoma of the cervix, according to some authors, arises from endocervical argyrophil cells or their precursors, multipotential neuroendocrine cells; one third to one half stain positively for neuroendocrine markers such as chromogranin, serotonin, or somatostatin. Lymphatic and vascular invasion are substantially more common in small-cell carcinoma.
Basaloid carcinoma (or adenoid-basal carcinoma), an extremely uncommon tumor, is characterized by nests or cords of small basaloid cells.
Primary sarcomas of the cervix (leiomyosarcoma, rhabdomyosarcoma, stromal sarcoma, carcinosarcoma) have been described occasionally.
Malignant lymphomas, primary or secondary in the cervix, have been reported sporadically. They behave (and should be treated) as other lymphomas.
According to some reports, prognosis is the same in younger and older patients (6); others have noted decreased survival in women under 35 (55) or 40 years (13), who have a higher frequency of poorly differentiated tumors.
Several authors have observed a correlation between race or socioeconomic characteristics and outcome of therapy (12).
In addition to stage and tumor volume, histologic type of the lesion and vascular or lymphatic invasion are important prognostic factors.
Most reports have shown no significant correlation between survival or tumor behavior and the degree of differentiation of squamous cell carcinoma or adenocarcinoma of the cervix (24).
In a study of women treated with radical hysterectomy, the 5-year disease-free survival rate was 90% in 181 patients with stage IB1 (less than 4 cm) and 72.8% in 48 patients with stage IB2 tumors (p = 0.02) (19).
Delgado et al. (14) described 3-year disease-free survival rates of 94.8%, 88.1%, and 67.6%, respectively, for occult, less than 3 cm, and greater than 3 cm stage I invasive squamous cell carcinoma of the cervix treated with radical operation.
Eifel et al. (16) and Perez et al. (49) reported a close correlation between size of primary tumor, incidence of pelvic recurrences, and survival after irradiation.
Several retrospective studies have demonstrated decreased survival and a greater incidence of distant metastases in patients with endometrial extension of primary cervical carcinoma (endometrial stroma invasion or replacement of endometrium by tumor only) (51).
Tumors involving the uterosacral space or causing hydronephrosis above the level of the pelvic brim are associated with a higher incidence of local recurrence and distant metastasis (10 and 11).
Some authors have noted no significant difference in recurrence rates between patients with diploid or aneuploid tumors (67), whereas others have observed a less favorable prognosis in tumors with a diploid or a tetraploid DNA (60).
Alterations in either the expression or function of cellular genes that control cell growth and differentiation in cervical cancer show no clear-cut use as prognostic markers (66).
The c-myc oncogene is amplified 3 to 30 times in approximately 20% of squamous cell carcinomas. Overexpression of c-myc is associated with a worse clinical outcome (57).
Host factors (e.g., anemia) affect the prognosis of patients with cervical carcinoma.
A higher incidence of pelvic recurrences and complications in patients with arterial hypertension (diastolic pressure greater than 110 mm Hg) has been described.
Decreased survival has been reported in patients with oral temperature higher than 100°F (33).
Controversy continues between those who advocate radical surgery and those who advocate radiation therapy for the treatment of early carcinoma of the uterine cervix; results are equivalent (37).
The use of irradiation as sole modality has declined; this may be related to earlier tumor detection because of greater awareness in physicians and patients, the widespread use of Pap smear screening, and greater use of surgery and chemoradiation in the treatment of these patients.
Treatment should involve close collaboration between the gynecologic oncologist and the radiation oncologist, with an integrated team approach vigorously pursued.
Patients with carcinoma in situ, which may include those with severe dysplasia, are usually treated with a total abdominal hysterectomy, with or without a small vaginal cuff. The decision to remove the ovaries depends on the patient age and the status of the ovaries.
When the patient wishes to have more children, carcinoma in situ may be treated conservatively in selected patients, with therapeutic conization, laser, or cryotherapy.
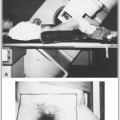
Intracavitary irradiation (tandem and ovoids) may be useful for the treatment of in situ carcinoma (45 to 50 Gy to point A), particularly in patients with strong medical contraindications for surgery or when there is multifocal carcinoma in situ in both the cervix and vagina (23).
Early invasive carcinoma of the cervix (stage IA2) is usually treated with a total abdominal or modified radical hysterectomy, but it can be treated with intracavitary brachytherapy alone (6,500 to 8,000 mgh [56 to 70 Gy to point A, respectively] in one or two insertions). High dose rate (HDR) brachytherapy can be prescribed to 36 to 45 Gy in 6 to 8 fractions depending on tumor size and stromal invasion.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree