, S. J. Valle1, W. Liauw1, 3 and D. L. Morris1
(1)
Department of Surgery, St George Hospital, Kogarah, NSW, 2217, Australia
(2)
College of Medicine, Al-Imam Muhammad ibn Saud Islamic University, Riyadh, Saudi Arabia
(3)
Cancer Care Centre, St George Hospital, Kogarah, NSW, 2217, Australia
4.1 Introduction
The occurrence of peritoneal carcinomatosis (PC) is serious in all malignancies and usually results in a poor prognosis. Cytoreductive surgery (CRS) and hyperthermic intraperitoneal chemotherapy (HIPEC) is an aggressive surgical approach combining visceral resections and peritonectomy procedures to remove macroscopic disease followed by HIPEC, targeting residual microscopic disease. CRS/HIPEC is the standard and potentially curative treatment for select patients with PC. This combined approach, however, has consistently demonstrated improved survival outcomes across a variety of disease types including appendiceal cancer, colorectal cancer, low-grade appendix mucinous neoplasm, peritoneal mesothelioma and ovarian cancer with peritoneal metastases [1–6], with randomised evidence demonstrating the superiority of CRS/HIPEC for colorectal cancer PC [2]. Although peritonectomy procedures are less likely to achieve disease control in selected patients with significant metastatic disease [7], CRS/HIPEC has been applied and reported in small series of patients with PC of unusual origin including tumours arising from sarcoma, desmoplastic small round cell tumour (DSRCT) and neuroendocrine tumours [8–10]. The role of CRS/HIPEC in these patients remains uncertain with limited data on the clinical efficacy of this treatment, as many reports outline specific outcomes in large heterogeneous groups. This goal of this manuscript was to review our single institution experience with CRS/HIPEC for patients with PC of unusual primaries followed by a literature review of studies that reported on greater than five patients of each unusual primary.
4.2 Methods
4.2.1 Patients
From 1,025 patients that underwent CRS/HIPEC at St George Hospital, Australia, between 1996 and 2016, 31 patients had an unusual primary tumour. The tumour included sarcoma (n = 10), desmoplastic small round cell tumour (n = 3), biliary (n = 3), pancreatic (n = 3), neuroendocrine (n = 3), hepatocellular carcinoma (n = 3), primary peritoneal carcinomatosis (n = 2), urachal adenocarcinoma (n = 2), cystadenoma of the liver (n = 2), gastrointestinal stromal tumour (n = 2) and breast cancer (n = 1). Outcomes were retrospectively reviewed from a prospectively entered database.
4.2.2 Preoperative Management
All patients underwent standard preoperative investigations, which included physical examination; double contrast-enhanced computed tomography (CT) scans of the chest, abdomen and pelvis; and MR study of the liver with Primovist, positron emission tomography and blood tumour markers. All patients were discussed at a multidisciplinary team meeting prior to surgery.
4.3 CRS/HIPEC
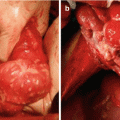
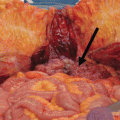
An initial assessment of the volume and extent of disease was recorded using the peritoneal carcinomatosis index (PCI), as described by Jacquet and Sugarbaker [11]. CRS was performed using Sugarbaker’s technique [11]. All sites and volumes of residual disease following CRS were recorded prospectively using the completeness of cytoreductive (CC) score as previously described [12]. After CRS, HIPEC was performed by installation of a heated chemoperfusate into the abdomen using the coliseum technique at approximately 42 °C, depending on tumour types. However, there is no fixed protocol for PC from unusual primary and was at the discretion of the medical oncologist. Drug choices included cisplatin, mitomycin C and oxaliplatin.
4.3.1 Postoperative Management
Perioperative complications in all patients were graded as previously described [13]. Major morbidity is defined as CDC grade III or IV. All of patients with the aggressive tumour were then followed up at 3-month intervals for the first 12 months and 6-month intervals thereafter until the last time of contact or death. The follow-up review included clinical examination, measurement of relevant tumour markers and assessment of abdominopelvic CT scans.
4.4 Results
The pathology and perioperative outcomes of patients are outlined in Table 4.1. Overall hospital mortality was 5.8% (n = 2). Major morbidity rate was 38% (n = 13). Mean ICU, HDU and total hospital stay was 4.5 days (standard deviation (SD) = 8.1), 3.0 days (SD = 2.7) and 26.3 days (SD = 21.6), respectively.
Table 4.1
Baseline characteristics of the study cohort
Total n = 34 | |
Gender n (%) | |
Male | 18 (52.9) |
Female | 16 (46.1) |
Age mean (SD) | 48.4 (14.1) |
Primary site | |
Cystic adenoma | 2 (5.8) |
Pancreatic cancer | 3 (8.8) |
Breast cancer | 1 (2.9) |
GIST | 2 (5.8) |
Urachal carcinoma | 2 (5.8) |
Primary peritoneal cancer | 2 (5.8) |
Neuroendocrine | 3 (8.8) |
Biliary adenocarcinoma | 3 (5.6) |
DSCRT | 3 (8.8) |
HCC | 3 (8.8) |
Sarcoma | 10 (31.2) |
PCI mean (SD) | 12.9 (8.3) |
CC score | |
0 | 32 (100) |
Transfusion mean (SD) | 4.4(4.6) |
Operation hours mean (SD) | 7.4 (2.7) |
HIPEC n (%) | |
Yes | 32 (94.1) |
No | 2 (5.9) |
EPIC | |
Yes | 21 (61.8) |
No | 13 (38.2) |
Mortality rate n (%) | 2 (5.8) |
Major morbidity rate n (%) | 13 (38.2) |
ICU stay mean (SD) | 4.5 (8.1) |
HDU stay mean (SD) | 3.0 (2.7) |
Total hospital stay mean (SD) | 26.3 (21.6) |
Median overall survival (OS) was 29.6 months (95%CI = 13.8–35.5) with a 1-year, 3-year and 5-year survival of 69.6%, 42.7% and 26.7%, respectively. Median follow-up time was 14.4 months (range = 0.2–86.7). Median disease-free survival (DFS) was 6.3 months (95%CI = 2.4–8.1). Survival outcomes by diagnoses and diagnoses with at least three patients were summarised in Tables 4.2 and 4.3, respectively.
Table 4.2
Summary of survival (months) for primaries with two or less patients
Cystic adenoma (n = 2) | Breast cancer (n = 1) | GIST (n = 2) | Urachal carcinoma (n = 2) | Primary peritoneal cancer (n = 2) | |
|---|---|---|---|---|---|
Patient 1 | 0.6 (alive) | 0.6 (died) | 51.1 (alive) | 21.3 (died) | 41.4 (died) |
Patient 2 | 12.0 (alive) | 4.9 (died) | 86.7 (alive) | 27.4 (alive) |
Table 4.3
Summary of median and overall survival for primaries with at least three patients
Total number (n) | 1-year OS (%) | 3-year OS (%) | 5-year OS (%) | Median OS (months) (95%CI) | Median DFS (months) (95%CI) | |
|---|---|---|---|---|---|---|
Pancreatic cancer | 3 | 66.7 | NR | NR | 9.6 (−) | 5.2 (−) |
Neuroendocrine | 3 | 100.0 | 33.3 | NR | 18.9 (0–32.4) | 14.6 (−) |
Biliary carcinoma | 3 | 33.3 | NR | NR | 12.0 (−) | 3.9 (−) |
HCC | 3 | 66.7 | 33.3 | 0 | 36.4 (−) | 5.1 (−) |
DSRCT | 3 | 100.0 | 100.0 | 33.3 | 64.9 (−) | 10.3 (−) |
Sarcoma | 10 | 60.0 | 60.0 | 0 | 50.5 (−) | 6.6 (1.3–11.9) |
4.5 Discussion
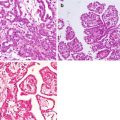
4.5.1 Desmoplastic Small Round Cell Tumour
Desmoplastic small round cell tumours (DSRCT) are a rare sarcoma with fewer than 500 cases reported between 1989 and 2015 and is a highly aggressive sarcoma predominantly occurring in males aged between 5 and 35 years [14]. DSRCTs are located almost exclusively in intra-abdominal locations and classically involve a large intra-abdominal mass in the retroperitoneum, pelvis, omentum or mesentery, with diffuse peritoneal deposits that spread along peritoneal and mesothelial surfaces [15, 16]. We previously reported on our patients (n = 3) with this tumour and showed a similar outcome [17]. Treatment of isolated peritoneal disease relies on perioperative chemotherapy, most commonly with cisplatin, combined with complete CRS and postoperative radiotherapy [18]. Hayes-Jordan et al. showed that HIPEC has improved survival outcomes with patients receiving neoadjuvant chemotherapy followed by CRS/HIPEC having a 3-year survival of 71% compared to 26% (p = 0.021) in patients who did not receive surgery or HIPEC [14]. In a recent study by the same group with the largest experience internationally, the median survival of patients with DSRCT that underwent CRS/HIPEC and complete cytoreduction with no extraabdominal disease was 63.1 months with a 100% 4-year survival [19] (Table 4.4). Our experience of these three patients showed that in concordance with the literature, disease-free survival was 10.3 months although the 3-year survival was 100%. The role of HIPEC for this indication remains controversial; however, complete CRS/HIPEC seems to be an effective therapy for patients with DSCRT.
Table 4.4
Literature review of patients with unusual primary that underwent CRS/HIPEC
Author | Year | Primary | Number (n) of patients | Median survival (months) | Disease-free survival (months) | 5-year survival |
|---|---|---|---|---|---|---|
Rossi [22] | 2004 | Peritoneal sarcomatosis | 60 | 34 | 22 | – |
Bonvalot [26] | 2005 | Peritoneal sarcomatosis | 38 | 29 | 12.5 | 45% (1-year survival) |
Lim [25] | 2007 | Peritoneal sarcomatosis | 28 | 5.5–16.9 | 2.3–4.4 | 11% |
Baratti [9] | 2010 | Peritoneal sarcomatosis | 37 | 26.2 | 12.1 | 24% |
Salti [32] | 2012 | Peritoneal sarcomatosis | 13 | 12 | 11 | – |
Randle [33] | 2013 | Peritoneal sarcomatosis | 10 | 21.6 | 15.3 | 43% |
Bryan [35] | 2015 | GIST | 18 | 40 | – | 56% (3-year survival) |
Hayes-Jordan [14] | 2010 | DSRCT | 24 | 12.4 | 12 | 71% |
Hayes-Jordan [19] | 2014 | DSRCT | 26 | 31.1 | – | – |
Honore [18] | 2015 | DSRCT | 23 | 25.7 | 15.5 | – |
Elias [8] | 2014 | Neuroendocrine tumour | 41 | NR | – | 69% |
Tentes [61] | 2012 | Pancreatic adenocarcinoma | 21 | 11 | 5 | 23% |
Bakrin [53] | 2013 | Primary peritoneal carcinomatosis | 36 | NR | 16.7 | 57.4% |
Tabrizian [58] | 2014 | Hepatocellular carcinoma | 14 | 35.6 (CC0/1) | 23 | – |
Krane [51] | 2012 | Urachal carcinoma | 5 | 27 | 13 | – |
Cardi [37] | 2013 | Breast carcinoma | 5 | 63.2 | NR | – |
4.5.2 Peritoneal Sarcomatosis
Approximately 36% of sarcomas originate in the abdominal viscera or retroperitoneum, and metastatic disease is most common to the lungs, liver or directly to peritoneal surfaces and adjacent organs [20]. For patients amenable to resection, local recurrence for abdominal sarcoma ranges from 35% to 82% [21, 22]. Sarcomatosis is defined as the intra-abdominal dissemination of sarcoma and may be present at initial diagnosis but is more frequently observed at recurrence presumably as a result of tumour spillage during the initial resection [23, 24]. When CRS/HIPEC is applied to sarcomatosis, outcomes have been unclear [10, 22, 25–31]. The median overall survival on literature review of peritoneal sarcomatosis post-CRS/HIPEC ranged from 12 to 39.6 months [22, 23, 25–27, 32] (Table 4.4). We report that in our patients, the median overall survival was 50.5 months. Randle et al. [33] reported that although complete cytoreduction is related to improved survival, adding HIPEC in sarcomatosis patients is controversial given the potential toxicity, significant recovery time and lack of a documented benefit. Highly selected sarcomatosis patients are still treated with CRS; however, they are no longer offered HIPEC [33]. A multi-institutional review may be helpful in further defining the role of CRS/HIPEC.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree