Summary of Key Points
- •
Performance status is universally recognized as an independent prognostic factor and typically correlates with the extent of tumor burden.
- •
As first-line therapy, platinum agent plus etoposide or irinotecan remains the standard of care for the treatment of small cell lung cancer (SCLC).
- •
The ideal number of chemotherapy cycles for SCLC has not been defined; however, four to six cycles are considered the standard based on results from randomized trials.
- •
Despite an initially high response rate to frontline platinum-based chemotherapy, extensive-stage SCLC will universally relapse, often within 3 to 6 months.
- •
Alternative chemotherapy strategies have focused on modifying the dosage and schedules of established regimens.
- •
Dose-dense regimens have shown mixed results.
- •
Most trials employing a dose-intensification strategy did not show a survival advantage over standard therapy for patients with extensive-stage SCLC, and higher doses were typically associated with greater toxicity.
- •
Patients who receive no further therapy have a median survival of less than 3 months.
- •
Patients who have previously received platinum-based therapy are grouped into two general categories reflecting the platinum-sensitivity status of their disease: platinum sensitive and platinum refractory.
- •
Topotecan is approved as second-line treatment for patients with platinum-sensitive, relapsed disease based on symptom control. An oral formulation of topotecan was also developed for patients’ convenience.
- •
Despite progress in the understanding of genomic alterations and signaling pathways in SCLC, clinical experiments with tyrosine kinase inhibitors, other small-molecule inhibitors, and antiangiogenic agents have been disappointing Other therapeutic areas of interest more recently evaluated include epigenetic modifiers, inhibitors of DNA repair and the cell cycle, immunocheckpoint inhibitors and inhibitors of the Notch pathway.
The chemosensitivity of small cell lung cancer (SCLC) was first identified 50 years ago with the recognition that methyl-bis-β-chloroethyl amine hydrochloride could cause tumor regression in more than 50% of patients. Since then, numerous antineoplastic agents have been shown to produce objective response rates of at least 20% in previously untreated patients. Older active agents included nitrogen mustard, doxorubicin, methotrexate, ifosfamide, etoposide, teniposide, vincristine, vindesine, nitrosureas, and cisplatin and its analog carboplatin. In the 1990s, six new agents were discovered to have activity against SCLC in untreated patients, including paclitaxel, docetaxel, topotecan, irinotecan, vinorelbine, and gemcitabine. In this century, two additional cytotoxic agents were evaluated: pemetrexed, a multitargeted antifolate agent evaluated as monotherapy in the relapse setting, and amrubicin, a topoisomerase II inhibitor that has produced impressive responses as first-line therapy. This chapter discusses first-line and second-line therapy for patients with extensive-stage SCLC.
First-Line Chemotherapy
Combination Chemotherapy
Given the large number of active agents in SCLC, the evaluation of combination regimens quickly ensued. In the 1970s, randomized trials demonstrated the superiority of combination chemotherapy over single-agent therapy. Furthermore, studies showed that simultaneous administration of multiple agents was more efficacious than the sequential administration of the same agents. Cyclophosphamide-based regimens were commonly used to treat SCLC, including cyclophosphamide, doxorubicin, and vincristine (CAV); cyclophosphamide, doxorubicin, and etoposide (CDE); and cyclophosphamide, etoposide, and vincristine (CEV).
After the introduction of cisplatin, randomized trials with a regimen of cisplatin and etoposide showed that this combination was as effective as CAV and less toxic. A meta-analysis of 36 trials demonstrated that regimens containing cisplatin and/or etoposide offered a significant survival advantage to patients with SCLC. Thus cisplatin and etoposide became the preferred regimen for the treatment of extensive-stage SCLC, yielding overall response rates of 65% to 85%, complete response rates of 10% to 20%, and a median survival of 8 months to 10 months. For patients with limited-stage SCLC, cisplatin and etoposide plus twice-daily thoracic radiotherapy was also considered the treatment of choice, producing an 87% overall response rate, a 56% complete response rate, a median survival of 23 months, and a 5-year survival rate of 44%. Carboplatin is frequently substituted for cisplatin because of its more favorable toxicity profile. One small randomized trial comparing cisplatin and etoposide with carboplatin and etoposide in patients with limited- and extensive-stage disease showed similar efficacy, but the carboplatin-based combination was significantly less toxic. A meta-analysis of individual data from 633 patients who participated in four clinical trials did not demonstrate any difference in efficacy between cisplatin- and carboplatin-based regimens, with a median survival of 9.6 months and 9.4 months, respectively. Significant differences in toxicity were found; more neutropenia, anemia, and thrombocytopenia occurred with carboplatin-based regimens, whereas more nausea, vomiting, neurotoxicity, and renal toxicity developed with cisplatin-based regimens.
Years elapsed before the discovery of newer cytotoxic agents such as the topoisomerase II inhibitors, taxanes, gemcitabine, and vinorelbine, which were shown to have antitumor activity in SCLC. Many studies have summarized the results from novel combinations that were evaluated in phase III trials ( Table 52.1 ). Enthusiasm for the combination of cisplatin and irinotecan (PI) arose when Japanese researchers halted their phase III trial prematurely after an interim analysis showed a survival benefit for PI over cisplatin and etoposide. One hundred and fifty-four patients were randomly assigned to receive either four cycles of etoposide (100 mg/m 2 ) on days 1, 2, and 3 with cisplatin (80 mg/m 2 ) on day 1 every 3 weeks or four cycles of irinotecan (60 mg/m 2 ) on days 1, 8, and 15, and cisplatin (60 mg/m 2 ) on day 1. Patients treated with PI had a significantly better overall response rate (84.4% vs. 67.5%; p = 0.02), median survival (12.8 months vs. 9.4 months), and 1-year survival rate (58.4% vs. 37.7%; p = 0.002) than patients treated with cisplatin and etoposide. The PI combination was associated with a higher rate of grade 3 or grade 4 diarrhea ( p = 0.01), whereas cisplatin and etoposide were associated with a higher rate of myelosuppression ( p = 0.0001). The Southwest Oncology Group (SWOG) conducted a confirmatory trial using the identical study design but found no survival benefit for PI. In this large trial of 651 patients, all efficacy parameters were very similar except for a trend toward improved progression-free survival time for PI (5.7 months vs. 5.2 months for cisplatin and etoposide; p = 0.07). Grade 3 or grade 4 neutropenia and thrombocytopenia were higher in the cisplatin and etoposide arm, whereas grade 3 or grade 4 nausea/vomiting and diarrhea were higher in the PI arm. A phase III superiority trial comparing a novel dose and schedule of the PI regimen (irinotecan [65 mg/m 2 ] with cisplatin [30 mg/m 2 ] given on days 1 and 8) with standard cisplatin and etoposide produced similar survival in both arms. In Europe, a different schedule of PI (irinotecan [65 mg/m 2 ] on days 1 and 8 with cisplatin [80 mg/m 2 ] on day 1) was assessed in comparison with standard cisplatin and etoposide. The data showed that the PI regimen was noninferior to cisplatin and etoposide, as hypothesized. The median overall survival rates were 10.2 months and 9.7 months, respectively, with a hazard ratio (HR) of 0.81 (95% confidence interval [CI], 0.61–1.01; p = 0.06). Overall response rates were 39% for PI and 47% for cisplatin and etoposide, and time to progression was 5.4 months and 6.2 months, respectively. The number of grade 3 or grade 4 adverse events was similar between the arms, but more patients in the PI arm had gastrointestinal toxicity and more patients in the cisplatin and etoposide arm had neutropenia. In 2013, Korean investigators reported the results from a phase III trial comparing PI with cisplatin and etoposide. Irinotecan was administered on days 1 and 8 with cisplatin on day 1, and cisplatin and etoposide were given in the standard fashion. The trial, however, did not demonstrate the superiority of PI (HR, 0.88; 95% CI, 0.73–1.05; p = 0.12). The median overall survival was 10.9 months for PI and 10.3 months for cisplatin and etoposide. The overall response rate was significantly higher for PI (62.3% vs. 48.2%; p = 0.0064), but progression-free survival was not significantly different (6.5 months vs. 5.8 months, respectively). The frequency of anemia, nausea, and diarrhea was greater with PI.
| Author (y) | Regimen | No. of Patients | Overall Response Rate (%) | Progression-Free Survival (mo) | Median Survival (mo) | 1-Year Survival Rate (%) |
|---|---|---|---|---|---|---|
| Noda et al. (2002) | PI PE | 77 77 | 84.4 a 67.5 | 6.9 b 4.8 | 12.8 c 9.4 | 58.4 37.7 |
| Lara, Jr. et al. (2009) | PI PE | 324 327 | 60 57 | 5.8 5.2 | 9.9 9.1 | 41 34 |
| Hanna et al. (2006) | PI PE | 221 110 | 48 44 | 4.1 4.6 | 9.3 10.2 | 35 35 |
| Zatloukal et al. (2010) | PI PE | 202 203 | 39 47 | 5.4 6.2 | 10.2 9.7 | 42 39 |
| Kim et al. (2013) | PI PE | 173 189 | 62 d 48 | 6.5 5.8 | 10.9 10.3 | NR NR |
| Hermes et al. (2008) | IC EC | 105 104 | NR NR | NR NR | 8.5 7.1 | 37 b 19 |
| Schmittel et al. (2011) | IC EC | 106 110 | 54 52 | 6.0 6.0 | 10.0 9.0 | 37 30 |
| Eckardt et al. (2006) | PT PE | 389 395 | 63 69 | 6.0 b 6.2 | 9.8 10.0 | 31 31 |
| Fink et al. (2012) | PT PE | 357 346 | 56 e 46 | 6.9 e 6.1 | 10.3 9.4 | 40 36 |
| de Jong et al. (2007) | CDE CT | 102 101 | 60 61 | 4.9 5.2 | 6.8 6.7 | 24 26 |
| Socinski et al. (2009) | PemC EC | 364 369 | 31 52 f | 3.8 5.4 g | 8.1 10.6 g | NR NR |
| Kotani et al. (2012) | AP IP | 142 142 | 78 72 | 5.1 5.7 | 15.3 18.3 | NR NR |
| Mavroudis et al. (2001) | PET PE | 62 71 | 50 48 | 11.0 b 9.0 | 9.5 10.5 | 38 37 |
| Reck et al. (2003) | CET CEV | 301 307 | 72 69 | 8.1 h 7.5 | 12.7 11.7 | 48 51 |
| Niell et al. (2005) | PET PE | 293 294 | 75 68 | 6.0 5.9 | 10.6 9.9 | 38 37 |
| Pujol et al. (2001) | PCDE PE | 117 109 | 76 b 61 | 7.2 i 6.3 | 10.0 9.3 | 40 j 29 |
Irinotecan has also been evaluated in combination with carboplatin. The IRIS study demonstrated superior survival for irinotecan plus carboplatin compared with oral etoposide plus carboplatin; however, overall survival in both study arms was low, at less than 9 months. Drug dosages and schedules were unconventional and lower than other published regimens, with irinotecan (175 mg/m 2 ) and carboplatin (area under the curve [AUC] 4) administered on day 1, and etoposide (120 mg/m 2 ) orally on days 1–5 with carboplatin (AUC 4) on day 1. Another trial, conducted in Germany, randomly assigned 216 patients to receive either irinotecan (50 mg/m 2 ) on days 1, 8, and 15 with carboplatin (AUC 5) or intravenous (IV) etoposide (140 mg/m 2 ) on days 1–3 with carboplatin (AUC 5). The irinotecan regimen was not found to be superior to the etoposide regimen in terms of overall survival (HR, 1.34; 95% CI, 0.97–1.85; p = 0.072). The median survival was 10 months and 9 months, respectively. The overall response rate and progression-free survival were similar for both treatment arms.
A meta-analysis of seven randomized trials including 2027 patients that compared irinotecan plus a platinum agent with etoposide plus a platinum agent showed a survival advantage for irinotecan regimens (HR, 0.81; 95% CI, 0.71–0.93; p = 0.003). No significant differences in progression-free survival or overall response rate were noted. Irinotecan regimens produced significantly less hematologic toxicity but more gastrointestinal toxicity than etoposide regimens. Overall, the data suggest that combinations of irinotecan or etoposide plus a platinum compound are reasonable options as first-line therapy for patients with extensive-stage SCLC.
Topotecan, a drug with activity for disease relapse, was evaluated in the frontline setting. Two large phase III trials with oral or IV topotecan plus cisplatin did not show a survival advantage over standard cisplatin and etoposide. Efficacy parameters were similar between the regimens, with median survival times of 9 months to 10 months. The IV topotecan regimen did produce a significantly higher overall response rate (56% vs. 46%; p = 0.01) and prolonged progression-free survival (7 months vs. 6 months; p = 0.004), but was associated with more hematologic toxicity.
Several other novel platinum combinations have been studied. One trial comparing paclitaxel plus carboplatin with CDE showed no benefit of the doublet over the standard regimen, but survival was modest in both arms, at less than 7 months. A phase III trial of the combination of pemetrexed and carboplatin unexpectedly showed inferior efficacy to standard treatment. In a previous randomized phase II study of pemetrexed plus cisplatin or carboplatin, the carboplatin arm produced a median survival of 10.4 months and was well tolerated. A phase III study, the Global Analysis of Pemetrexed in SCLC Extensive Stage, was designed to show noninferiority of pemetrexed (500 mg/m 2 ) and carboplatin (AUC 5) compared with etoposide and carboplatin. With 733 patients randomly assigned to treatment, the study was terminated prematurely when the predefined futility end point for progression-free survival showed inferiority of the experimental arm. In the final analysis, overall survival was inferior (HR, 1.56; 95% CI, 1.27–1.92; p < 0.01). The median overall survival was 8.1 months for pemetrexed and carboplatin and 10.6 months for etoposide and carboplatin. The median progression-free survival was 3.8 months for pemetrexed and carboplatin and 5.4 months for etoposide and carboplatin ( p < 0.01), and the overall response rate also favored the etoposide and carboplatin combination (52% vs. 31%; p < 0.001). Significant neutropenia and more febrile neutropenia were seen in the etoposide arm. By contrast, death during therapy or within 30 days was higher for the pemetrexed arm than for the etoposide arm (16% vs. 10%; p = 0.032), and the rate of toxicity-related death was higher (1.4% vs. 0%; p = 0.028).
Another novel cytotoxic agent with promising early results that failed to show a survival advantage in the phase III setting was amrubicin. A phase III randomized study showed that amrubicin plus cisplatin was inferior to PI. Two hundred and eighty-four patients were randomly assigned to receive either amrubicin (35 mg/m 2 to 40 mg/m 2 ) on days 1–3 and cisplatin (60 mg/m 2 ) every 3 weeks or cisplatin (60 mg/m 2 ) on day 1 and irinotecan (60 mg/m 2 ) on days 1, 8, and 15 every 4 weeks. The median overall survival for amrubicin plus cisplatin was 15 months, compared with 18.3 months for PI (HR, 1.33; 95% CI, 1.01–1.74; p = 0.681), and this result exceeded the noninferiority margin. The progression-free survival was 5.1 months for amrubicin plus cisplatin and 5.7 months for PI with an overall response rate of 78% and 72%, respectively. An increased incidence of grade 4 neutropenia (79% vs. 23%) and febrile neutropenia (32% vs. 11%) was found in the amrubicin plus cisplatin arm.
The favorable toxicity profiles of most of the newer agents led investigators to explore the possibility of integrating them into an active doublet (see Table 52.1 ). Three randomized trials evaluating the addition of paclitaxel to cisplatin and etoposide or carboplatin and etoposide did not produce a survival benefit over traditional doublets and were associated with increased toxicity. French investigators evaluated a four-drug regimen in which they added cyclophosphamide and 4′-epidoxorubicin to cisplatin and etoposide (PCDE). A significant improvement in the complete response rate (13% vs. 21%; p = 0.02) and overall survival (9.3 months vs. 10.5 months; p = 0.0067) was noted for PCDE. However, PCDE was associated with a significantly higher hematologic toxicity rate, with 22% of patients having a documented infection, compared with 8% in the cisplatin and etoposide arm ( p = 0.0038). Toxicity-related death rates were similar, at 9% for PCDE and 5.5% for cisplatin and etoposide. Since the early 1990s, there have been no major breakthroughs with newer chemotherapy agents in the first-line setting. A platinum agent plus etoposide or irinotecan remains the standard of care for the treatment of SCLC.
Alternative Chemotherapy Strategies
Alternative chemotherapy strategies have focused on modifying the dosage and schedules of established regimens, including dose intensification, alternating non–cross-resistant chemotherapy, and prolonged treatment durations. However, with the discovery of molecularly targeted agents, investigators have largely abandoned the pursuit of optimizing current chemotherapy regimens.
Dose Intensification
Dose intensity is defined as the dose per meter squared per week. Dose intensification can be accomplished by increasing the dose administered or by shortening the interval between doses (dose density). Results from preclinical tumor models suggested that one of the simplest ways to overcome drug resistance was dose escalation. In the late 1970s, Cohen et al. randomly assigned patients to receive either standard dosages of cyclophosphamide, methotrexate, and lomustine or a higher dose of cyclophosphamide and lomustine plus a standard dose of methotrexate. Patients treated in the high-dose arm had a higher overall response rate that led to prolonged survival, and a subset of these patients were long-term survivors. These data resulted in a series of seven randomized trials comparing high-dose and conventional-dose chemotherapy in patients with limited-stage and extensive-stage SCLC. Most of these trials were conducted in the 1980s and did not show a clinical benefit. The Spanish Lung Cancer Group reexamined this question in 2004. They compared high-dose epirubicin (100 mg/m 2 ) plus cisplatin (100 mg/m 2 ) administered on day 1 with standard cisplatin and etoposide (cisplatin [100 mg/m 2 ] on day 1 and etoposide [100 mg/m 2 ] on days 1–3) in 402 patients with SCLC. Efficacy results were similar between the arms. A study of patients with limited-stage disease published in 1989 showed a superior 2-year survival rate of 43% when the dose of cisplatin and cyclophosphamide was increased by 20% in the first cycle of a PCDE regimen, compared with a 2-year survival rate of 23% for standard PCDE.
Dose-dense regimens have shown mixed results. One such combination was an intense weekly regimen of cisplatin (25 mg/m 2 ) for 9 consecutive weeks, vincristine (1 mg/m 2 ) on even weeks for 9 weeks, and doxorubicin (40 mg/m 2 ) and etoposide (80 mg/m 2 ) on days 1–3 on odd weeks for 9 weeks (CODE). This was the first regimen to be associated with an impressive 2-year survival rate of 30% among 48 patients with extensive-stage SCLC. The investigators were able to administer close to the intended full doses of all four agents, thereby increasing the dose intensity by twofold. The National Cancer Institute of Canada–Cancer Treatment Group (NCIC–CTG) in collaboration with SWOG conducted a phase III trial comparing the CODE regimen and conventional alternating CAV/cisplatin and etoposide for patients with extensive-stage SCLC. Response rates were higher in the CODE arm, but no differences were found in progression-free or overall survival. Although rates of neutropenia and fever were similar, toxicity-related deaths occurred in 9 of 110 patients receiving CODE, compared with one of 109 patients receiving CAV/cisplatin and etoposide ( p = 0.42). Given the high toxicity-related death rate and similar efficacy, CODE was not recommended. Japanese investigators subsequently demonstrated that adding granulocyte colony-stimulating factor (G-CSF) to CODE increased the mean total dose intensity received, reduced neutropenia and febrile neutropenia, and significantly prolonged survival (59 weeks vs. 32 weeks; p = 0.0004). This led to a phase III trial of CODE plus G-CSF compared with CAV/cisplatin and etoposide. The response rate was significantly higher for CODE, but there was no survival advantage. The toxicity-related death rate with CODE plus G-CSF was low, with only four reported deaths.
European investigators evaluated the dose-dense strategy with or without colony stimulation in seven phase III trials published between 1993 and 2002. Two trials showed a survival advantage for the dose-dense arm, and the other trials reported similar outcomes between the standard and experimental arms. The trial by Steward et al. showed a significant prolongation in survival with dose intensification of vincristine, ifosfamide, carboplatin, and etoposide (ICE) chemotherapy compared with standard dosing of this regimen. The median survival time was 443 days in the dose-dense arm and 351 days in the standard arm ( p = 0.0014), with 2-year survival rates of 33% and 18%, respectively. There was no difference in response rate, despite the 26% increase in dose intensity in the experimental arm. The British Medical Research Council randomly assigned 403 patients to receive doxorubicin, cyclophosphamide, and etoposide in two or three weekly schedules. In this trial, a 34% escalation in dose density was achieved. Although the response rates in the two arms were similar, a significant improvement was found in the complete response rate in the dose-dense arm (40% vs. 28%; p = 0.02) that translated into a 2-year survival benefit (13% vs. 8%; p = 0.04). Subgroup analysis showed that the survival advantage among patients with extensive disease was as large as that for patients with limited disease.
A possible explanation for the failure of the previous trials is that the dose intensity was insufficient to produce a survival benefit. To definitively answer the question about dose intensification, studies were conducted using stem cell rescue, which would allow for a 200% to 300% dose escalation of chemotherapy. Multiple small studies have shown this approach to be feasible. The original studies focused on patients who had a response with conventional cytotoxic therapy and then received high-dose consolidation with stem cell rescue. A randomized trial testing this late-intensification strategy was reported by Humblet et al. in 1987. One hundred and one patients received standard induction chemotherapy, and 45 patients with chemotherapy sensitivity were randomly assigned to receive either one additional cycle with high-dose cyclophosphamide, carmustine, and etoposide or conventional doses of the same drugs. In this highly selected group of patients, the median overall survival was 68 weeks for the high-dose arm compared with 55 weeks for the conventional therapy ( p = 0.13).
Because of its improved safety and feasibility, peripheral blood stem cell transplantation has largely replaced autologous marrow transplants. Japanese investigators reported promising results from a phase II study of high-dose ICE with autologous peripheral blood stem cell transplantation in 18 patients with limited-stage SCLC after concurrent, hyperfractionated chemoradiation therapy. The complete response rate was 61% and the median survival time was 36.4 months. One toxicity-related death was reported. At the time of publication, a randomized trial based on these results was ongoing. Three randomized trials using high-dose ICE chemotherapy with peripheral blood rescue as first-line treatment for SCLC have also been reported. The largest trial included 318 patients with predominantly limited-stage SCLC and compared six cycles of a dose-dense ICE regimen every 14 days, with G-CSF–mobilized whole-blood hematopoietic progenitors, with six cycles of the standard 28-day ICE regimen. Despite doubling of the median dose intensity with the dose-dense regimen (182% vs. 88%, respectively), the median survival time and the 2-year survival rate were comparable (14.4 months and 22% vs. 13.9 months and 19%, respectively). By contrast, an identical study by Buchholz et al. was halted after 70 patients were enrolled. They reported a favorable median survival of 30.3 months ( p = 0.001), a 2-year survival rate of 55%, and a time to progression of 15 months ( p = 0.0001) for the dose-intense arm compared with a median survival of 18.5 months, a 2-year survival rate of 39%, and a time to progression of 11 months for the standard-dose arm in this small, single-institution study. The European Group for Blood and Marrow Transplant conducted a similar study. The study was closed after 140 of the planned 340 patients were enrolled because of poor accrual. The median dose intensity for the high-dose arm was 293%, but this dose did not yield a survival benefit; the median survival time was 18.1 months and the 3-year survival rate was 18% for the high-dose arm, compared with 14.4 months and 19%, respectively, for the standard ICE arm. None of the subgroups benefited from high-dose ICE.
Overall, most trials using a dose-intensification strategy did not show a survival advantage over standard therapy for patients with extensive-stage SCLC, and the higher doses were typically associated with greater toxicity. This approach should be abandoned in patients with extensive disease. In limited-stage SCLC, the optimal drug doses remain unclear, with several studies suggesting a possible benefit. Continued evaluation of dose intensity in the curative setting is reasonable.
Alternating Non–Cross-Resistant Chemotherapy Regimens
To achieve maximal antitumor effects using multiple active agents, they should be administered simultaneously at their optimal single-agent dose. However, because drug toxicities often overlap, strict adherence to this approach is often not possible in the clinical setting. In the 1980s, Goldie et al. suggested that alternating two non–cross-resistant chemotherapy regimens of relatively comparable efficacy could minimize the development of drug resistance while avoiding excessive host toxicity. This strategy was particularly appealing for SCLC because both CAV and cisplatin and etoposide are highly active against SCLC and contain agents from divergent drug classes. Three randomized phase III trials were performed to evaluate CAV and CAV alternating with cisplatin and etoposide. Studies from the United States and Japan showed similar efficacy between the study arms, whereas the NCIC–CTG reported superior efficacy for the alternating regimen, with overall response rates of 80% and 63%, respectively ( p < 0.002), and survival times of 9.6 months and 8.0 months ( p = 0.03). Investigators at the NCIC–CTG went on to test this approach in patients with limited-stage SCLC. Patients were randomly assigned between two induction regimens, either alternating CAV/cisplatin and etoposide or sequential therapy with three cycles of CAV followed by three cycles of cisplatin and etoposide. Chemotherapy was followed by radiotherapy in patients with a disease response. The therapeutic outcomes in the study groups were not significantly different. SWOG conducted a similar study and found no advantage for the alternating CAV/cisplatin and etoposide regimen over the etoposide, vincristine, doxorubicin, and cyclophosphamide regimen in patients with limited-stage disease.
The European Organization for Research and Treatment of Cancer reported a trial testing two relatively non–cross-resistant regimens: CDE and vincristine, carboplatin, ifosfamide, and mesna. Patients with extensive-stage SCLC were randomly assigned to receive either a maximum of five courses of CDE or an alternating regimen consisting of CDE in cycles 1, 3, and 5 and vincristine, carboplatin, ifosfamide, and mesna in cycles 2 and 4. The trial accrued only 148 of the 360 planned patients. The median survival time was 7.6 months in the standard arm and 8.7 months in the alternating arm ( p = 0.243).
Although no survival benefit for the alternating drug hypothesis was demonstrated, the emergence of newer active agents for the treatment of SCLC justified revisiting this strategy. The North Central Cancer Treatment Group conducted a trial of etoposide and cisplatin alternating with topotecan and paclitaxel. The overall response rate was 77%, including four complete responses among 44 evaluable patients. The median survival was 10.5 months, with 1- and 2-year survival rates of 37% and 12%, respectively. This alternating regimen was associated with a high rate of grade 3 and grade 4 neutropenia (95%) despite the use of filgrastim in cycles 2, 4, and 6. The Hellenic Oncology Research Group treated 36 previously untreated patients with extensive-stage SCLC with cisplatin and etoposide alternating with topotecan. The overall response rate was 64%, and 14% of patients had a complete response. Grade 3 and grade 4 neutropenia occurred in 39% of patients during the cycles of cisplatin and etoposide and in 55% after the topotecan treatment. These limited data incorporating newer chemotherapy agents into an alternating strategy were disappointing. Taken together, alternating newer and/or older cytotoxic agents to overcome drug resistance is an unsuccessful strategy and should not be pursued.
Treatment Duration and Maintenance Therapy
The ideal number of chemotherapy cycles for SCLC has not been defined; however, four to six cycles is considered the standard based on the results from the randomized trials described previously. Clinical trials specifically designed to investigate the role of prolonged treatment using a consolidation or maintenance approach have been performed. Three of 14 trials produced positive results. All three trials were initiated in 1982. In two trials of patients with limited-stage SCLC, two cycles to four cycles of consolidation therapy with cisplatin and etoposide were given to patients who had a response after induction CAV with or without thoracic radiotherapy. The remaining trial randomly assigned patients with nonprogressing limited or extensive-stage disease to four additional cycles of CEV or observation. Although this trial showed that four cycles of CEV were inferior, a second randomization to salvage chemotherapy compared with palliative care given at the time of disease progression demonstrated that the subset of patients who received eight cycles of CEV with or without salvage therapy did not live longer than patients who received four cycles of CEV and salvage therapy at the time of relapse. The role of consolidation and/or maintenance therapy with topotecan was evaluated by the Eastern Cooperative Oncology Group (ECOG). Two hundred and twenty-three patients with nonprogressing, extensive-stage SCLC were randomly assigned to receive either four cycles of topotecan or observation. Progression-free survival from the date of randomization was significantly better with topotecan than with observation alone (3.6 months vs. 2.3 months; p < 0.001), but overall survival from randomization was not significantly different between the arms (8.9 months vs. 9.3 months; p = 0.43). A meta-analysis of 14 randomized trials of maintenance chemotherapy involving 1806 patients was published in 2013. Maintenance chemotherapy failed to increase survival when compared with observation alone, with an odds ratio for 1-year mortality of 0.88 (95% CI, 0.66–1.19; p = 0.414). Maintenance treatment did, however, significantly prolong progression-free survival for patients with extensive-stage disease (HR, 0.72; 95% CI, 0.58–0.89; p = 0.003). This benefit was limited to patients who had switch maintenance therapy. Overall, chemotherapy after four to six cycles of a combination regimen is not warranted. Patients should be followed closely for signs and symptoms of relapse. Clinical trials evaluating maintenance regimens with molecularly targeted agents were ongoing at the time of publication.
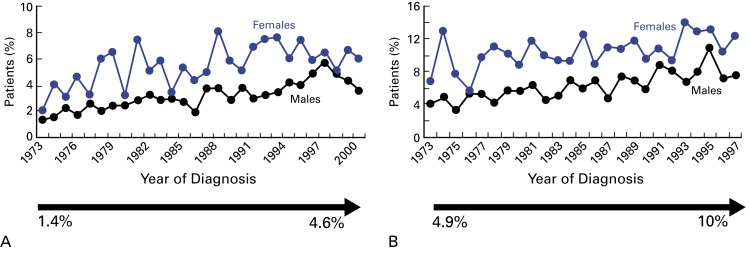
In summary, a platinum-based doublet with etoposide or irinotecan remains the standard of care for patients with SCLC. Although extensive research has not altered the standard of care for SCLC in many years, an analysis of the Surveillance, Epidemiology, and End Results database showed a modest but significant improvement in survival with current therapies. In 1973, the 2-year survival rate for extensive-stage SCLC was 1.5%, compared with 4.6% in 2000, whereas the 5-year survival rate for limited-stage SCLC increased from 4.9% to 10% during a similar period ( Fig. 52.1 ). Moreover, the recent genomic characterization of SCLC provides optimism that novel, efficacious agents are forthcoming.
First-Line Chemotherapy for Older Patients
According to the Surveillance, Epidemiology, and End Results database, 42% of patients with SCLC were aged 70 years or older at diagnosis. Similar age distributions are seen worldwide. Furthermore, the 5-year survival rate for SCLC was significantly worse for older patients than for younger patients ( p < 0.0001) and had not changed over the 15 years studied. For the period between 1998 and 2003, 5-year survival rates were 6.5% for patients younger than 70 years of age, 3.4% for patients aged 70 to 79 years, and 2.4% for patients aged 80 years or older.
Retrospective reviews to identify prognostic factors in SCLC have shown variable results with regard to age. The largest experience comes from SWOG. An analysis of 2580 patients enrolled in six SWOG studies, of whom approximately 10% were older, showed that patients over the age of 70 years had a significant risk of death, with a HR of 1.5 ( p ≤ 0.0001) for limited-stage disease and a HR of 1.3 ( p = 0.006) for extensive-stage disease. By contrast, a smaller study from 1991 reviewed 614 patients with limited- and extensive-stage disease from the University of Toronto clinical trial database and showed that age over 70 years was not a significant predictor of a poorer outcome. A meta-analysis published by Pignon et al. in 1992 examined 2140 patients with limited-stage disease from 13 randomized trials that were designed to determine the role of thoracic radiotherapy combined with chemotherapy compared with chemotherapy alone. The relative risk of death in patients older than 70 years of age receiving combination therapy was 1.07, higher than that of older patients receiving chemotherapy alone. Since this meta-analysis, a review of two NCIC–CTG trials, BR.3 and BR.6, involving 618 patients with limited-stage SCLC who received the same chemotherapy regimen, showed no difference in survival between patients aged younger and older than 70 years. In the United States Intergroup study comparing once-daily and twice-daily radiotherapy for limited-stage SCLC, survival of younger patients was better than that for patients older than 70 years of age, with borderline significance ( p = 0.051).
Advanced age has been perceived as a strong rationale for the use of less aggressive therapies or no therapy, for fear of increasing toxicity. The literature is conflicting on this topic. Some retrospective reviews have reported that older age is associated with an increased risk of chemotherapy-related morbidity and mortality, whereas other studies have shown that despite toxicity and dose reductions, older patients receive a survival benefit with chemotherapy and/or radiotherapy compared with no treatment. A review by the Royal Marsden Hospital investigated the survival outcomes of 322 older patients (aged 70 years or older) with SCLC treated with chemotherapy from 1982 to 2003. Patients treated between 1995 and 2003 had a median survival of 43 weeks and a 1-year survival rate of 37%, compared with 25 weeks and 14%, respectively, for patients treated between 1982 and 1994 ( p < 0.001). Patients who received a platinum combination had significantly better survival ( p < 0.001) than patients who received single agents or another combination. No survival difference was found between a cisplatin and a carboplatin regimen. In a 2005 analysis of 54 older patients with limited-stage disease who participated in the North Central Cancer Treatment Group phase III trial of cisplatin and etoposide plus twice-daily or once-daily thoracic radiotherapy, survival was not different from that of their younger counterparts despite higher toxicity. These results corroborate the United States Intergroup findings. More data are needed from phase III trials regarding age-specific outcomes.
To formally address the question of dose tolerability among older patients with SCLC, Ardizzoni et al. randomly assigned patients aged 70 years or older either to four cycles of cisplatin (25 mg/m 2 ) on days 1–2 with etoposide (60 mg/m 2 ) IV on days 1–3 every 3 weeks (the attenuated-dose regimen; 28 patients) or to cisplatin (40 mg/m 2 ) on days 1–2 plus etoposide (100 mg/m 2 ) IV on days 1–3 with prophylactic G-CSF (the full-dose regimen; 67 patients). Patients treated with the attenuated-dose regimen had a poorer outcome. The response rate was 39% in the attenuated-dose arm and 68% in the full-dose arm, with 1-year survival rates of 18% and 39%, respectively. No grade 3 or grade 4 myelotoxicity was reported in the attenuated-dose group, but 10% was noted in the full-dose group. There was one toxicity-related death in the full-dose arm. The median number of cycles was four in both groups; 75% of patients in the attenuated-dose group and 72% in the full-dose group completed all planned cycles. Japanese investigators conducted a phase III trial to test whether nearly full doses of carboplatin and etoposide were superior to their standard regimen for older patients, which consisted of a split dose of cisplatin and etoposide. Older was defined as an age of 70 years or more with an ECOG performance status of 0 to 2. Patients younger than 70 years of age with a performance status of 3 were also allowed to participate. A total of 220 patients with extensive-stage SCLC were entered in the study, with 110 patients receiving carboplatin (AUC 5) on day 1 and etoposide (80 mg/m 2 IV) on days 1–3 every 3–4 weeks for four cycles, and 109 patients receiving cisplatin (25 mg/m 2 ) on days 1–3 with etoposide (80 mg/m 2 IV) on days 1–3 every 3–4 weeks for four cycles. G-CSF was recommended in both treatment arms. As many as 92% of the patients met the criteria for older status, and 8% were poor risk. Objective response rates were identical in both treatment arms (73%). The median survival for the carboplatin and etoposide arm was 10.6 months and the 1-year survival rate was 41%, as compared with 9.9 months and 35%, respectively, for the split-dose cisplatin and etoposide arm. The rate of grade 3 or grade 4 neutropenia was high in both arms (95% for the carboplatin and etoposide arm and 90% for the split-dose cisplatin and etoposide arm). A significant difference in the rate of grade 3 or 4 thrombocytopenia was noted (56% for carboplatin and etoposide and 16% for split-dose cisplatin and etoposide; p = 0.01). There were four treatment-related deaths, three in the carboplatin and etoposide arm and one in the split-dose cisplatin and etoposide arm. The authors concluded that either regimen was a reasonable treatment option.
The optimal chemotherapy regimen for older patients with SCLC is not known. We have learned that chronologic age should not be the sole determinant of treatment decisions. Physiologic age determined by comorbidities and performance status provides a clearer framework for guiding treatment decisions. Among patients aged 70 years or older, categories such as fit elderly (performance status of 0 or 1) and frail (performance status of 2–4) are emerging as beneficial terms in both the clinical and research settings. Despite the limited data, we are encouraged that a survival benefit can be achieved in a subset of older patients with acceptable toxicity. As the older population continues to increase, it is crucial that we develop evidence-based treatment plans. Additional clinical research in this population is needed.
First-Line Chemotherapy for Patients With Poor Performance Status
Performance status is universally recognized as an independent prognostic factor and typically correlates with the extent of tumor burden. Several retrospective studies of large databases have confirmed that shorter survival times among patients with SCLC are associated with poor performance status. Despite poor survival, patients with a performance status of 2 have routinely been eligible for clinical trials because our experience has taught us that patients whose poor performance status is attributed to tumor burden can respond to treatment with meaningful symptom palliation, improved performance status, and prolonged survival. However, the number of patients with poor performance status enrolling in clinical trials is low, and outcome data specific to performance status are not available.
Clinical trials specifically including patients with poor performance status are few and were conducted more than 20 years ago. Two trials evaluated the oral formulation of etoposide because it was presumed to be efficacious but less toxic. The first study randomly assigned previously untreated patients with a performance status of 2–4 to either oral etoposide (50 mg twice a day) for 10 days (171 patients) or standard chemotherapy with cisplatin and etoposide or CAV (168 patients). The primary end point was palliation of symptoms at 3 months. The data safety and monitoring board stopped the trial early because of an inferior survival rate with oral etoposide. Survival was 130 days in the oral etoposide arm and 183 days in the standard arm ( p = 0.03). Palliation rates were similar in both arms (41% vs. 46%, respectively). Grade 2 or greater hematologic toxicity was low in both arms (21% vs. 26%, respectively). The second trial, conducted by the London Lung Cancer Group, enrolled patients younger than 75 years of age with a performance status of 2 or 3 or patients equal to or older than 75 years of age with any performance status to receive 100 mg etoposide orally for 5 days (75 patients) or CAV alternating with cisplatin and etoposide (80 patients). The authors hypothesized that oral etoposide would produce a similar survival rate but with improved quality of life. This study, too, was stopped prematurely because of a significantly inferior survival rate in the oral etoposide arm. The median survival was 4.8 months, with a 1-year survival rate of 9.8% for oral etoposide, compared with 5.9 months and 19.3%, respectively, for CAV/cisplatin and etoposide ( p < 0.05). Grade 3 and grade 4 toxicities were infrequent and similar between the treatment groups, except that more nausea and vomiting were reported in the CAV/cisplatin and etoposide arm. In another study, the Medical Research Council Lung Cancer Working Party randomly assigned 310 patients with poor performance status either to a four-drug regimen of etoposide, cyclophosphamide, methotrexate, and vincristine (control arm) or to a less intense two-drug regimen of etoposide and vincristine. No differences were found in symptom palliation, response rates, or survival times between the groups; however, more early deaths occurred with the four-drug regimen. Grade 2 or greater hematologic toxicity and mucositis were also worse with the four-drug regimen.
Despite the lack of data, experts generally agree that if a poor performance status is due to the disease itself, patients should be offered standard platinum-based chemotherapy with close monitoring because they have a reasonable chance of symptom palliation and prolonged survival.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree