The thyroid gland
The thyroid gland is one of the largest endocrine glands in the body and is located immediately below the larynx and anterior to the upper part of the trachea. It consists of two lateral lobes connected by a narrow band of tissue, called the isthmus. The isthmus usually overlies the region from the second to the fourth cartilages. The lobes of the thyroid contain many hollow, spherical structures referred to as follicles, the functional units of the thyroid gland. Each follicle is filled with a thick substance called the colloid. The major constituent of the colloid is a large glycoprotein called thyroglobulin (Tg). Unlike other endocrine glands, which secrete their hormones once they are produced, the thyroid gland stores considerable amounts of thyroid hormones in the colloid until they are required for the body. The thyroid gland secretes two major hormones, which are required for the normal operation of a variety of physiologic processes affecting virtually every organ system in the body. Regulation of thyroid hormone secretion is tightly controlled by the hypothalamic–pituitary–thyroid axis.
Physiology of the thyroid–pituitary–hypothalamic axis
The thyroid gland synthesizes the two hormones, thyroxine (T 4 ) and 3,5,3′-triiodothyronine (T 3 ). The thyroid cells extract iodine from the circulation. In the thyroid cells, the iodine is oxidized in the presence of H 2 O 2 and thyroid peroxidase (TPO) and immediately bound to Tg. This iodinated protein moves to the apex of the thyroid cell where tyrosine is iodinated into mono- and diiodotyrosine. At the apex of the thyroid cell, the mono-iodotyrosine and diiodotyrosine molecules are coupled to form T 3 and T 4 , respectively. The synthesis of T 4 and T 3 takes place on the surface of Tg.
These hormones are then stored in the lumen of the thyroid follicle. During secretion, the intrafollicular contents are incorporated by endocytosis, then hydrolyzed, and T 4 , T 3 , and Tg molecules are secreted into circulation. All these processes (the uptake of iodine, oxidation, hormone synthesis, and secretion of the thyroid hormones) are stimulated by thyroid-stimulating hormone (TSH), which is secreted from the anterior pituitary gland ( Fig. 3.1 ) in response to thyrotropin-releasing hormone (TRH) release from the hypothalamus. Virtually every aspect of thyroid hormone synthesis and secretion is stimulated by TSH. In addition to regulating thyroid hormone secretion, it maintains structural integrity of the gland. When TSH is absent, the thyroid gland atrophies, and sustained TSH stimulation results in increased follicular cell size (hypertrophy) and number (hyperplasia).
When serum thyroid hormone concentrations decline, TRH is released and circulates to the anterior pituitary to stimulate secretion of TSH, and release of TSH by the pituitary stimulates the thyroid gland to produce thyroid hormones and secrete them into circulation. Conversely, circulating T 3 and T 4 inhibit TSH secretion, thereby decreasing subsequent thyroid hormone synthesis and secretion.
Peripheral tissues, particularly liver and kidney, play an important role in storage, metabolism, and production of active thyroid hormone (T 3 ). In these organs, an enzyme, deiodinase, converts T 4 to T 3 . Eighty percent of T 3 in circulation is derived through this peripheral mechanism, whereas the rest of the T 3 is secreted directly from the thyroid gland. The liver also synthesizes three proteins that bind thyroid hormones in circulation: thyroid-binding globulin (TBG), transthyretin (TTR; previously called thyroid-binding prealbumin), and albumin. In addition to intrathyroidal stores, the extrathyroidal tissues also store thyroid hormone with a volume of distribution of about 10 L.
T 4 is the principal hormone secreted by the thyroid gland. All the T 4 in the circulation is derived from thyroidal secretion. In contrast, only about 20% of circulating T 3 is of thyroidal origin. Most of the T 3 in blood is produced enzymatically in nonthyroidal tissues by 5′-monodeiodination of T 4 . In fact, T 4 appears to function as a prohormone for the production of the more biologically active form of thyroid hormone, T 3 . In the circulation, most (~99.98%) of the T 4 is bound to specific plasma proteins, TBG (60%–75%), TTR/prealbumin (TBPA) (15%–30%), and albumin (~10%). Approximately 99.7% of T 3 in the circulation is bound to plasma proteins, specifically TBG. This represents a 10-fold weaker protein binding than seen for T 4 . Thus only 0.02% of T 4 and 0.3% of T 3 are free. Protein-bound thyroid hormones do not enter cells and are thus considered to be biologically inert and function as storage reservoirs for circulating thyroid hormone. In contrast, the minute-free hormone fractions readily enter cells and exert their biological effects. In the pituitary, the negative feedback of thyroid hormone on TSH secretion is mediated primarily by T 3 that is produced at the site from the free T 4 (FT 4 ) entering the thyrotroph cells.
A dynamic equilibrium exists in circulation between the free and bound forms. The tissue activity of the thyroid hormone is provided chiefly by the free hormones, although some bound thyroid hormone is also available to tissues for utilization. In most of the tissues, the circulating free T 3 (FT 3 ) is responsible for the biological effect of the thyroid hormone. Some tissues, however, utilize circulating T 4 as well. The circulating FT 4 is converted in the pituitary and probably brain tissues by local deiodinase type II into intracellular T 3 . This T 3 produced within the cells augments the biological effect of the FT 3 derived from circulation.
The cellular effects of the thyroid hormones are produced through T 3 receptors. Two types of thyroid hormone receptors (TRs) have been identified, each with two isoforms: Thyroid receptor (TR) α-I, TR α-II, TR β-I, and TR β-II. Although α-I and the β isoforms are utilized by T 3 to produce the thyroid hormone effect, the α-II isoform may antagonize the T 3 function. This T 3 receptor is unique in the superfamily in that it remains attached to chromatin in the absence of T 3 and inhibits the transcription of genes stimulated by T 3 . Although in most of the tissues, the ligand-bound receptor bound to thyroid hormone response elements (TRE) increases the transcription of T 3 -responsive genes, in the pituitary gland, T 3 -bound receptor bound to TRE will suppress the production of TSH.
Roughly 90 µg of T 4 , 30 µg of T 3 , and 47–50 mmol of reverse T 3 (rT 3 ) are produced daily. Both the thyroid gland and extrathyroidal tissues are involved in T 3 formation. The secretion of the daily amounts of hormones by the thyroid is fairly constant and is stimulated by the hypothalamic–pituitary axis but inhibited by circulating FT 4 and FT 3 . TSH is secreted episodically. The hypothalamic hormone TRH stimulates the synthesis and secretion of TSH by the pituitary gland. Somatostatin and dopamine dampen the secretion of TSH. Environmental influences can also affect the hypothalamic–pituitary axis. For example, the exposure to cold immediately after birth results in a sudden rise in TSH secretion. The hypothalamic–pituitary axis attempts to maintain a constant supply of thyroid hormones to peripheral tissues. The circulating free thyroid hormones T 4 and T 3 exercise an inhibitory influence on the secretion of TSH and TRH. Thus a regulated hypothalamic-pituitary-thyroid-peripheral tissue axis maintains a fairly constant circulating free thyroid hormone blood level. When the free hormone levels decline, the TSH increases, acting to normalize the circulating FT 4 and FT 3 concentrations. On the other hand, when the FT 4 and FT 3 concentrations increase, the TSH secretion is inhibited.
Metabolism
Thyroid hormones are metabolized by a number of processes including deiodination, sulfation, conjugation, and side-chain cleavage. The major route of metabolism is through deiodination. T 4 is metabolized chiefly by deiodinases that remove iodine atoms sequentially. Ultimately, thyronine with no iodine atoms is produced. The removal of iodine from the outer ring as the first step produces T 3 , the active hormone. The enzyme deiodinase I is present in the liver and kidney. It has an active site for selenium and at pH 6.5–7.5 removes the iodine from the outer ring, producing T 3 . A similar enzyme present in the brain (deiodinase II) also produces T 3 by removing iodine from the outer ring. It does not require selenium for its activity and is active at pH 6.5–7.5. It produces T 3 in the brain and pituitary gland. The inner ring deiodination by deiodinase I at pH 8.0–8.5 and by deiodinase III (skin, brain, and placenta) produces rT 3 , which is inactive. The subsequent deiodination of the T 3 and rT 3 produces inactive molecules. The production of T 3 by deiodinase I can be decreased in diseases of the liver and kidney, systemic diseases, as well as by certain drugs.
Many bodily functions are influenced by thyroid hormones. Although thyroid hormones do not target a specific site, they affect nearly every tissue in the body, causing a variety of physiologic responses. One of the most significant effects of the thyroid hormones is on the basal metabolic rate, through oxygen consumption and heat production. Through the action of thyroid hormones, cellular oxidation is induced in most tissues, resulting in increased oxygen consumption and heat production. The thyroid hormones also stimulate carbohydrate metabolism. Use of glucose by cells is increased as is the rate of glucose absorption by the gastrointestinal tract. Calcium, water balance, and liver function are also influenced by thyroid hormones. In hypothyroidism, hypercalcemia occurs and glucuronic acid conjugation by the liver is impaired. Also, water is retained in extracellular compartments. Furthermore, because thyroid hormones stimulate carbohydrate metabolism, they are necessary for lipogenesis in liver and fat cells. Mobilization of fatty acids from fat tissue occurs in presence of thyroid hormones, which increases plasma free fatty acids (FFAs). Thyroid hormones also influence cholesterol levels.
Signs and symptoms
A number of signs and symptoms are well-established manifestations of thyroid dysfunction. Risk factors identifiable in personal history include previous thyroid dysfunction, goiter, surgery or radiotherapy affecting the thyroid gland, diabetes, vitiligo, pernicious anemia, leukotrichia, medications such as lithium, or iodine-containing compounds. Risk factors identifiable in family history include thyroid disease, pernicious anemia, diabetes, and primary adrenal insufficiency.
Abnormal results in commonly obtained laboratory tests are an indication of thyroid dysfunction. In hypothyroidism, there may be accompanying hypercholesterolemia, hyponatremia, anemia, creatine kinase and lactate dehydrogenase elevations, and hyperprolactinemia, and for hyperthyroidism, accompanying features may include hypercalcemia, alkaline phosphatase elevation, and hepatocellular enzyme elevation. The above clinical/laboratory findings warrant thyroid function testing, especially if they are sustained for 2 weeks or more, occur in combination, have not been present previously during documented euthyroidism, or occur in individuals with increased risk of thyroid disease.
Screening for thyroid dysfunction
Thyroid dysfunction meets many criteria for a condition that completely justifies population screening: The prevalence of various forms of thyroid dysfunction are substantial; overt hypothyroidism and hyperthyroidism have well-established clinical consequences; the serum TSH assay is an accurate, widely available, safe, and relatively inexpensive test, diagnostic for all common forms of hypo- and hyperthyroidism; and effective therapies for both hypo- and hyperthyroidism are available.
Screening of all newborn children for hypothyroidism is well accepted. In addition, serum TSH measurement in adults every 5 years has been shown in decision analysis to be cost-effective, especially in women and older persons. The American Thyroid Association guidelines recommend that all adults have serum TSH measured beginning at age 35 years and every 5 years thereafter. More frequent screening may be appropriate in individuals at high risk of developing thyroid dysfunction. With the advent of sensitive TSH measurements, serum TSH measurement is the single most reliable test to diagnose all common forms of hypo- and hyperthyroidism, especially in the ambulatory setting.
Variables affecting testing of thyroid function
For practical purposes, variables such as age, gender, race, season, phase of menstrual cycle, cigarette smoking, exercise, fasting, or phlebotomy- induced stasis have minor effects on the reference intervals for thyroid tests in ambulatory adults. Physiological variables include the serum TSH/FT 4 relationship, age, pregnancy, and biologic variation.
The serum TSH/FT 4 relationship
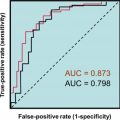
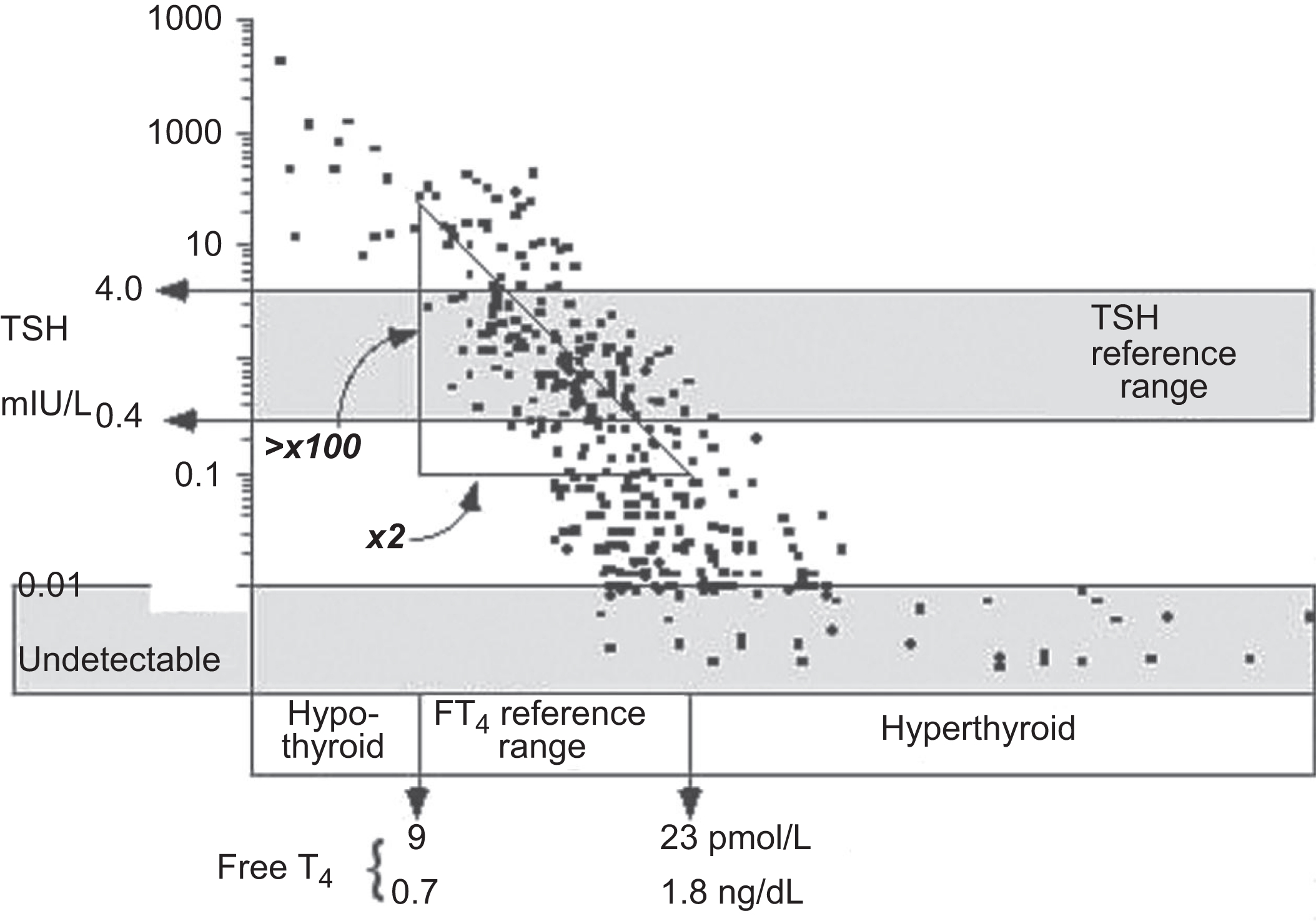
With the advent of ultrasensitive TSH assays, the strategy for screening for thyroid dysfunction is a TSH-first algorithm with testing for FT 4 when the TSH is above or below range. It is important to understand the normal relationship between serum levels of FT 4 and TSH when interpreting thyroid tests. An intact hypothalamic–pituitary axis is a prerequisite for TSH measurements to be used to determine primary thyroid dysfunction. When hypothalamic–pituitary function is normal, a log/linear inverse relationship between serum TSH and FT 4 concentrations is produced by negative feedback inhibition of pituitary TSH secretion by thyroid hormones. Thus it follows that high TSH and low FT 4 are characteristic of primary hypothyroidism, and low TSH and high FT 4 are characteristic of primary hyperthyroidism. In fact, now that the sensitivity and specificity of TSH assays have improved, it is recognized that the indirect approach (serum TSH measurement) offers better sensitivity for detecting thyroid dysfunction than does FT 4 testing.
There are two reasons for using a TSH-centered strategy for ambulatory patients. As shown in Fig. 3.1 , serum TSH and FT 4 concentrations exhibit an inverse log/linear relationship such that small alterations in FT 4 will produce a much larger response in serum TSH.
Furthermore, twin studies have demonstrated that each individual has a genetically determined FT 4 set-point. Any mild FT 4 excess or deficiency will be sensed by the pituitary, relative to that individual’s FT 4 set-point, and cause an amplified, inverse response in TSH secretion. It follows that in the early stages of developing thyroid dysfunction, a serum TSH abnormality will precede the development of an abnormal FT 4 because TSH responds exponentially to subtle FT 4 changes that are within the population reference limits. This is because population reference limits are broad, reflecting the different FT 4 set-points of the individual members of the cohort of normal subjects studied.
Serum FT 4 measurement is a more reliable indicator of thyroid status than TSH when thyroid status is unstable, such as during the first 2–3 months of treatment for hypo- or hyperthyroidism. Patients with chronic, severe hypothyroidism may develop pituitary thyrotroph hyperplasia that can mimic a pituitary adenoma but resolves after several months of levothyroxine (L-T 4 ) replacement therapy. In hypothyroid patients suspected of intermittent or noncompliance with L-T 4 replacement therapy, both TSH and FT 4 should be used for monitoring. Noncompliant patients may exhibit discordant serum TSH and FT 4 values (high TSH/high FT 4 ) because of persistent disequilibrium between FT 4 and TSH.
Currently, measurement of the serum TSH concentration is the most reliable indicator of thyroid status at the tissue level. Studies of mild (subclinical) thyroid hormone excess or deficiency (abnormal TSH/normal range FT 4 and FT 3 ) find abnormalities in markers of thyroid hormone action in a variety of tissues (heart, brain, bone, liver, and kidney). These abnormalities typically reverse when treatment to normalize serum TSH is initiated.
It is important to recognize the clinical situations where serum TSH or FT 4 levels may be diagnostically misleading. These include abnormalities in hypothalamic or pituitary function, including TSH-producing pituitary tumors. Also, serum TSH values are diagnostically misleading during transition periods of unstable thyroid status, such as occurs in the early phase of treating hyper- or hypothyroidism or changing the dose of L-T 4 . Specifically, it takes 6–12 weeks for pituitary TSH secretion to reequilibrate to the new thyroid hormone status. These periods of unstable thyroid status may also occur following an episode of thyroiditis, including postpartum thyroiditis when discordant TSH and FT 4 values may also be encountered. Drugs that influence pituitary TSH secretion (i.e., dopamine and glucocorticoids) or thyroid hormone binding to plasma proteins may also cause discordant TSH values.
Effects of chronological age on thyroid test reference ranges
Despite studies showing minor differences between older and younger subjects, adult age-adjusted reference ranges for thyroid hormones and TSH are not deemed necessary. In children, the hypothalamic–pituitary–thyroid axis undergoes progressive maturation and modulation. Specifically, there is a continuous decrease in the TSH/FT 4 ratio from the time of midgestation until after the completion of puberty. As a result, higher TSH concentrations are typically seen in children. This maturation process dictates the use of age-specific reference limits.
Lower serum total and FT 3 concentrations (measured by most methods) are seen with pregnancy, during the neonatal period, in the elderly, and during caloric deprivation. Furthermore, higher total and FT 3 concentrations are typically seen in euthyroid children. This suggests that the upper T 3 limit for young patients (younger than 20 years of age) should be established as a gradient: between 6.7 pmol/L (0.44 ng/dL) for adults; up to 8.3 pmol/L (0.54 ng/dL) for children under 3 years of age.
Pregnancy
Pregnancy is a physiological state that affects the relationship among thyroid hormones in a complex, but predictable, fashion. During pregnancy, estrogen production increases progressively elevating the mean TBG concentration. TBG levels plateau at 2–3 times the prepregnancy level by 20 weeks of gestation. This rise in TBG results in a shift in the total T 4 (TT 4 ) and total T 3 (TT 3 ) reference range to approximately 1.5 times the nonpregnant level by 16 weeks of gestation. These changes are associated with a fall in serum TSH during the first trimester, such that subnormal serum TSH may be seen in approximately 20% of normal pregnancies. This decrease in TSH is attributed to the thyroid-stimulating activity of human chorionic gonadotropin (hCG) that has structural homology with pituitary TSH. The peak rise in hCG and the nadir in serum TSH occur together at about 10–12 weeks of gestation. In approximately 10% of such cases (i.e., 2% of all pregnancies), the increase in FT 4 reaches supranormal values and, when prolonged, may lead to a syndrome entitled “gestational transient thyrotoxicosis” that is characterized by more or less pronounced symptoms and signs of thyrotoxicosis. This condition is frequently associated with hyperemesis in the first trimester of pregnancy.
The fall in TSH during the first trimester of pregnancy is associated with a modest increase in FT 4 . Thereafter in the second and third trimesters, there is now consensus that serum FT 4 and FT 3 concentrations decrease to approximately 20%–40% below the normal mean, a decrease in free hormone that is further amplified when the iodide nutrition status of the mother is restricted or deficient. In some patients, FT 4 may fall below the lower reference limit for nonpregnant patients. Serum Tg concentrations typically rise during normal pregnancy. Patients with differentiated thyroid carcinomas (DTCs) with thyroid tissue still present typically show a twofold rise in serum Tg with a return to baseline by 6–8 weeks postpartum. Mounting evidence suggests that hypothyroidism during early pregnancy has a detrimental effect on fetal outcome (fetal wastage and lower infant IQ). Thus prepregnancy or first-trimester screening for thyroid dysfunction using serum TSH and thyroperoxidase antibody (TPOAb) measurements is important both for detecting mild thyroid insufficiency (TSH >4.0 mIU/L) and for assessing risk for postpartum thyroiditis (elevated TPOAb). Initiation of L-T 4 therapy should be considered if the serum TSH level is >4.0 mIU/L in the first trimester of pregnancy and if elevated serum TPOAb concentrations are identified (as per the recent AACE guidelines for hypothyroidism). A high serum TPOAb concentration during the first trimester is a risk factor for postpartum thyroiditis. Serum TSH should be used to assess thyroid status during each trimester when pregnant patients are taking L-T 4 therapy, with more frequent measurement if L-T 4 dosage is changed.
Pathological variables
Medications
Medications can cause both in vivo and in vitro effects on thyroid tests. Serum TSH concentration is affected less by medications than thyroid hormone concentrations. For example, estrogen-induced TBG elevations raise serum TT 4 levels but do not affect the serum TSH concentration because pituitary TSH secretion is controlled by the FT 4 independent of binding protein effects. Glucocorticoids in large doses can lower the serum T 3 level and inhibit TSH secretion. Dopamine also inhibits TSH secretion and may even mask the raised TSH level of primary hypothyroidism in sick, hospitalized patients. Propranolol is sometimes used to treat manifestations of thyrotoxicosis and has an inhibitory effect on T 4 to T 3 conversion. Iodide, contained in solutions used to sterilize the skin and radiopaque dyes and contrast media used in coronary angiography and computed tomography scans, can cause both hyper- and hypothyroidism in susceptible individuals. Amiodarone used to treat heart patients has complex effects on thyroid gland function that can induce either hypothyroidism or hyperthyroidism in susceptible patients with positive TPOAb. Lithium can cause hypo- or hyperthyroidism in as many as 10% of lithium-treated patients, especially those with a positive TPOAb titer. Some therapeutic and diagnostic agents (i.e., phenytoin, carbamazepine, or furosemide/frusemide) may competitively inhibit thyroid hormone binding to serum proteins in the specimen and acutely increase FT 4 , resulting in a reduction in serum TT 4 values through a feedback mechanism. Intravenous heparin administration, through in vitro stimulation of lipoprotein lipase, can liberate FFAs, which inhibit T 4 binding to serum proteins and falsely elevate FT 4 .
Nonthyroidal illness
Patients who are seriously ill often have abnormalities in their thyroid tests but usually do not have thyroid dysfunction. These abnormalities are seen with both acute and chronic critical illnesses and thought to arise from a maladjusted central inhibition of hypothalamic-releasing hormones, including TRH. The terms nonthyroidal illness (NTI) as well as “euthyroid sick” and “low-T 4 syndrome” are often used to describe this subset of patients. As shown in Fig. 3.2 , the spectrum of changes in thyroid tests relates both to the severity and stage of illness, as well as to technical factors that affect the methods and in some cases the medications given to these patients.
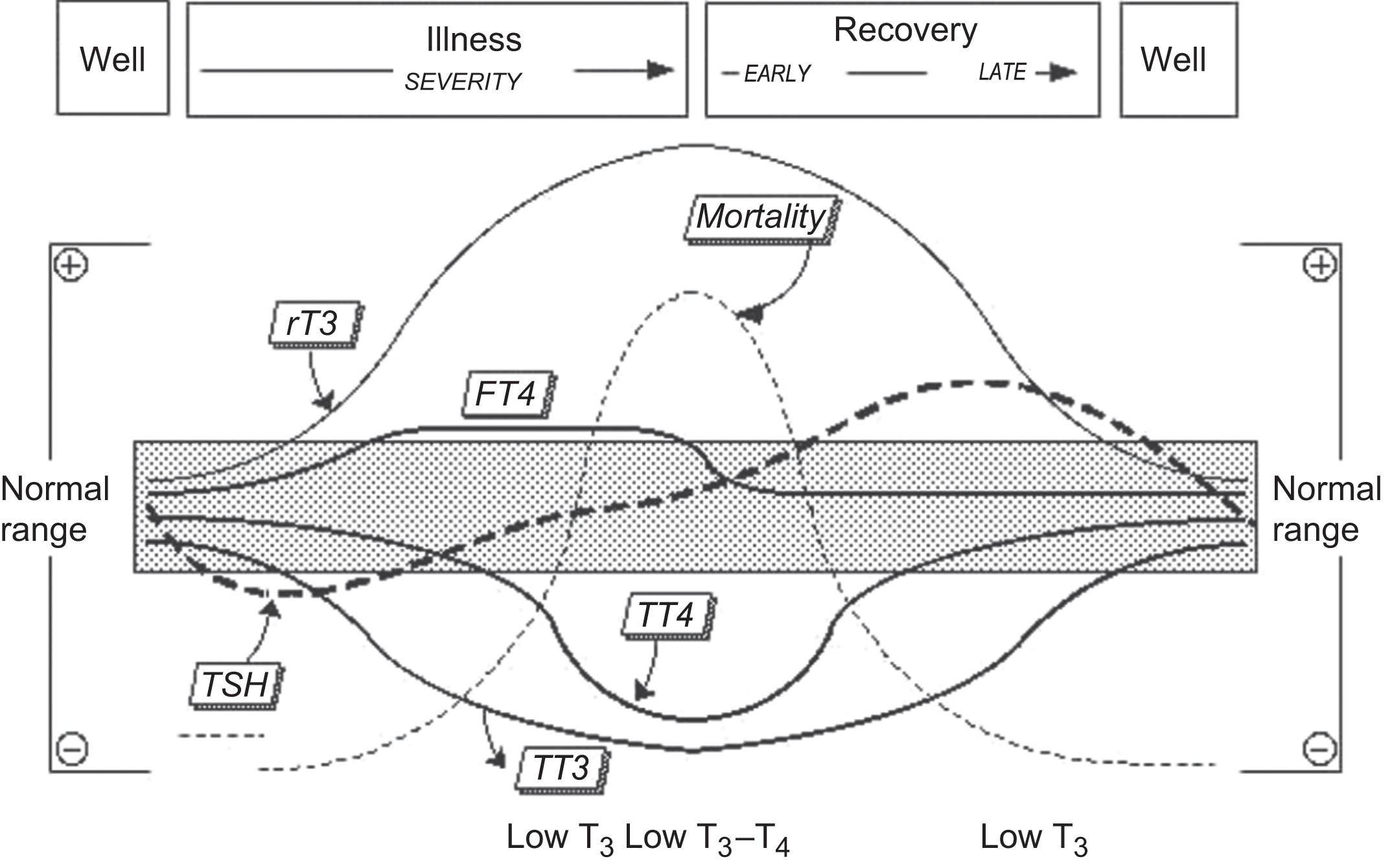
Most hospitalized patients have low serum TT 3 and FT 3 concentrations, as measured by most methods. As the severity of the illness increases, serum TT 4 typically falls because of a disruption of binding protein affinities, possibly caused by T 4 -binding inhibitors in the circulation. If a low TT 4 is not associated with an elevated serum TSH (>20 mIU/L), and the patient is not profoundly sick, a diagnosis of central hypothyroidism secondary to pituitary or hypothalamic deficiency should be considered.
TSH in the absence of dopamine or glucocorticoid administration is the more reliable test for NTI patients. Estimates of free or total T 4 in NTI should be interpreted with caution in conjunction with a serum TSH measurement. Both T 4 + TSH measurements are the most reliable way for distinguishing true primary thyroid dysfunction (concordant T 4 /TSH abnormalities) from transient abnormalities resulting from NTI per se (discordant T 4 /TSH abnormalities). An abnormal FT 4 result in a hospitalized patient should be confirmed by a reflex TT 4 measurement. If both TT 4 and FT 4 are abnormal (in the same direction), a thyroid condition may be present. When TT 4 and FT 4 are discordant, the FT 4 abnormality is unlikely due to thyroid dysfunction and more likely a result of the illness, medications, or an artifact of the test.
A raised total or FT 3 is a useful indicator of hyperthyroidism in a hospitalized patient, but a normal or low T 3 does not rule it out. rT 3 testing is rarely helpful in the hospital setting because paradoxically normal or low values can result from impaired renal function and low binding protein concentrations. Furthermore, the test is not readily available in most laboratories.
It is clear that the diagnosis and treatment of thyroid dysfunction in the presence of a severe NTI are not simple and are best done with the help of an endocrinologist. Although the diagnostic specificity of TSH is reduced in the presence of somatic illnesses, a detectable serum TSH value in the 0.02–20 mIU/L range, when measured by an assay with a functional sensitivity ≤0.02 mIU/L, usually rules out significant thyroid dysfunction, provided that hypothalamic–pituitary function is intact and the patient is not receiving medications that affect pituitary TSH secretion. However, in general, it is best to avoid routine thyroid testing in hospitalized patients if at all possible.
Specimen variables
A few studies have examined the effects of storing blood samples on serum concentrations of total and free thyroid hormones, TSH and Tg. In general, these studies suggest that thyroid hormones are relatively stable whether stored at room temperature, refrigerated, or frozen. Hemolysis, lipemia, and hyperbilirubinemia do not produce significant interference in immunoassays, in general. However, FFAs can displace T 4 from serum-binding proteins, which may partly explain the low TT 4 values often seen in NTI. Heterophilic antibodies may be encountered in patient sera. Heterophilic antibodies fall into two classes. They can be human anti-mouse antibodies (HAMAs) or other specific human anti-animal immunoglobulins (HAAAs) such as human anti-rabbit antibodies. Either HAMAs or HAAAs affects immunometric assays (IMAs) more than competitive immunoassays by forming a bridge between the capture and signal antibodies, thereby creating a false signal and resulting in an inappropriately high value. Approaches to eliminate interference from HAMAs and HAAAs include the use of chimeric antibody combinations and blocking agents to neutralize the effects of these heterophilic antibodies on their methods.
Disease-specific variation
Caloric deprivation
Acute caloric deprivation lowers total and FT 3 within the first 24 h reaching a nadir in 1–2 weeks. rT 3 increases during this period. Total T 4 is unchanged. FT 4 levels are normal or slightly increased. TSH, after a slight downward shift, returns to baseline within 4 days. These effects are reversed by as little as 50 g of glucose or protein, but not by fat.
Hepatic disorders
Patients with hepatic cirrhosis have low serum T 3 levels due to decreased production. T 4 levels are either normal or low (due to decreased albumin and TTR). Serum FT 4 and FT 3 are generally within the normal range. TSH levels are within normal limits but may occasionally be minimally elevated.
Increased T 4 and T 3 levels are seen in acute infectious hepatitis, chronic active hepatitis, and biliary cirrhosis. This increase is due to increased production and release of TBG.
Renal failure
Patients with chronic renal failure have low T 3 , but normal rather than elevated rT 3 . T 4 levels are low, but FT 4 and TSH levels are normal. The thyroid hormone blood levels in nephrotic syndrome are similar, although patients lose T 4 , T 3 , and TBG through the kidney.
Acquired immunodeficiency syndrome and acquired immunodeficiency syndrome—related complex
The circulating thyroid hormone blood concentrations in patients infected with human immunodeficiency virus (HIV) are somewhat different from patients with other NTIs. An unexplained rise in serum TBG produces high circulating T 4 levels. FT 4 is normal. T 3 is also normal rather than decreased. rT 3 is normal or low. The serum TBG levels continue to increase as the patient moves from an asymptomatic stage to acquired immunodeficiency syndrome (AIDS)-related complex and finally to AIDS. The serum TSH concentrations are normal or may be slightly high. Ultimately, circulating T 4 and T 3 concentrations decline signifying a preterminal stage. Thyroid function in patients with HIV who are treated with highly active antiretroviral therapy (HAART) appears to be variable. Elevated or suppressed TSH with normal T 4 as well as isolated low FT 4 concentrations have all been reported. Development of Graves’ thyrotoxicosis during immune cell reconstitution after HAART initiation, with increased T 4 and T 3 , low TSH, and detectable anti-TSH receptor antibodies, has also been reported.
Psychiatric disorders
A minority of patients with acute psychiatric illnesses may have elevated T 4 , FT 4 , and free thyroid index (FTI) at the time of admission. Serum T 3 levels are generally normal or minimally elevated. rT 3 is increased. Serum TSH levels may be elevated. TSH response to TRH may be blunted. Rapid reversal of these parameters, however, takes place.
Helpful clues to distinguish thyroid disease from nonthyroidal causes of abnormal hormone levels
The following clues are helpful when confronted with thyroid parameters from a patient with NTI.
- •
In hyperthyroidism, TSH is below 0.04 mU/L, and FT 4 may be high-nor-mal or high. This degree of TSH suppression is extremely unlikely in NTI alone.
- •
In moderate to severe primary hypothyroidism, TSH will be unquestionably high and FT 4 low. Such a combination of TSH and FT 4 levels is uncommon in NTI.
- •
Confusion arises when FT 4 is low and TSH is either low-normal or TSH is minimally high. These combinations of parameters are compatible with NTI, mild primary hypothyroidism, or pituitary disease. FTI, calculated from T 4 and T 3 resin uptake ratio (RUR), may provide a clue. Although T 4 is low in NTI, primary hypothyroidism, and pituitary disease, RUR is almost always elevated or at the upper limit of normal in NTI and is almost always low-normal or lower than normal in primary hypothyroidism and hypopituitarism. rT 3 is rarely needed and is available in few laboratories. rT 3 is high in NTI except in some liver disorders, renal failure, or HIV-related disorders and low in hypothyroidism. However, equilibrium, dialysis followed by mass spec assessment of free hormone is the best indicator.
- •
When faced with an ill patient with confusing thyroid hormone values that suggest the diagnosis of hypopituitarism, measurement of serum cortisol values may be of help. In severe illness, plasma cortisol levels are generally high. This assessment, however, cannot be accomplished if the patient is receiving exogenous glucocorticoids.
- •
Finally, assessment of antithyroid antibodies may provide a clue to the presence of autoimmune thyroid disease.
- •
Concomitant thyroid disease and NTI can confuse the picture. Occasionally, NTI will normalize thyroid indices and mask thyroid disease. The most common example is the normalization of minimally elevated TSH of mild primary hypothyroidism. Mildly elevated T 4 and T 3 concentrations of hyperthyroidism may also become normal with superimposed NTI.
Thyroid disorders
Hyperthyroidism
Hyperthyroidism is a state in which the body is exposed to an excessive amount of thyroid hormone. Common causes include Graves’ disease, toxic nodule, toxic multinodular goiter, and thyroiditis. Clinical hyperthyroidism can also be produced by ingestion of excessive amounts of thyroid hormones, T 4 or T 3 . Except for thyroiditis, the change from euthyroidism to a hyperthyroid state and progression of hyperthyroidism is usually a slow and gradual process. In fully developed hyperthyroidism, TSH levels are below 0.04 mU/L, and FT 4 concentrations are elevated. However, both these parameters do not become abnormal simultaneously. Usually, TSH concentrations fall to less than 0.04 mU/L before FT 4 levels exceed the normal range. The patient at this stage may be asymptomatic, and a state of “subclinical hyperthyroidism” is said to exist. Additionally, in Plummer’s disease and early Graves’ disease, the T 3 levels rise before the increase in T 4 . When the TSH is suppressed to less than 0.04 mU/L with elevated T 3 concentrations and normal FT 4 concentrations, the patient is said to have “T 3 toxicosis.” It is important to appreciate that TSH concentrations may decline below the population normal range before T 4 or FT 4 concentrations become elevated ( Table 3.1 ).