Steroidogenesis inhibitors
Ketoconazole
Metyrapone
Mitotane
Etomidate
Centrally acting agents
Cabergoline
Pasireotide
Glucocorticoid receptor antagonist
Mifepristone
Note: With the exception of pasireotide and mifepristone, these agents are used “off-label” in Cushing’s syndrome
Steroidogenesis Inhibitors
These agents inhibit one or several enzymatic steps leading to cortisol biosynthesis in the adrenal glands (Table 1). They can be used to control hypercortisolism regardless of the underlying etiology [3, 6]. Measuring 24 h urine free cortisol (UFC ) is helpful in dose titration . Two different therapeutic strategies can be employed: either achieving UFC normalization by titrating the dose of medical therapy or completely suppressing endogenous cortisol synthesis with backup glucocorticoid replacement (“block and replace” regimen). The latter regimen can be particularly helpful in patients with cyclic (intermittent or periodic) hypercortisolism but requires meticulous follow-up in order to avoid glucocorticoid excess resulting from residual (incompletely suppressed) endogenous cortisol synthesis.
Table 1
Currently available steroidogenesis inhibitors
Name | Dose range | Remarks |
|---|---|---|
Ketoconazole | 200–600 mg po bid–tid | Rapid onset of action |
Requires regular monitoring of liver chemistries | ||
Metyrapone | 250–1000 mg po qid | Rapid onset of action |
Preferred in pregnancy | ||
Mitotane | 0.5–3.0 g po tid | Very gradual onset of action |
Adrenolytic in higher doses | ||
Preferred in adrenocortical carcinoma | ||
Etomidate | 0.03 mg/kg iv as a bolus, followed by infusion (0.1–0.3 mg/kg/h) | Useful in patients with severe hypercortisolism |
Use limited by intravenous route and potential for sedation |
All steroidogenesis inhibitors may lead to hypoadrenalism as a result of excess enzymatic blockade of cortisol biosynthesis . Therefore, patients on these agents need to be monitored for clinical and biochemical evidence of hypoadrenalism. Pituitary corticotroph tumors, which maintain some degree of feedback regulation by glucocorticoids, may increase their corticotropin (ACTH) output in response to treatment with steroidogenesis inhibitors, potentially overriding enzymatic blockade in some cases [3, 6].
Ketoconazole
Ketoconazole is an imidazole derivative that inhibits several steps in adrenal steroidogenesis , including 11,20-lyase (desmolase), 17-alpha hydroxylase, and 11-beta hydroxylase [8]. Limited data suggested that ketoconazole might also have direct inhibitory effects on ACTH secretion from pituitary corticotrophs, but this is not widely accepted [8]. Ketoconazole was originally licensed as an antifungal agent and has been prescribed “off-label” to control hypercortisolism. Used as monotherapy in patients with Cushing’s disease, ketoconazole has been reported to control hypercortisolism in 70 % of treated patients based on pooled analyses of 8 small, retrospective studies that included a total of 82 patients [8]. However, a more recent multicenter study found that ketoconazol e use led to UFC normalization in approximately 50 % of patients with Cushing’s disease [9]. Ketoconazole has also been effective in controlling hypercortisolism in approximately 50 % of patients with the ectopic ACTH syndrome [8]. As this medication has a rapid onset of action, it can be particularly helpful among patients with severe manifestations of cortisol excess.
Common, but generally mild, adverse effects associated with ketoconazole use may include gastrointestinal symptoms (nausea, dyspepsia), rash, and headache [3]. Hypogonadism may also develop in men as a result of inhibition of testosterone synthesis. Severe adverse effects are uncommon, including idiosyncratic hepatotoxicity (occurring in approximately 1 in 15,000 treated patients) [10]. Regular monitoring of liver chemistries is recommended in treated patients, who need to be warned of possible symptoms associated with liver toxicity. Asymptomatic transaminitis is more common (occurring in approximately 12 % of patients) and generally improves or resolves with a decrease in medication dose [8].
Ketoconazole absorptio n is significantly higher in the presence of an acidic environment in the stomach. Accordingly, use of medications that raise gastric pH, including proton pump inhibitors or H2 receptor antagonists, is best avoided in patients receiving ketoconazole therapy. Of note, ketoconazole is metabolized in the liver by the CYP450 3A4 enzyme, raising the potential for drug–drug interactions with other medications (such as several “statin s”) that are substrates of the same enzyme or those that either inhibit or induce this enzymatic activity [11, 12].
Metyrapone
Metyrapone inhibits 11-beta hydroxylase , which catalyzes the last step in cortisol synthesis [3]. As a corollary, several steroid precursors accumulate in patients receiving this medication, including 11-deoxycortisol, 11-deoxycorticosterone, as well as several androgenic precursors. Since 11-deoxycortisol often cross-reacts with cortisol in immunoassays, serum cortisol levels may be overestimated in patients on this therapy (depending on the assay used).
Metyrapone was reported to control hypercortisolism in up to 75 % of 53 patients with Cushing’s disease treated for up to 16 weeks based on serum cortisol data (using cortisol day curves) [13]. A more recent study found that metyrapone use led to UFC normalization in approximately 50 % of patients with Cushing’s disease [14]. Escape from its salutary effects may occur in a minority of patients with Cushing’s disease. Metyrapone has also been effective in controlling hypercortisolism in substantial proportions (40–75 %) of patients with the ectopic ACTH syndrome, as well as those with benign or malignant adrenal pathologies [14]. Metyrapone has a rapid onset of action, which can be quite helpful when prompt control of severe hypercortisolism is needed. Metyrapone is considered the preferred medical agent to control hypercortisolism during pregnancy , but is not licensed for use specifically for this indication [15].
Common adverse effects (25 %) associated with metyrapone use include nausea, vomiting, and dizziness [14]. In addition, the accumulation of precursors with mineralocorticoid activity (including 11-deoxycortisol and 11-deoxycorticosterone) may lead to hypertension, edema, and hypokalemia. Similarly, androgenic precursors that accumulate as a result of metyrapone therapy may lead to hirsutism and acne in women [3].
Mitotane
Mitotane inhibits several steps in adrenal steroidogenesis , including the cholesterol side-chain cleavage enzyme, 3-beta hydroxysteroid dehydrogenase, and 11-beta hydroxylase. In addition, it is adrenolytic when used long term in higher doses (>4 g/daily) [3]. This latter effect has led to its use in adrenocortical carcinoma, either as adjuvant postoperative therapy or as treatment in patients with advanced disease [16–18]. In patients with adrenocortical carcinoma, monitoring of systemic levels is advisable with a goal to maintain plasma mitotane levels ≥14 mg/L, which correlate with higher likelihood of achieving tumor control [19]. Used as monotherap y, mitotane is effective in controlling hypercortisolemia in 72–83 % of patients with Cushing’s disease, but has been used in only a few centers worldwide for this indication [16, 20]. Of note, its onset of action is slow, requiring several weeks to months to reach maximal effect in individual patients. As a consequence, mitotane monotherapy is not appropriate when rapid control of severe hypercortisolism is needed. Adrenal insufficiency may occur over time, necessitating the administration of glucocorticoid replacement in treated patients. Escape from its effects on cortisol synthesis is unlikely with long-term use.
Mitotane use may lead to several adverse effects , including gastrointestinal (nausea, vomiting, dyspepsia, diarrhea) and neurologic (dizziness, ataxia, dysarthria, confusion) symptoms, which may limit its use [3]. Other side effects include rash, gynecomastia, abnormal liver chemistries, and dyslipidemia. Rare adverse events include hemorrhagic cystitis, ophthalmic, and hematologic abnormalities. Mitotane is highly lipophilic and can persist in the adipose tissue for months or years after it is stopped. In view of its long half-life and teratogenicity, pregnancy should be avoided for up to 5 years after mitotane discontinuation [3].
Mitotane increases systemic corticosteroid-binding globulin (CBG) levels and accelerates cortisol clearance. Consequently, glucocorticoid replacement doses need to be higher in patients treated with mitotane therapy.
Etomidate
Etomidate is primarily used in anesthesia induction. However, it also inhibits 11-beta hydroxylase, leading to rapid suppression of cortisol synthesis within hours, even in subhypnotic doses [21]. Etomidate can be particularly helpful when rapid control of severe hypercortisolism is needed, especially in patients unable to take oral medications, but requires careful monitoring to avoid excessive sedation [22]. Etomidate is the only intravenous preparation that can be used to control hypercortisolism [22]. However, its use is limited to hospitalized patients with severe hypercortisolism.
Novel Agents Under Investigation
Osilodrostat (LCI699) is a novel 11-beta hydroxylase and aldosterone synthase inhibitor that is currently under study in patients with Cushing’s disease. In a phase II, proof-of-concept trial, osilodrostat administration led to UFC normalization in 92 % of 12 patients with Cushing’s disease who were treated for 70 days [23]. Whether escape from its effects may occur remains to be established. Of note, approximately 79 % of patients treated with LCI699 achieved normal UFC in a 6 month extension of the phase II study that included 19 patients [12, 24]. The efficacy and safety of osilodrostat are being investigated in a phase III study.
Osilodrostat appears to be well tolerated in most patients. However, fatigue, headache, gastrointestinal symptoms , and dizziness may occur. Hypertension, edema, and hypokalemia may develop as a consequence of accumulation of precursors with mineralocorticoid activity, and hirsutism or acne may occur as a result of accumulation of androgenic precursors.
Levoketoconazole is a ketoconazole enantiomer that is also under investigation in Cushing’s disease. Based on preliminary data, it may have increased potency and duration of action and potentially a lower risk of hepatotoxicity [12].
Abiraterone is an inhibitor of 17-alpha hydroxylase and 17,20-lyase activity and has been used to suppress androgen synthesis in patients with castration-resistant advanced prostate cancer [25]. Based on its mechanism of action, it would be predicted to be potentially efficacious in patients with Cushing’s syndrome. However, clinical studies are required to examine this possibility.
Subgroups of adrenal masses in patients with bilateral macronodular adrenal hyperplasia or adrenal adenomas may express a wide variety of receptors, including those engaging glucose-dependent insulinotropic peptide (GIP) , luteinizing hormone (LH)/human chorionic gonadotropin (hCG), vasopressin (V1, V2, V3), serotonin (5HT4 and 5HT7), angiotensin (AT1), glucagon, or beta adrenergic receptors [26]. Based on these considerations, medications that inhibit some of these receptors or pathways, including octreotide or pasireotide (inhibiting GIP secretion), leuprolide (inhibiting LH secretion), and propranolol (inhibiting beta adrenergic receptors), have shown at least transient effectiveness in controlling hypercortisolism in small numbers of patients with adrenal masses expressing the respective receptors [26–29].
Centrally Acting Agents
These agents are directed at suppressing ACTH synthesis and/or release and may be efficacious in controlling hypercortisolism in patients with ACTH-dependent Cushing’s syndrome, primarily those with Cushing’s disease (Table 2) [3, 4, 30]. In addition, they might lead to a decrease in pituitary tumor size in patients with Cushing’s disease or Nelson’s syndrome. Currently, these medications are used primarily for their antisecretory effects, since data on tumor control are limited.
Table 2
Currently available centrally acting agents
Name | Dose range | Remarks |
|---|---|---|
Cabergoline | 0.5–7.0 mg po weekly | Escape (loss of effectiveness) may occur over time |
Potential risk of valvulopathy in high doses | ||
Pasireotide | 0.3–0.9 mg sc bid | Hyperglycemia or diabetes mellitus may develop |
Glucose, hepatic function, and electrocardiographic monitoring advised |
Cabergoline
Cabergoline is a dopamine receptor (type 2 specific) agonist , which is licensed as therapy for hyperprolactinemia, but has also been used “off-label” to treat patients with Cushing’s disease [3, 31]. Its potential effectiveness is predicated by the presence of dopamine receptors in the majority of corticotropinomas [32]. Cabergoline administration may control hypercortisolism in 30–40 % of patients with ACTH-secreting pituitary adenomas [33, 34]. However, escape from its effects may occur over time. It should also be noted that cabergoline doses that are required to control hypercortisolism are generally larger (1–7 mg/week) than those that are effective in the majority of patients with hyperprolactinemia (0.5–2.0 mg/week). In contrast to cabergoline, bromocriptine, an older dopamine receptor agonist, is largely ineffective in patients with Cushing’s disease.
Cabergoline administration is generally tolerated well. However, nausea, vomiting, and dizziness may occur and are more common among patients receiving high doses. Other less common adverse effects include headache, nasal congestion, constipation, digital vasospasm, anxiety, depression, exacerbation of psychosis, or a variety of manifestations of impulsivity [31, 35]. When administered in high doses in patients with Parkinson’s disease, cabergoline use was associated with cardiac valvulopathy, which is presumed to occur as a consequence of serotonin receptor (5HT2B) activation [36, 37]. While cabergoline use in doses typically required to treat hyperprolactinemia (0.5–2.0 mg/week) appears to be safe with regard to cardiac valvulopathy, it is less clear whether its long-term use in higher doses (up to 7.0 mg/week) needed to control hypercortisolism may increase the risk of valvular damage [38]. Periodic echocardiography seems prudent in patients receiving such higher cabergoline doses. However, there are currently no data examining the cost-effectiveness of echocardiography in detecting valvulopathy in this population.
Pasireotide
Pasireotide is a somatostatin receptor agonist with expanded specificity, which activates type 1, 2, 3, and 5 somatostatin receptor isoforms [11, 30]. It is thought that stimulation of the type 5 receptor isoform accounts for its efficacy in patients with Cushing’s disease [39]. In contrast, octreotide, which activates type 2 and (weakly) type 5 somatostatin receptors, has very limited efficacy in patients with Cushing’s disease. Of note, type 5 and type 2 somatostatin receptor isoforms are expressed by approximately 84 % and 74 % of corticotropinomas, respectively [32].
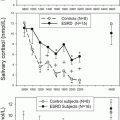
The efficacy of pasireotide administration was established in a phase 3, multicenter clinical trial of 162 adults with Cushing’s disease, who were randomly allocated to either of two pasireotide starting doses (600 mcg twice daily and 900 mcg twice daily) and were treated for 12 months. Control of hypercortisolism , based on UFC normalizatio n, was reported in 15 % and 26 % of patients who received the lower and higher pasireotide starting dose without need for dose uptitration, respectively [40]. In addition, pasireotide therapy led to weight loss, decrease in blood pressure, and improved quality of life as well as a decrease in tumor size among patients with measurable tumor mass (by 9.1 % and 43.8 % in patients receiving the lower and higher pasireotide starting dose, respectively). Pasireotide has been approved by the FDA and EMA for use in patients with Cushing’s disease who have failed pituitary surgery or are not surgical candidates. Pasireotide LAR, a long-acting form of pasireotide, is under evaluation in a phase III clinical trial as a possible therapy in patients with Cushing’s disease [41].
Similar to octreotide, pasireotide administration is associated with possible gastrointestinal adverse events (nausea, abdominal pain, diarrhea, gallstones or sludge, mild transaminitis). Asymptomatic sinus bradycardia, QT prolongation, and hair loss may also occur. In addition, pasireotide therapy appears to be associated with the development of hyperglycemia or diabetes mellitus. Indeed, hyperglycemia developed in 73 % of patients in the phase 3 trial [40]. The hyperglycemic effects of pasireotide occur as a consequence of inhibition of insulin secretion, which is partly attributable to suppression of incretin secretion from the gastrointestinal tract [42]. Self-monitoring of blood glucose is advisable in patients treated with pasireotide. Hyperglycemia may be treated with metformin therapy with possible stepwise addition of incretin mimetics, dipeptidyl peptidase inhibitors, and/or insulin. In addition to monitoring for hyperglycemia, pasireotide-treated patients are advised to undergo periodic evaluation of serum electrolytes, liver function tests, electrocardiograms, and gallbladder ultrasound examinations.
Novel Agents and Targets Under Investigation
The retinoic acid receptor appears to have a role in the regulation of proopiomelanocortin and ACTH synthesis [43, 44]. Accordingly, cognate retinoic acid receptor agonists may be of potential benefit in patients with Cushing’s disease. Preliminary data suggest some evidence of in vitro and in vivo effectiveness of retinoic acid in Cushing’s disease, but its clinical use has not been adequately investigated [45].
The epidermal growth factor receptor is often expressed in corticotropinomas [46]. Recent in vitro and preclinical data suggest a role for epidermal growth factor receptor inhibition with gefitinib in controlling tumor size and hypercortisolism [46]. While epidermal growth factor receptor inhibition (with gefitinib) is of potential interest as a treatment strategy in patients with Cushing’s disease, its efficacy and safety in this population remain to be explored in clinical studies.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree