Background
Embryology, surgical anatomy and physiology
Embryology
The thyroid gland originates from the first pharyngeal arch and begins as an out-pouching of endodermal tissue from the base of the tongue, called the foramen caecum, as early as the fourth week of gestation. This soft tissue mass descends over the ensuing weeks anterior to airway structures, halting at the level of the second to fourth tracheal cartilages by the seventh week. This route of descent is the embryological origin of the thyroglossal tract, duct or cyst, and the distal portion constitutes the pyramid of the thyroid. The parathyroid cell masses and the ultimobranchial bodies also descend with the rudimentary thyroid over this period of development. Bilateral thyroid buds then develop with associated arterial supply, venous and lymphatic drainage.
Thyroid anatomy
The posterolateral aspect of each lobe features a variably sized protuberance of thyroid tissue known as the tubercle of Zuckerkandl (TZ). This is the site of fusion of the ultimobranchial body (containing the precursors of the parafollicular C cells) and the median thyroid process. Medial to the TZ is a thickened area of pretracheal fascia, known as the ligament of Berry, tethering the thyroid to the trachea.
Circulation to the thyroid gland involves two arterial and two to three venous draining vessels bilaterally. The superior thyroid artery is a branch of the external carotid artery and enters the superior pole of each lobe. The inferior thyroid artery branches off from the thyrocervical trunk and is more variable in its course; its branches typically enter the lateral aspect of the gland in an oblique fashion. Superior, middle and inferior thyroid veins drain the gland, into the internal jugular and the brachiocephalic veins. The presence of the middle thyroid vein is variable.
Lymphatic drainage roughly follows the order of perithyroidal nodes, the pre- and paratracheal nodes (including the prelaryngeal nodes), then the jugular chain of nodes.
Recurrent laryngeal nerve and external branch of superior laryngeal nerve anatomy
The recurrent laryngeal nerve (RLN), also known as the inferior laryngeal nerve, is a branch of the vagus nerve. After winding around the aortic arch on the left, and the right subclavian artery, the RLNs course superomedially from the root of the neck to continue in the tracheo-oesophageal groove. The right RLN has an oblique course and the left a more vertical course, in the neck. The posteromedial aspect of the gland is therefore in close approximation to the last extralaryngeal segment of the nerve. In the last 1–2 cm of its extralaryngeal course, the RLN is juxtaposed between the lateral side of the ligament of Berry and the medial side of the TZ, plastered in place by an overlying fascia containing the tertiary branches of the inferior thyroid artery. It is here, just before entering the larynx under the cover of the cricopharyngeus muscle, that the RLN is most constant in position and also most prone to injury during thyroid surgery due to the difficulty in freeing it from the structures enveloping it. Up to 72% of RLNs divide into two or more branches before entering the larynx. The anterior-most branch carries most of the motor fibres to the laryngeal muscles, and therefore is the most important branch to preserve.
The external branch of the superior laryngeal nerve (EBSLN), also a branch of the vagus nerve, is the motor supply of the cricothyroid muscle. In its course from the carotid sheath to the cricothyroid muscle, it comes close to the junction of the upper pole and the upper pole vessels. The Cernea Classification describes the various variations, which have relevance in surgical dissection ( Fig. 2.1 ).
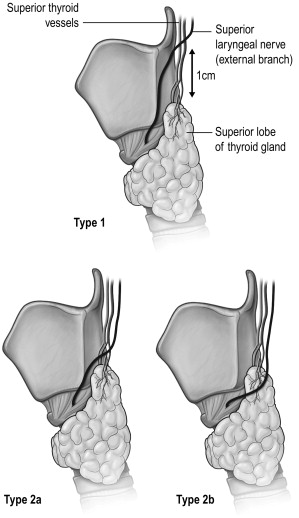
Parathyroid anatomy
Parathyroid cell masses from the third and fourth pharyngeal pouches form the inferior and superior parathyroid glands respectively. They descend from their pouch origins to the final positions in close association with the developing thyroid and thymus glands. Therefore, it is not surprising that they maintain such close relationships with these glands. The majority of non-pathological superior parathyroid glands are found in the vicinity of the cricothyroid junction (77%), often closely related to the TZ and RLN, or under the capsule on the posterior surface of the superior thyroid pole (22%). The superior glands lie posterior to the RLNs. The inferior parathyroid glands, having travelled a longer distance, are more variable in their locations. They can be found on the surface of the inferior thyroid pole (42%), in the uppermost part of the thymic horn (39%), lateral to the inferior thyroid pole (15%) or in other ectopic locations (2%). They lie anterior to the RLNs.
Thyroid physiology
The physiological unit of the thyroid gland is the thyroid follicle. Each follicle is lined by follicular cells and contains colloid. Follicular cells are responsible for absorption and transport of iodide, production of thyroglobulin (Tg), and thyroid hormone removal from Tg binding for systemic secretion. The colloid represents the gland’s reservoir of thyroid hormone. Two forms of thyroid hormone are produced: T 3 (tri-iodothyronine) and T 4 (tetra-iodothyronine or thyroxine). The production of thyroid hormone involves iodination of the tyrosyl residues on the Tg molecules to produce mono- and di-iodotyrosine molecules, which then couple to form T 3 and T 4 molecules. The thyroglobulin-bound T 3 and T 4 complexes are stored in the colloid. Upon endocytosis back into the follicular cells, T 3 and T 4 molecules are released from Tg molecules before being secreted into the systemic circulation.
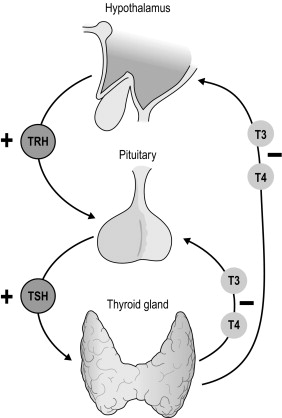
Thyroid hormone secretion in health is dependent upon a classical feedback loop. Reduced levels of T 3 and T 4 induce thyroid-stimulating hormone (TSH) release directly from the anterior pituitary and also indirectly through hypothalamic stimulation of thyrotropin-releasing hormone (TRH) release (which in turn induces TSH secretion). Such pathways are suppressed in periods of thyroid hormone excess. TSH exerts its effect via TSH receptors to increase iodide trapping, Tg synthesis, as well as T 3 /T 4 production and secretion ( Fig. 2.2 ).
Circulating hormone is both free and protein bound, and this is maintained in equilibrium to ensure equal whole-body distribution. Free hormone is physiologically active and exerts its effect upon peripheral tissues through nuclear thyroid hormone receptors. Whilst T 4 is more abundant in the circulation, T 3 is more potent physiologically. The majority of T 3 is derived from de-iodination of T 4 by peripheral tissues. The physiological effect of thyroid hormone is apparent in all metabolically active tissues, the manifestations of which become most apparent in disease states.
Clinical history and examination
Pathologies of the endocrine organs, including the thyroid gland, give rise to a broad spectrum of symptoms and signs. The clinician must be thorough in both history-taking and physical examination, not only of the organ in question, but also other organ systems that the endocrinopathy may affect. A schema for assessing patients with thyroid complaints is suggested in Box 2.1 , while Box 2.2 lists the questions that are essential to keep in mind when formulating a management plan. Some symptoms and signs of thyroid pathologies are listed in Tables 2.1 and 2.2 . These are by no means exhaustive. A grading system for goitre size has been published by the World Health Organisation ( Table 2.3 ).
- 1.
Local symptoms and signs
- 2.
Thyroid status – systemic symptoms and signs
- 3.
Family history
- 4.
Risk factors
- 5.
Medications
- 6.
Known history of thyroid pathology/surgery
- 7.
General health/fitness for surgery
Questions to be kept in mind when gathering information to formulate a treatment plan:
- 1.
Does the patient have a benign or malignant condition?
- 2.
Are the local symptoms in keeping with the pathology?
- 3.
Is the thyroid causing hyper- or hypothyroidism?
- 4.
What are the valid treatment options?
- 5.
Is there an indication for surgery?
- 6.
What surgery is indicated?
- 7.
What perioperative measures might be required?
| Local | Systemic |
|---|---|
Mass
| Hyperthyroidism
|
| General | Hyperthyroidism |
|
|
| Grade 0 | No palpable or visible goitre |
| Grade 1 | A goitre that is palpable but not visible when the neck is in the normal position (i.e. the thyroid is not visibly enlarged) |
| Grade 2 | A swelling in the neck that is clearly visible when the neck is in the normal position and is consistent with an enlarged thyroid when the neck is palpated |
Investigation of the thyroid
Investigations of the thyroid are used to confirm thyroid functional status, define anatomical extent of the pathology, determine likelihood of malignancy, and detect the presence of autoimmune disease.
Blood tests
Thyroid function tests
Thyroid function homeostasis is dependent upon the pituitary–thyroid axis feedback loop ( Fig. 2.2 ). Thyroid function tests (TFTs) identify hypo- or hyperfunction through quantification of not only circulating thyroid hormones (T 3 and T 4 ) but, more importantly, thyroid-stimulating hormone (TSH or thyrotropin). In keeping with the negative feedback loop, thyroid hyperfunction results in a suppressed TSH, in the presence of increased circulating T 3 and T 4 . Conversely, reduced hormone levels lead to an increase in TSH. (also see section on hyperthyroidism).
Thyroid antibodies
Thyroglobulin antibody
Thyroglobulin antibody (TgAb) is a highly sensitive marker of Hashimoto’s disease and over 99% of patients with this condition will have elevated antibodies. Elevated levels may also be seen in Graves’ disease. The presence of TgAb should also be noted when monitoring Tg levels for surveillance after treatment of papillary or follicular thyroid cancer. TgAb may interfere with Tg assays and lead to spurious levels.
Thyroid peroxidase antibody
Thyroid peroxidase antibody (TPOAb) is commonly elevated in Graves’ disease but may also be seen in cases of thyroiditis. The test lacks sensitivity and specificity for Graves’ disease and is therefore only useful with a clear clinical suspicion of disease. Systemic autoimmune diseases may also lead to TPOAb positivity that may not be of any clinical significance.
TSH receptor antibody
TSH receptor antibodies (TRAbs) may be directly stimulatory or exert an inhibitory action on thyroid TSH receptors, leading to related changes in thyroid hormone secretion. Stimulatory antibodies are encountered in Graves’ disease and are particularly useful where the clinical diagnosis proves difficult to make. TRAbs are also identified in euthyroid Graves’ opthalmopathy, unilateral Graves’ eye disease, subclinical hyperthyroidism and thyroiditis. TRAbs can also cross the placenta and, in pregnancy, a positive test predicts for neonatal thyroid dysfunction.
Biomarkers of malignant disease
Thyroglobulin
Assessment of serum Tg is employed in surveillance of patients who have undergone total thyroidectomy and radioactive iodine ablation for differentiated thyroid cancers. Thyroglobulin serves as a biochemical marker of disease recurrence or progression in those with residual disease.
Calcitonin
Calcitonin is produced by the parafollicular C cells of the thyroid gland. These cells represent the cellular origin of medullary thyroid carcinoma (MTC), and calcitonin is elevated in cases of this disease. Calcitonin serves as a sensitive marker of disease recurrence and progression in MTC, and progressively higher levels at diagnosis are associated with larger tumours, regional lymph node metastases and distant metastases. Routine measurement of calcitonin in the work-up of thyroid nodules is recommended by the European Thyroid Cancer Taskforce; however, the American Thyroid Association (ATA) has not made a recommendation for or against this practice in their updated guidelines.
Carcinoembryonic antigen
Whilst not as specific as calcitonin, carcinoembryonic antigen (CEA) is also employed as a biomarker of disease in MTC. When elevated in the absence of an obvious primary tumour elsewhere, the thyroid gland should be investigated.
Imaging studies
Ultrasonography
Ultrasound (US) is the imaging modality of choice for evaluation of the thyroid gland and associated lymph nodes. It is accessible, inexpensive, non-invasive and well tolerated. Surgeon-performed thyroid US is increasingly becoming a standard skill set and it has been shown that surgeon-performed US leads to beneficial changes in diagnosis and management. It also affords the clinician the added advantage of an intimate anatomical knowledge of the region to be dissected, which is of particular benefit in re-operative surgery or where selective lymph node dissection is anticipated.
US features that are suspicious for malignancy include microcalcifications, intranodular hypervascularity, hypoechoeic nodules, irregular margins and extracapsular extension (see Box 2.3 ).
High suspicion
- •
Family history of thyroid malignancy or multiple endocrine neoplasia
- •
Rapid tumour growth
- •
Very firm nodules
- •
Fixation to adjacent structures
- •
Vocal cord paralysis
- •
Associated cervical lymphadenopathy
- •
Distant metastasis (lungs or bones)
Moderate suspicion
- •
Age < 20 years or > 60 years
- •
Male sex
- •
Solitary nodule
- •
History of head and neck irradiation
- •
Firm texture or possible fixation of nodule
- •
Nodule > 4 cm in diameter and partially cystic
- •
Compressive symptoms: dysphagia, dysphonia, hoarseness, dyspnoea, cough
Sonographic features of suspicion
- •
Hypoechogenicity
- •
Microcalcifications
- •
Absent halo
- •
Irregular margin
- •
Invasive growth
- •
Regional lymphadenopathy
- •
High intranodular flow by Doppler
Nodules and lymph nodes may also be aspirated under US guidance, ensuring precise sampling and avoidance of complications. US-guided fine-needle aspiration (FNA) has been shown to produce lower rates of non-diagnostic and false-negative cytology specimens, when compared to FNA performed by palpation only.
Nuclear medicine studies
Thyroid isotope scanning employs intravenous radiolabelled iodine ( 131 I or 123 I) or technetium pertechnetate ( 99m Tc), which are taken up by active thyroid cells and detected by gamma-ray cameras. Isotope scanning may be used in determining the cause of hyperthyroidism, identifying ectopic thyroid tissue or postoperative remnant tissue and detecting metastases of differentiated thyroid cancers. It is also used for surveillance after treatment of differentiated thyroid cancers.
Computed tomography
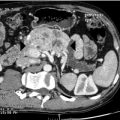
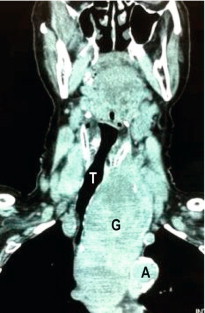
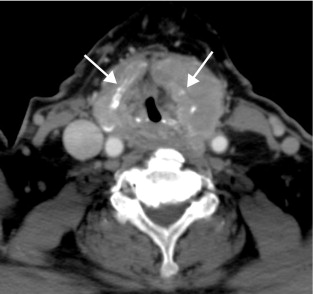
Computed tomography (CT) scanning gives detailed anatomical definition of the thoracic inlet and associated structures, and is therefore of utility in the management of retrosternal disease. The degree of tracheal compression and distortion of adjacent structures by a bulky retrosternal goitre can be adequately defined ( Fig. 2.3 ). The presence of mediastinal, pulmonary or more distant metastases in thyroid cancer can also be quantified. Often, CT scanning will dictate the surgical approach, with a large retrosternal goitre or low mediastinal metastases being indications for sternotomy. In locally aggressive cancers, CT scanning is a useful modality in assessing invasion of the aerodigestive tract and internal jugular veins ( Fig. 2.4 ).
Tissue diagnosis
Fine-needle aspiration cytology (FNAC), along with clinical examination and US scanning, form the basis of the triple test for a thyroid nodule. Any solitary or dominant nodule over 1 cm should be put through the triple test, including FNAC.
The interpretation of FNAC results has recently been standardised with the introduction of the Bethesda classification, which divides FNAC results into six categories ( Table 2.4 , Fig. 2.5 ). Each category correlates with an estimated risk of malignancy, which aids surgical decision-making (see section on management of differentiated thyroid cancers).
| Category | Description | Risk of malignancy (%) | Usual management |
|---|---|---|---|
| I | Non-diagnostic | 1–4 | Repeat FNAC with US |
| II | Benign | 0–3 | Clinical follow-up |
| III | Atypia of undetermined significance or follicular lesion | 5–15 | Repeat FNAC |
| IV | Follicular neoplasm or suspicious for follicular neoplasm | 25–30 | Lobectomy |
| V | Suspicious for malignancy | 60–75 | Total thyroidectomy or lobectomy +/− frozen section |
| VI | Malignant | 97–99 | Total thyroidectomy or lobectomy |

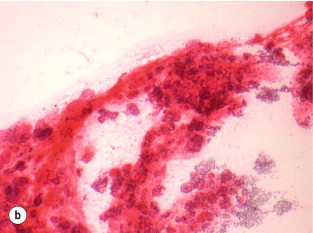
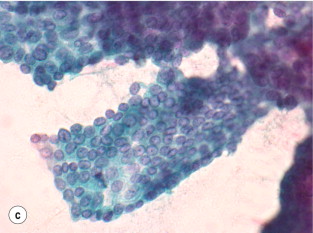
Incidental thyroid pathology
Thyroid pathology is increasingly being found incidentally during investigations for coexisting pathology. US for carotid vascular disease, staging CT for head and neck malignancy and positron emission tomography (PET) scanning frequently identify either incidental nodular disease or tracer uptake consistent with thyroid pathology. The approach to such findings follows the general principles outlined for clinically evident nodules, with dedicated ultrasound scan and biopsy of suspicious nodules, or nodules larger than 1 cm.
Surgical pathologies of the thyroid
Benign conditions
Benign goitre
‘Goitre’, derived from the Latin word ‘ gutter ’ meaning throat, simply refers to an enlarged thyroid gland. A goitre can be diffuse if the thyroid is evenly enlarged or multinodular if the enlarged thyroid comprises multiple nodules of varying sizes. It can also be toxic when some nodules develop autonomous production of thyroid hormones; retrosternal if the inferior border extends below the level of the sternal notch and clavicles; or malignant if the goitre harbours malignancy. Benign multinodular goitre (MNG) is endemic in parts of the world due to iodine deficiency, and in 2003, the worldwide prevalence of goitre was 15.8%.
Causes of multinodular goitre
Iodine deficiency
Goitre is considered endemic when its prevalence in a region is over 10%, and iodine deficiency is the primary cause of endemic MNG. Iodine deficiency is mainly due to a low dietary intake in areas of iodine-poor soil, regardless of altitude. While some of the most severely iodine-deficient regions are high up in the mountains, such as the Pyrenees, the Himalayas and the Cordillera of the Andes, populations in coastal areas, large cities and highly developed countries can also be found to be iodine deficient. The Sydney basin on Australia’s eastern coast is one example.
Genetics
Although no single causative gene with a clear mode of inheritance has been described, the familial clusters and higher concordance rates in monozygotic twins with sporadic MNG point towards a genetic aetiology. Genes implicated in familial goitre include the thyroglobulin gene, the thyroid-stimulating hormone receptor gene, the Na + /I − symporter gene and the MNG marker 1 on chromosome 14. A defect in any of these genes can result in dyshormonogenesis, leading to compensatory goitre formation. Further studies are required for the significance of these genes to be extrapolated to the general population.
Goitrogens
Thiocyanate is the goitrogen found in cassava and vegetables of the brassica family (e.g. cabbage, Brussels sprouts, cauliflower, mustard and turnip). Their goitrogenic effects are usually seen in areas where these food types are the staple, and especially where the iodine intake is also borderline.
Gender
The female-to-male ratio of sporadic MNG ranges between 5:1 and 10:1; however, the reason for this is poorly understood.
Drugs
Some drugs have been implicated in the development of MNG by inducing a state of chronic hypothyroidism with an increased TSH secretion. For instance, long-term lithium treatment results in goitre in up to 50% of treated patients.
Pathogenesis
There are two stages in the development of MNG, which may be separated by a long period of time, sometimes as long as decades. The early stimulus for generalised thyroid hyperplasia is most commonly due to iodine deficiency in endemic areas, whereas in sporadic MNG, genetic predisposition or ingested goitrogens may be the stimulus. The second stage of MNG formation is due to focal somatic mutations. Although most of the mutations result in enlarged colloid follicles, focal hyperplasia, hypertrophy, adenoma or even carcinoma can all contribute to the MNG. Over time, these nodules become intersected by areas of fibrosis.
Management of benign MNG
Surgery is the only effective way of treating compressive symptoms of the aerodigestive tract caused by MNG. As such, this constitutes the main indication for surgery. Other indications include MNG with nodules suspicious of malignancy on FNA, toxic MNG and retrosternal goitre.
Total thyroidectomy (TTx) has replaced subtotal thyroidectomy (STTx) as the procedure of choice for benign MNG. The major issue with STTx is recurrence, with long-term follow-up data showing eventual recurrence in up to 50% of patients. Furthermore, if secondary surgery is subsequently required for symptomatic recurrent goitre, the risk of complications rises. A significantly higher complication rate has been reported in patients undergoing re-operative thyroidectomy for recurrence after initial STTx, compared to those who had a primary TTx.
Thyroid cysts
Thyroid cysts are usually benign and account for up to a third of surgically excised solitary thyroid lesions. However, up to 10% of mixed solid and cystic thyroid lesions can be malignant in nature. FNA under US guidance and targeting the solid component of a mixed solid/cystic lesion ensures optimal cellular harvest. Indications for surgery include malignant or suspicious cytology, large cyst (> 4 cm), rapid refill after aspiration, heavily bloodstained aspirate, and a history of head and neck irradiation.
Malignant conditions
The incidence of thyroid cancer has increased exponentially over the last three or so decades according to data from countries such as Australia, the USA, Canada and France. This steep rise in incidence is due to increased diagnosis of papillary thyroid carcinoma (PTC), especially microcarcinomas, with mortality due to thyroid cancer remaining consistently low (5-year relative survival of 96%). Females are four times more likely to be diagnosed with thyroid cancer than males.
Papillary thyroid carcinoma accounts for 80% of thyroid cancers. This type of thyroid cancer, originating from thyroid follicular cells, is the commonest type. Follicular thyroid cancer (FTC), also originating from follicular cells, accounts for 15% of thyroid cancers. PTC and FTC are collectively known as differentiated thyroid cancers (DTCs). The remaining 5% of thyroid cancer consists of medullary thyroid cancer (MTC), poorly differentiated thyroid cancer (PDTC), anaplastic thyroid cancer (ATC), lymphoma and metastatic cancer to the thyroid from a distant primary site.
Molecular biology of thyroid cancers
The underlying molecular mechanisms that result in thyroid cancer development have gradually been elucidated in the last 20 years. A brief summary is given here.
Papillary thyroid carcinoma
The molecular studies over the last two decades have led to the observation that PTC is characterised by genetic lesions that activate the mitogen-activated protein kinase (MAPK) signalling pathway. These genetic lesions can be produced by chromosomal rearrangements such as RET / PTC , TRK and AKAP9 / BRAF oncogenes, or point mutations such as BRAF and RAS oncogenes.
The RET proto-oncogene encodes a receptor-type tyrosine kinase. In PTC, RET is mutated by chromosomal rearrangements where the tyrosine kinase domain is fused to a variety of donor genes causing constitutive activation of the tyrosine kinase domain. RET / PTC1 and RET / PTC3 are the commonest combinations, and they account for over 90% of all RET rearrangements in PTC. Tumours with RET / PTC rearrangements are typically of the classical variant of PTC.
The BRAF protein is the B-isoform of the intracellular Raf kinase within the MAPK signalling cascade. The BRAF gene is mutated in a variety of human cancers, and by far the commonest mutation involves a valine-to-glutamate substitution at residue 600 ( BRAF V600E ). This substitution results in constitutive activation of Raf kinase and subsequently up-regulation of the MAPK pathway. BRAF V600E is detected in 29–69% of PTC cases, and can be associated with the classical and tall cell variants of PTC, as well as poorly differentiated and anaplastic thyroid carcinomas. Some studies report association of BRAF V600E with more aggressive clinicopathological features; however, this view is not universal.
Follicular thyroid carcinoma
The common genetic mutations associated with FTCs are RAS mutations, PAX8 / PPAR -γ rearrangement and phosphoinositide 3-kinase (PI3K)/protein kinase B (Akt) pathway deregulation.
Oncogenic mutations may involve any of the three members of the RAS gene family. RAS mutations reportedly occur in up to 50% of FTCs, 40% of follicular adenomas, 25% of Hurthle cell carcinomas (HCCs) and 20% of follicular variant PTCs. They are also seen frequently in PDTC and ATC. The presence of RAS mutations in follicular adenomas is a clue to the adenoma–carcinoma sequence in the pathogenesis of FTCs. Significant correlation between RAS mutations to metastases and poor prognosis has been found.
The PAX8 / PPAR -γ rearrangement results in a fusion of the DNA-binding domain of PAX8 to the peroxisome proliferator-activated receptor PPAR-γ. The fusion protein stimulates proliferation of thyrocytes by an unknown mechanism. The PI3K/Akt pathway is central for many cellular events such as growth, proliferation and apoptosis. Its constitutive activation by mutations is a common feature in many cancers, including FTC.
Differentiated thyroid cancers (PTC and FTC)
Risk factors
The most well-established environmental risk factor for thyroid cancer is exposure to ionising radiation. PTC is the type of thyroid cancer that is associated with radiation exposure, which induces damage to cellular DNA, commonly causing RET/PTC chromosomal rearrangements. The effect is most pronounced in children, and the latency period ranges from 5 to 30 years. In a patient with a history of radiation exposure, the overall risk of malignancy in a nodule is 30–40%; therefore, an initial TTx is recommended.
No susceptibility gene for hereditary non-medullary thyroid cancer (HNMTC) has been identified; however, epidemiological studies have shown that they are more aggressive than sporadic disease. The risk of developing thyroid cancer is 5 to 10 times higher in patients with a first-degree relative who has thyroid cancer, when compared to the general population. Features such as early age at presentation, reversed gender distribution, large tumour size, tumour multicentricity and aggressive tumour biology are clues to suspect such a kindred. Until specific gene(s) are identified, a detailed family history is the only way to identify these at-risk families. HNMTC may also be part of another familial syndrome, such as familial adenomatous polyposis ( APC ), Cowden syndrome ( PTEN ), Carney complex type 1 ( PRKAR1 α), McCune–Albright syndrome ( GNAS1 ) and Werner syndrome ( WRN ) (see Chapter 4 for further details).
Pathology
Papillary thyroid carcinoma
Papillary thyroid carcinoma is an epithelial malignant tumour of the thyroid gland, which still retains follicular cell differentiation and is characterised by unique nuclear features. The nuclei typically show a clear or ground-glass appearance, and irregularities of the nuclear contours can often be seen as nuclear grooves and pseudo-inclusions.
Since the diagnostic features of PTC are mainly in the nuclei, it is possible to confidently make the diagnosis on cytology. In addition, fragments of papillae may be seen, along with other features such as ropy colloid, multinucleated giant cells and psammoma bodies. Psammoma bodies are rounded and concentrically laminated calcifications, which are found in association with tumour cells, within lymphatic spaces or within the tumour stroma.
Papillary thyroid carcinomas show positive staining for cytokeratins, thyroglobulin (Tg) and thyroid transcription factor-1 (TTF-1) on immunohistochemistry. They are negative for synaptophysin and chromogranin. Metastatic papillary carcinomas from the thyroid are typically positive for both Tg and TTF-1, while metastases from pulmonary papillary carcinomas may be positive for TTF-1 and are usually negative for Tg. Metastatic papillary carcinomas from other sites are usually negative for both Tg and TTF-1.
Histopathological variants
- •
Microcarcinomas
- •
Papillary thyroid microcarcinoma (PTMC) is defined by the World Health Organisation (WHO) as a PTC that is 1.0 cm or less in largest dimension. Its prevalence in autopsy series varies widely, from 6–7% in the USA to 35% in Finland. It is being diagnosed with increasing frequency due to the widespread use of US and FNA biopsy, as well as the improved resolution of ultrasonography. This increase has contributed significantly to the overall increase in incidence of newly diagnosed PTC. The prognosis for this group of patients had been shown to be excellent in a large study, despite 30% nodal involvement on presentation. A study from the Mayo Clinic also showed that PTMC does not affect overall survival, and that neither postoperative radioactive iodine (RAI) nor total thyroidectomy (or completion thyroidectomy) reduced recurrence rates compared to unilateral lobectomy. However, multifocal tumours and nodal positivity were predictors of recurrence.
- •
Follicular variant
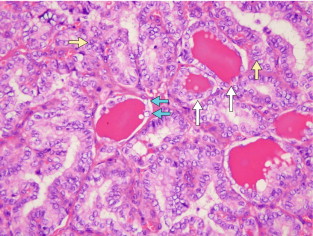
The follicular variant of PTC can be a challenge for pathologists to diagnose accurately. This variant displays follicular architecture, but retains the nuclear features of PTC ( Fig. 2.6 ). It can be confused with follicular adenoma or carcinoma. However, it is important to distinguish the follicular variant of PTC from FTC because the prognosis of these patients is similar to that of patients with PTC rather than FTC.
- •
Aggressive variants
The tall cell variant is an uncommon variant that is usually found in older patients. It has a more aggressive clinical behaviour, and is often associated with necrosis, mitotic activity and extrathyroidal extension (which are all aggressive features). Columnar and diffuse sclerosing variants are two other variants associated with more aggressive disease behaviour.
Follicular thyroid carcinoma
Like PTC, FTC is a malignancy of the epithelial follicular cells. However, unlike PTC, it lacks diagnostic nuclear features, so the diagnosis of FTC can only be made on histological examination ( Fig. 2.7 ). Aspirates that are hypercellular, with a microfollicular pattern and scant colloid, suggest follicular neoplasm (adenoma or carcinoma). FTC can only be diagnosed by histological confirmation of capsular invasion and/or vascular invasion. The main variants are conventional or oncocytic types. The oncocytic type is also known as Hurthle cell carcinoma.
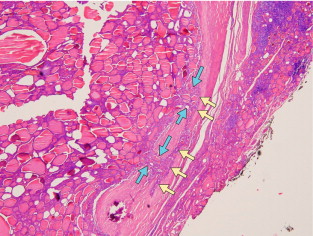
Minimally invasive follicular carcinoma is used by some to describe tumours that show only capsular invasion, as distinct from grossly encapsulated angioinvasive follicular carcinoma. The latter carries a higher risk of distant metastasis secondary to the angioinvasion.
Hurthle cell variant
Also known as oncocytic variant or oxyphil cell carcinoma, these tumours are composed of predominantly (> 75%) oncocytic cells. Mitochondrial abnormalities are thought to be the cause of these tumours. Unlike the conventional type of FTC, where lymph node involvement is seen in < 5% of patients, the oncocytic variants are associated with lymph node metastases in up to 30% of patients.
Staging
As many as 17 staging and prognostic systems for DTC have been reported in the literature since 1979. The sixth edition AJCC/IUCC staging system is currently one of the most commonly used systems. Other commonly used systems include AMES (Age, Metastasis, Extent, Size) from the Lahey Clinic, AGES (Age, Grade, Extent, Size) and MACIS (Metastasis, Age, Completeness of resection, Invasion, Size) from the Mayo Clinic, and EORTC staging from the European Organisation for Research and Treatment of Cancer. Some interesting observations can be made from these systems. Like all other staging systems, pathological features such as tumour size, grade, extent and presence of metastasis feature uniformly throughout the different classification systems. However, nodal status is notably absent in many of these systems apart from the AJCC system. This relates to the fact that these systems were mostly developed to predict disease-specific survival, and so far most studies do not correlate nodal status with survival, although the evidence is not conclusive at this stage. This must be borne in mind when using these prognostic systems during patient follow-up for recurrence. The ATA published a three-tier risk stratification that is useful for the purpose of surveillance ( Table 2.5 ). Another unique factor in the AJCC staging for DTC is the inclusion of age. Patients under the age of 45 have excellent prognosis regardless of nodal status, and a small decrease in survival in the presence of metastases. As such, the highest stage for patients under 45 years of age is Stage II.