Fig. 11.1
Schematic representation of the most important hormonal mechanisms regulating canine luteal function (Modified from Kowalewski et al. (2014b)). A detailed explanation is provided in the text. COX2/PTGS2 cyclooxygenase 2 (PTGS2), PTGES PGE2 synthase, PRLR PRL receptor, STAR steroidogenic acute regulatory protein, 3βHSD (HSD3B2) 3β-hydroxysteroid-dehydrogenase, sER smooth endoplasmic reticulum, VEGF vascular endothelial growth factor
Follicular luteinization can be observed as early as 6 days before first significant LH increase (LH surge) and is reflected in slowly increasing P4 levels from basal values of 0.2–0.4 ng/ml to the levels of 0.6–1.0 ng/ml observed at the preovulatory LH surge (Concannon 2009). Coincident with the onset of the LH surge (0.5–3 days after the estrogen peak), the preovulatory luteinization becomes very intense and P4 increases rapidly, concomitantly with still decreasing estrogen levels (Fig. 11.2). Ovulation takes place at 48–60 h (2–3 days) after the initial LH surge (Concannon 2009) and is accompanied by relatively high circulating P4 levels of about 5 ng/ml (Concannon et al. 1989) (Figs. 11.1 and 11.2). The phenomenon of preovulatory luteinization, described for the first time in the dog by Bischoff in his work (Bischoff 1845), is not unique to the dog and, even if not that intense, can also be observed, for example, in pigs, rodents, and primates. As recently shown, analogous to other species, ovulation is associated with high PGE2 and PGF2α concentrations in the forming CL (Tsafriri et al. 1972; Iesaka et al. 1975; Kowalewski et al. 2014a), implicating their involvement in this process. Following ovulation, and before the end of estrus (male acceptance), the structural formation of the CL begins. The remaining long-lasting phase of luteal activity is commonly referred to in the literature as diestrus. Progesterone concentrations rise rapidly and vary widely between individual animals, reaching maximal circulating levels of 30–35 ng/ml (sometimes even up to 80 ng/ml or higher) within 15–30 days (Concannon et al. 1989; Concannon 2011). The turning point of luteal steroidogenic activity is indicated by the onset of a slow luteal regression as becomes obvious by the gradually decreasing P4 levels. Especially following implantation and placenta formation, the mean circulating P4 levels tend to be numerically but not statistically higher in pregnant dogs (Steinetz et al. 1989), precluding the use of P4 levels as an endocrine marker for pregnancy detection. These diverging P4 levels observed during the second half of gestation, compared with nonpregnant cycles, are likely to result from the increased prolactin (PRL) levels also measured during the same period approximately 4–6 weeks following the LH surge (reviewed in (Concannon 2009; Kowalewski et al. 2014a)). This enhanced PRL secretion could be initiated by the simultaneously or slightly earlier (days 25–30 from the preovulatory LH surge) increasing secretion of relaxin from placental syncytiotrophoblast (Klonisch et al. 1999). Such a PRL-releasing role for relaxin was reported for pigs and monkeys (Bethea et al. 1989; Li et al. 1993). Similarly as for P4, the high peripheral PRL concentrations observed in overtly pseudopregnant bitches also preclude using this hormone as a reliable marker for detection of pregnancy. Instead, placental relaxin is the only suitable endocrine marker of pregnancy.
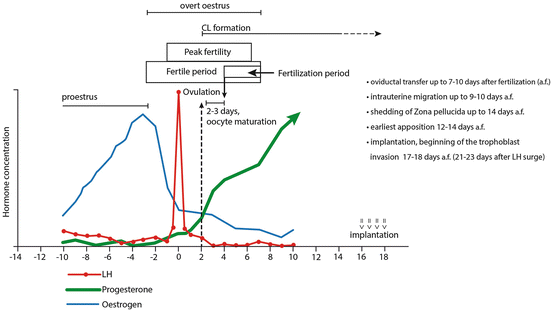

Fig. 11.2
The timeline of endocrine pre- and postovulatory events and timing of early embryonic development in the dog (Modified after England and Pacey (1998)). A detailed explanation is provided in the text. Briefly, the strong preovulatory luteinization results in relatively high circulating progesterone levels of about 5 ng/ml at the time of ovulation, which takes place 48–60 h (2–3 days) after the first significant LH increase above the basal level (referred to as LH surge). The latter occurs 0.5–3 days after the estrogen peak. Following ovulation and before the end of male acceptance (overt estrus), the structural formation of the CL begins. The long-lasting phase of luteal activity is commonly referred to as diestrus. The oocyte maturation and completion of first meiotic division is delayed in the dog and takes place 2–3 days after ovulation (i.e., 4–5 days after the initial preovulatory LH surge). The fertilization period is long and variable due to the extended life span of intrauterine spermatozoa (up to 7 days) and oviductal oocytes (7–8 days including up to 5 days, which follow the oocyte maturation). (a.f. after fertilization)
At approximately day 60 of the luteal life span, the P4 profiles, which up until then are similar in pregnant and nonpregnant dogs, start to diverge. At this time, the steep P4 decline, which is observed in pregnant dogs, signals the initiation of prepartum luteolysis (Nohr et al. 1993). This is accompanied by a concomitant increase in PGF2α concentrations in the maternal circulation, indicating the role of PGF2α during both luteolysis and parturition. The placenta fetalis appears to be the main source of this PGF2α increase (Kowalewski et al. 2010; Gram et al. 2013, 2014b).
Interestingly, the nonpregnant uterus does not exert any effect on luteal function, since normal ovarian cyclicity is maintained following hysterectomy (Hoffmann et al. 1992). In other words, there is no acute uterine luteolytic mechanism in the absence of pregnancy. Moreover, any luteolytic function of prostaglandins produced by the CL could be ruled out (Hoffmann et al. 1992; Kowalewski et al. 2006b, 2009; Gram et al. 2013). As a result thereof, a physiological pseudopregnancy can be observed, characterized by the aforementioned similar luteal life span in pregnant and nonpregnant animals, until shortly before parturition.
The lack of an active luteolytic principle indicates that luteal regression in dogs is a passive degenerative process and a part of the inherently controlled luteal life span. This apparently biologically preprogrammed aging process allows extension of the luteal phase in nonpregnant dogs beyond the time equivalent to parturition. Consequently, luteal function in pseudopregnant bitches fades out slowly, generating gradually decreasing P4 concentrations. This, following the highest P4 concentrations measured at the mid-luteal phase, can even last as long as 1–3 months. Once peripheral P4 reaches levels below 1 ng/ml, the canine reproductive cycle, per definition, enters anestrus (see review Kowalewski 2012, 2014). In contrast to the actively regulated prepartum luteolysis, which is associated with strong apoptotic signals occurring concomitantly with the prepartum PGF2α increase, during the extended luteal regression only sporadic apoptotic signals can be observed (Hoffmann et al. 2004b; Luz et al. 2006; Kowalewski 2014).
Furthermore, estrogens seem to be entirely of luteal origin, as suggested from the absence of detectable placental aromatase activity (Hoffmann et al. 1994; Nishiyama et al. 1999) and the time-dependent luteal expression of aromatase (Papa and Hoffmann 2011). Displaying variable serum concentrations, the profile of estradiol production during diestrus/pregnancy parallels to some extent that of P4. It increases slightly after the end of estrus, is higher in the mid-luteal phase, and decreases thereafter. Importantly, the mid-diestrus increase is not pregnancy-specific and the rapid prepartum drop, which additionally suggests its ovarian origin (Hoffmann et al. 1994; Onclin et al. 2002), is striking. Similarly striking is the lack of prepartum increase of cortisol. Its erratic presence in the maternal circulation seems not to be mandatory for normal parturition and was linked to maternal stress (Hoffmann et al. 1994). However, as suggested by Concannon and collaborators (Concannon et al. 1978), the cortisol levels observed in the peripheral circulation may not reflect possible increases at the uterine and/or placental levels. Both the estradiol and cortisol secretion patterns further emphasize the differences in mechanisms of endocrine control of canine pregnancy compared with most other domestic animal species.
11.2.2 Morphological Aspects and Regulatory Mechanisms
The exceptionally intense preovulatory luteinization of ovarian follicles in dogs is mirrored in a strong folding of proliferating theca interna layers. Unlike in most other species investigated so far, morphologically and ultrastructurally the canine CL consists of only one steroidogenic cell type. The cellular origin of the canine CL was, until lately, a subject of scientific debate (Concannon 2011; Kowalewski 2014). However, our recent histological documentation of postovulatory follicles revealed the presence of strongly proliferating theca cells associated with vascular structures and separated by remnants of basement membrane from the not yet vascularized luteinizing granulosa cells (Kowalewski et al. 2014a). Nevertheless, further investigations are needed that would elucidate the process of development of the uniform CL cell population.
The early luteal phase, which follows ovulation, is characterized by strong proliferative and vasculogenic activity driven, at least in part, by hypoxia (Papa et al. 2014) and associated with increased infiltration of immune cells (Hoffmann et al. 2004a), cumulatively leading to continuously and rapidly increasing steroidogenic activity. The latter is directly reflected in elevated expression of steroidogenic acute regulatory (STAR) protein and 3-β-hydroxysteroid-dehydrogenase (3βHSD, HSD3B2) (Kowalewski et al. 2006a; Kowalewski and Hoffmann 2008), which is translated into dynamically rising luteal P4 output as discussed above. During this time, the CL is responsible for providing P4 required for facilitating uterine responsiveness for embryo implantation. Around implantation, which in dogs takes place at approximately day 17–18 after mating (Amoroso 1952; Kehrer 1973), the canine CL is already well developed, reaching the mature stage characterized by maximal steroidogenic activity, shortly thereafter, at about day 20–25 after ovulation. Acting at the level of their receptors, P4 and estrogens are among the luteotrophic factors, and treatment with an antiprogestagen at any time during gestation unequivocally results in luteolysis (Kowalewski et al. 2009). Although both PRL and LH are luteotrophic, with PRL being the predominant luteotrophin as early as day 25 after the preovulatory LH peak, early luteal development appears to be characterized by a period of transitional gonadotropic independence (see review Kowalewski 2014). During this time, locally produced (intra-CL) prostaglandins, and especially PGE2, appear to be among the most important luteotrophic factors (Kowalewski et al. 2006b, 2008a, 2009). The development of the CL is associated with increased PGE2 and low PGF2α luteal content (Kowalewski et al. 2014a).
Recently, in addition to our in vitro studies (Kowalewski et al. 2013) with dispersed canine lutein cells, compelling evidence for the luteotrophic function of prostaglandins in canine CL has been provided in vivo by applying a selective cyclooxygenase 2 (COX2/PTGS2) inhibitor during early diestrus (Janowski et al. 2014; Kowalewski et al. 2014a). This treatment resulted in significantly decreased levels not only of the respective PGE2-synthase (PTGES)-encoding gene but also in suppression of luteal PGE2 production. The impaired CL function was associated with suppressed STAR expression, clearly indicating the causality between PTGS2 function and PGE2 synthesis in the canine CL (Kowalewski et al. 2014a). Additionally, as indicated by both our in vivo and in vitro experiments, PGE2 seems to be involved indirectly in the luteotrophic function of PRL by stimulating expression of its receptor (PRLR). This seems to be a new regulatory pathway not previously described for species more dependent than dogs on LH for luteal maintenance, e.g., cattle and pigs (Kowalewski et al. 2014a).
The slowly ongoing luteal regression appears to be a process of both cellular degeneration and structural remodeling. The role of luteotrophic support during this period of the luteal life span seems to be in sustaining CL function, rather than in active stimulation of P4 production, thereby supporting maintenance of canine gestation. The first signs of structural degeneration can be observed ultrastructurally as early as day 30 after ovulation. At this time, the smooth endoplasmic reticulum (sER) exhibits whirl-like structures, moves toward the periphery of the lutein cell, and at approximately day 45 encircles large lipid droplets (Fig. 11.1). This, together with large lipid vacuoles observed all over the cytoplasm, can be seen as a further indication of fatty degeneration (Hoffmann et al. 2004b). Further structural remodeling and proliferation of connective tissue components complete the process of progressive reduction of luteal function and corpus albicans formation.
11.3 Early Embryonic Development
Exact knowledge of oocyte maturation and timing of embryonic development is mandatory for fully understanding reproductive physiology; yet in the bitch the data concerning these events are largely imprecise. This is mostly due to the different and imprecise “starting points” concerning several endocrinological, morphological, and behavioral events related to the time of ovulation, such as the LH surge, peripheral progesterone levels, acceptance of the male, or cytological appearance (see Holst and Phemister 1971; Archbald et al. 1980; Renton et al. 1991; Bysted et al. 2001; Concannon et al. 2001; Hatoya et al. 2006).
Oocyte maturation, and completion of the first meiotic division, which is uniquely delayed in the dog, takes place 2–3 days after ovulation, i.e., 4–5 days after the preovulatory LH surge (Concannon et al. 1989) (Fig. 11.2). Oocyte maturation in Canidae is completed within the oviduct, unlike all other mammals in which this process occurs within the ovarian follicle; this complicates efforts to study fertilization and early embryo development in vitro in the dog.
Moreover, the uterus, and not the oviductal ampulla or isthmus, seems to be the major sperm reservoir in the bitch (Bischoff 1845; Pacey et al. 2000; Rijsselaere et al. 2014). The “fertilization window,” i.e., the time period when a bitch can be successfully mated, is relatively long and variable. Thus, mating can occur as early as 5 days before ovulation or as late as 6 days afterward (Fig. 11.2). This is because of the extended life span of intrauterine spermatozoa (up to 7 days, considerably longer than in most other domestic species) and of oviductal oocytes (7–8 days, including up to 5 days, which follow the delayed oocyte maturation) (Bischoff 1845; Concannon 2009). Also, embryonic development in dogs is slow. It takes longer for embryos to reach the uterotubal junction and enter the tip of uterine horns, 7–10 days following fertilization, compared to 3–4 days in other species (Holst and Phemister 1971; Concannon 2009), at this time being as early as the 16 cell stage, but usually morulae or early blastocysts (Holst and Phemister 1971). This agrees with the earliest descriptions by Bischoff (1845), who never observed the uterine presence of embryos before day 8 after mating.
The subsequent intrauterine transcornual migration and distribution of embryos take up to 9–10 days (Shimizu et al. 1990; Bysted et al. 2001) and are not influenced by the number of oocytes ovulated from either of the ovaries (Tsutsui et al. 2002). Blastocysts can still be enclosed by the zona pellucida as late as day 19 after the LH surge (around 14 days after oocyte maturation/fertilization) (Concannon et al. 2001). The apposition of blastocysts to the uterine epithelium can, however, already be observed as early as between days 12 and 14 after oocyte maturation (fertilization), and the associated uterine swellings, implantation, and, shortly afterward, invasion, normally occur by days 17–18 after fertilization (Amoroso 1952; Kehrer 1973). The latter, taking place at the end of the first third of gestation, is rather late considering the entire length of canine pregnancy (Fig. 11.2).
11.4 Role of Progesterone in Implantation and Placentation in Dogs
By regulating the secretory activity of the endometrium and stimulating expression of genes involved in uterine receptivity both in cyclic and pregnant animals, P4, at least in part, indirectly regulates embryonic development by maintaining the uterus in a state of physiological receptivity for the conceptus (see review Dorniak and Spencer 2013). By expressing the so-called “decidualization markers”, P4 is responsible for the morphological remodeling processes of the endometrium during decidua formation. Adequate levels of circulating P4 are essential for successful establishment of early pregnancy and, if too low, can lead to embryo loss in some species, e.g., in cattle (Mann and Lamming 2001; Lonergan 2011). Other than that, the canine uterus is normally exposed to P4 levels, which by far exceed those required for establishment and maintenance of pregnancy. As reported by Concannon (Concannon et al. 2001; Concannon 2009), exogenous supplementation with P4 in bitches that were ovariectomized as early as at day 14 after the LH surge maintained pregnancy even though serum concentrations of this hormone were chronically below 5 ng/ml; the PRL concentrations following implantation did not differ from the levels expected during normal pregnancy (Concannon et al. 2001). Therefore, based on this study, it has been concluded that P4 is the predominant, if not the only, luteal steroid needed for implantation and placentation in previously estrogenized bitches. Unlike in humans and several domestic animal species, and as already indicated above, there is no large increase in estrogens during canine pregnancy, and levels observed in the second half of gestation never exceed those observed shortly before ovulation (Concannon et al. 2001). The use of natural P4 in these experiments precludes any possible estrogenic effect exerted by synthetic hormones. However, it should be noted that the synthetic capabilities of canine embryos toward conversion of progesterone or androgens to estrogens are not known.
11.5 Preimplantation Embryo-Maternal Communication
Being the only domestic animal species devoid of an active luteolytic principle in the absence of pregnancy, and therefore exhibiting similar hormonal profiles during early pregnancy and pseudopregnancy, the dog appears to lack an important regulatory mechanism that would facilitate reproductive events and allow for faster procreation, i.e., by shortening the nonpregnant cycle. Needless to say, this situation strongly contrasts with what is observed in livestock, in which cyclicity is maintained due to periodic secretion of a uterine luteolysin, i.e., PGF2α. Consequently (reviewed elsewhere), to allow successful establishment of pregnancy, different strategies for the maternal recognition of pregnancy have evolved to prevent luteolysis and preserve endogenous progesterone concentrations by extending the luteal life span. Briefly, in ruminants this task is fulfilled by IFN-τ produced by the early embryo (Bazer et al. 1991; Spencer and Bazer 2004). Being produced between days 10 and 21–25, its function, among others, is to suppress PGF2α production by inhibiting ERα and oxytocin receptor (OXTR) expression. In pigs, the trophoblast produces estrogens, which are responsible for redirection of uterine PGF2α secretion from endocrine to exocrine secretion and thus prevent luteolysis, which normally occurs on days 15–16 of the estrous cycle (Bazer and Thatcher 1977; Spencer and Bazer 2004). This switch from endocrine to exocrine secretion starts with embryonic estrogen production between days 10 and 12 (Bazer 1989). Nevertheless, in the dog, as in these other species, there must be some kind of synchronization between blastocyst development and uterine preparation for pregnancy in order to support gestation, even though its mechanism is not through suppressing luteolysis.
Some earlier studies addressed the biochemical and endocrine milieu characteristic of early canine gestation in attempts to find new early pregnancy detection markers. A major change in the circulating levels of acute phase proteins was found on days 28–37 after fertilization in the blood of pregnant animals, but not in corresponding healthy nonpregnant bitches (Evans and Anderton 1992). Similarly, concomitantly with increased levels of relaxin, strongly elevated concentrations of fibrinogen and serum C-reactive protein-like (CRP-LI) were found in pregnant dogs between days 21 and 30 after the LH surge and maintained until day 50 (Eckersall et al. 1993; Concannon et al. 1996). However, acting as mediators of inflammation, these acute phase proteins may be triggered by infections or trauma. Thus, the rise observed at pregnancy cannot be seen as pregnancy-specific, since it is apparent that dogs suffering from some infections could be falsely classified as being pregnant (Evans and Anderton 1992).
At the uterine level, efforts were undertaken to characterize secretion of proteins synthesized during pregnancy. Uteri collected from dogs during early nonpregnant diestrus were compared with their counterparts obtained from dogs prior to embryos traversing the uterotubal junction and during their free-floating phase prior to implantation. Two major protein complexes, designated as cP5 and cP6, were detected as differentially expressed; one of them (cP6) proved highly similar to retinol binding proteins (Buhi et al. 1992, 1993, 1995). While their secretion differed by day, it did not differ by status, i.e., in pregnant vs. nonpregnant dogs.
More recently, some insight into immunological processes possibly involved in regulating embryo-maternal contact was provided by investigating the expression of several cytokines and growth factors in the early pregnant, preimplantation uterus (day 10 of gestation), and comparing their expression in corresponding nonpregnant uteri. Among the most interesting findings was the expression of CD8, IL4, and IFNγ mRNA, which seemed to be targeted to the preimplantation uterus, whereas the expression of CD4, TNFα, and IL6 was found abundantly in the nonpregnant uterus (Schafer-Somi et al. 2008). Additionally, in the same study, transcripts encoding for TGFβ, IL2, IL10, and LIF were only detected in the early pregnant uterus. In another study from the same group, the expression of Fas ligand (FasL) and its receptor (Fas) mRNA did not differ between early pregnant and nonpregnant uteri (Schafer-Somi et al. 2012). On the other side, the 10-day-old embryos tested positively for several factors, such as GM-CSF, IL1β, IL6, IL8, LIF, and CD4 (Schafer-Somi et al. 2008) (Fig. 11.3). Although most of these data were generated utilizing a qualitative transcriptional approach and thus require further confirmation, they clearly indicate differential, possibly embryo-mediated, regulation of uterine function and modulation of the uterine immune response in order to avoid embryo rejection and facilitate implantation, which is known to occur in other species.
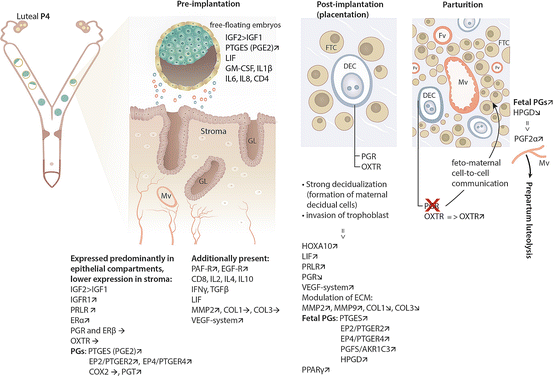

Fig. 11.3
Schematic representation of signals participating in embryo- and feto-maternal communication in the dog and underlying effects exerted on the pregnant canine uterus. Preimplantation, effects exerted by free-floating embryos; arrows indicate uterine expression of different factors compared with their expression levels in the nonpregnant uterus. Post-implantation and at parturition, schematic pictures represent fragments of the canine endotheliochorial placenta; maternal decidual cells (DEC) are present in a direct contact with fetal trophoblast cells (FTC). DEC are the only cells expressing progesterone receptor (PGR) within the canine placenta. Following implantation, utero-placental expression of several regulatory factors is indicated. The role of decidual cells in the maintenance of pregnancy and their involvement in the endocrine cascade within the canine placenta resulting in prepartum output of luteolytic PGF2α are presented. Thus, blocking PGR function in the placenta materna (decidual cells) leads to activation of fetal PGs synthesis. A detailed explanation is provided in the text. Mv maternal blood vessel, GL endometrial glands, Fv fetal vessels, PGs prostaglandins
In addition to these earlier studies, lately we have investigated the uterine expression of several target genes potentially involved in the process of early decidualization (the so-called “decidualization markers”) (Kautz et al. 2014). The uterine response to the presence of free-floating embryos during the preimplantation stage of pregnancy (days 10–12 of gestation, determined by uterine flushings) was investigated. The majority of embryos, 63 %, were hatched blastocysts while the remaining 37 % were unhatched blastocysts. Dogs inseminated but determined as nonpregnant in uterine flushing were used as controls in these studies. The effects of seminal plasma alone were not separately investigated.
Being implicated in various cellular activities like proliferation and differentiation, insulin-like growth factors 1 and 2 (IGF1 and IGF2) and PRL are among the most prominent decidualization markers (Irwin et al. 1994; Ramathal et al. 2010). Functionally, the mitogenic activity of IGF1 and IGF2 is mediated mainly through their type 1 receptor (IGF1R) (see review Yu et al. 2011). As for the canine uterus, increased preimplantation expression of IGF2, but not of IGF1, was found. While it was not affected at the level of transcript expression, IGF1R was clearly detectable and abundantly expressed at the protein level during early pregnancy. Similarly, at the same time, IGF2 expression was higher than that of IGF1 in the hatched embryos, suggesting a possible predominant role of IGF2 during the establishment of early canine pregnancy. Both IGF1 and IGF2 were below the detection limits in the unhatched embryos (Kautz et al. 2014). The presence of early preimplantation embryos was also associated with increased expression of receptors for two other important growth factors, i.e., platelet-activating factor (PAF) and epidermal growth factor (EGF) (Schafer-Somi et al. 2013) (Fig. 11.3).
Interestingly, in contrast to rodents and primates (Prigent-Tessier et al. 1999; Tseng and Mazella 1999) in which PRL is one of the most prominent decidualization markers involved, e.g., in regulating endometrial glandular secretory activity (Jabbour et al. 1998), the canine uterus does not exhibit a strong capacity to express PRL. Its expression at the transcript level was low and frequently below the detection limit in our studies. Thus, the uterus does not seem to contribute strongly to the overall circulating PRL in the dog. However, the highly upregulated PRLR expression (Kowalewski et al. 2011b; Kautz et al. 2014) could serve to compensate for low PRL expression and thereby to locally increase its relative availability. The expression of LH receptor, which was recently suggested to be involved in implantation in mice (Gridelet et al. 2013), was significantly reduced in the early pregnant canine uterus (Kautz et al. 2014).
As for the steroidogenic hormone receptors, ERα was significantly upregulated in early pregnant canine uteri compared with nonpregnant diestrus controls, while ERβ and progesterone receptor (PGR) remained unaffected (Kautz et al. 2014). Similarly to the situation with PRL, the increased expression of ERα could possibly counteract the low availability of estradiol of luteal origin during canine pregnancy (Fig. 11.3). The oxytocin receptor (OXTR) could be involved in mechanisms regulating the distribution and positioning of free-floating embryos before attachment and is a known mediator of local prostaglandin effects. As for the canine uterus, its uterine expression varied greatly between early pregnant and nonpregnant bitches, so no further conclusions about its role could be drawn (Gram et al. 2014a; Kautz et al. 2014). It is noteworthy that, throughout the above-cited studies (Kautz et al. 2014), stronger uterine signals for the protein expression of the respective genes (IGF1, IGF2, IGFR1, ERα, ERβ, PGR, OXTR) were observed in the endometrial epithelial compartments, i.e., in luminal and glandular epithelium, compared with the weaker signals found in the uterine stromal cells. This was interpreted as an indication of the early stage of uterine differentiation observed at the beginning of pregnancy (preimplantation stage), which is further supported by the unaltered expression of E-cadherin (CDH1) (Kautz et al. 2014), a cell adhesion protein known for controlling migratory activity in different cell types.
11.5.1 Prostaglandins
Concerning the prostaglandin family members, our attention was especially drawn to the increased expression of prostaglandin transporter (PGT) and of PTGES and its two G protein-coupled receptors, PTGER2 and PTGER4, observed in the early pregnant canine uterus (Kautz et al. 2014) (Fig. 11.3). This interest has been further strengthened in our recent studies on the biological function and expression of PTGES in uterine and placental tissues throughout canine gestation (Gram et al. 2014b). Based on its subcellular localization bound to the endoplasmic reticulum, the only currently available canine PTGES, which was cloned in our laboratory (GenBank: NM_001122854; Kowalewski et al. 2008a), appears to be a microsomal protein corresponding to the inducible microsomal isoform known from other species (Gram et al. 2014b). In addition, as presented above, being one of the most important luteotrophic factors in the dog, acting mostly through the cAMP/PKA signaling pathway, PGE2 was shown to accelerate decidualization mediated by P4 and cAMP (Brar et al. 1997). Similar effects were observed in rats (Kennedy and Doktorcik 1988). Furthermore, by inhibiting IL2 and expression of its receptor in human decidua, PGE2 was shown to block activation of maternal leukocytes with potential anti-trophoblast function, thereby revealing an immunosuppressive but embryo-protective effect (Parhar et al. 1989). The embryo- and luteo-protective role of endometrial PGE2 has been discussed for pigs, in which additionally, species-specifically, estrogen also increases PGE2 secretion (see review Ziecik et al. 2011). Similar effects could also apply to the canine species as suggested by the concomitantly and abundantly expressed PTGES levels in hatched compared to unhatched preimplantation embryos (Kautz et al. 2014). Together with the strongly increased PTGES expression in the course of cAMP-induced decidualization of canine uterine stromal cells isolated from early dioestrus(own data, unpublished), there is emerging evidence identifying PGE2 as an important factor involved in canine decidualization. This effect could be amplified by the local PGE2 feedback loop since PTGER2 and PTGER4 are known mediators of cAMP/PKA pathway-coupled effects. The local effects of prostaglandins could be additionally coordinated by the clearly detectable expression of endometrial HPGD (15-hydroxyprostaglandin dehydrogenase) that is responsible for biochemical deactivation of prostaglandins (Gram et al. 2013; Kautz et al. 2014). Finally, as also observed in our previous experiments (Kautz et al. 2014), together with IGF2, the expression of PTGES was abundant in hatched embryos flushed from preimplantation canine uteri, indicating a possible functional interplay between these two entities as embryo-derived factors regulating canine decidualization (Fig. 11.3).
11.5.2 Structural Remodeling
Remodeling of uterine matrix is essential for preparation of invasive growth of the cytotrophoblast, especially when the highly invasive canine placentation type is considered. As an integral part of it, extracellular matrix proteins (ECM), which are secreted by the decidualizing uterus in response to the changing uterine milieu and which interact with other components of the uterine environment, are critical players during implantation and placentation. Several processes important for cell adhesion, migration, and differentiation are mediated by these components, such as integrins, e.g., aberrant expression of some of the integrin family members is associated with infertility and recurrent pregnancy loss (Lessey et al. 1995; Liu et al. 2012).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree






