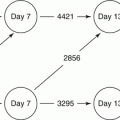
Fig. 9.1
Timeline of events during early pregnancy in the mare
9.11 Conclusions
Indirect conclusions can be drawn only about the time frame during which maternal recognition of pregnancy occurs. The conceptus has to be recognized before Day 14 after ovulation, as this is the time that luteolysis is initiated in the mare (Ginther et al. 2011). Although expression of oxytocin receptors is similar for nonpregnant and pregnant mares on Days 10 and 12 after ovulation, expression of oxytocin receptors is less for pregnant than cyclic mares on Day 14 after ovulation (de Ruijter-Villani et al. 2014; Sharp et al. 1997; Starbuck et al. 1998). Continuous infusions of oxytocin from 8 days after ovulation prolong luteal phase, whereas continuous infusions of oxytocin from 10 days after ovulation initiate luteolysis (Stout et al. 1999), indicating that maternal recognition of pregnancy is initiated by Day 10 postovulation. Wilsher and coworkers (2010) recently suggested reevaluation of the concept that maternal recognition of pregnancy occurs on or before Day 10. Surprisingly, all six asynchronous transfers of 10-day-old blastocysts into recipient mares on Day 12 of diestrus resulted in establishment of pregnancy and survival of the conceptus to the “heartbeat stage” of development of the embryo (end of observation period). Notwithstanding, various mechanisms lead to luteal maintenance when using repeated/continuous administration of oxytocin versus transfer of a blastocyst as in vitro factors secreted by the conceptus lead to a rapid (within 24 h) reduction in secretion of PGF2α by endometrial explant cultures (Ealy et al. 2010). It seems likely that the mechanism leading to luteal maintenance via repeated or continuous treatments with oxytocin must be initiated well before maternal recognition of pregnancy occurs.
References
Ababneh MM, Troedsson MH (2013a) Endometrial phospholipase A2 activity during the oestrous cycle and early pregnancy in mares. Reprod Domest Anim 48:46–52. doi:10.1111/j.1439-0531.2012.02023.x PubMed
Ababneh MM, Troedsson MH (2013b) Ovarian steroid regulation of endometrial phospholipase A2 isoforms in horses. Reprod Domest Anim 48:311–316. doi:10.1111/j.1439-0531.2012.02151.x PubMed
Ababneh MM, Troedsson MH, Michelson JR, Seguin BE (2000) Partial characterization of an equine conceptus prostaglandin inhibitory factor. J Reprod Fertil Suppl (56):607–613
Ababneh M, Ababneh H, Shidaifat F (2011) Expression of cytosolic phospholipase A2 in equine endometrium during the oestrous cycle and early pregnancy. Reprod Domest Anim 46:268–274. doi:10.1111/j.1439-0531.2010.01657.x PubMed
Adams AP, Oriol JG, Campbell RE, Oppenheim YC, Allen WR, Antczak DF (2007) The effect of skin allografting on the equine endometrial cup reaction. Theriogenology 68:237–247. doi:10.1016/j.theriogenology.2007.04.058 PubMedCentralPubMed
Albihn A, Waelchli RO, Samper J, Oriol JG, Croy BA, Betteridge KJ (2003) Production of capsular material by equine trophoblast transplanted into immunodeficient mice. Reproduction 125:855–863PubMed
Allen WR, Rowson LE (1973) Control of the mare’s oestrous cycle by prostaglandins. J Reprod Fertil 33:539–543PubMed
Allen WR, Stewart F (2001) Equine placentation. Reprod Fertil Dev 13:623–634PubMed
Amoroso EC, Hancock JL, Rowlands IW (1948) Ovarian activity in the pregnant mare. Nature 161:355PubMed
Antczak DF, Bright SM, Remick LH, Bauman BE (1982) Lymphocyte alloantigens of the horse. I. Serologic and genetic studies. Tissue Antigens 20:172–187PubMed
Arnold CE, Love CC (2013) Laparoscopic evaluation of oviductal patency in the standing mare. Theriogenology 79:905–910. doi:10.1016/j.theriogenology.2012.12.004 PubMed
Atli MO, Kurar E, Kayis SA, Aslan S, Semacan A, Celik S, Guzeloglu A (2010) Evaluation of genes involved in prostaglandin action in equine endometrium during estrous cycle and early pregnancy. Anim Reprod Sci 122:124–132. doi:10.1016/j.anireprosci.2010.08.007 PubMed
Bae SE, Watson ED (2003) A light microscopic and ultrastructural study on the presence and location of oxytocin in the equine endometrium. Theriogenology 60:909–921PubMed
Banu SK, Arosh JA, Chapdelaine P, Fortier MA (2003) Molecular cloning and spatio-temporal expression of the prostaglandin transporter: a basis for the action of prostaglandins in the bovine reproductive system. Proc Natl Acad Sci U S A 100:11747–11752. doi:10.1073/pnas.1833330100 PubMedCentralPubMed
Battut I, Colchen S, Fieni F, Tainturier D, Bruyas JF (1997) Success rates when attempting to nonsurgically collect equine embryos at 144, 156 or 168 hours after ovulation. Equine Vet J Suppl (25):60–62
Bazer FW, Spencer TE, Ott TL (1997) Interferon tau: a novel pregnancy recognition signal. Am J Reprod Immunol 37:412–420PubMed
Beers SA, Buckland AG, Koduri RS, Cho W, Gelb MH, Wilton DC (2002) The antibacterial properties of secreted phospholipases A2: a major physiological role for the group IIA enzyme that depends on the very high pI of the enzyme to allow penetration of the bacterial cell wall. J Biol Chem 277:1788–1793. doi:10.1074/jbc.M109777200 PubMed
Behrendt-Adam CY, Adams MH, Simpson KS, McDowell KJ (1999) Oxytocin-neurophysin I mRNA abundance in equine uterine endometrium. Domest Anim Endocrinol 16:183–192PubMed
Berglund LA, Sharp DC, Vernon MW, Thatcher WW (1982) Effect of pregnancy and collection technique on prostaglandin F in the uterine lumen of Pony mares. J Reprod Fertil Suppl 32:335–341PubMed
Betteridge KJ, Mitchell D (1972) Retention of ova by the Fallopian tube in mares. J Reprod Fertil 31:515PubMed
Betteridge KJ, Mitchell D (1974) Direct evidence of retention of unfertilized ova in the oviduct of the mare. J Reprod Fertil 39:145–148PubMed
Betteridge KJ, Eaglesome MD, Mitchell D, Flood PF, Beriault R (1982) Development of horse embryos up to twenty two days after ovulation: observations on fresh specimens. J Anat 135:191–209PubMedCentralPubMed
Birts CN, Barton CH, Wilton DC (2008) A catalytically independent physiological function for human acute phase protein group IIA phospholipase A2: cellular uptake facilitates cell debris removal. J Biol Chem 283:5034–5045. doi:10.1074/jbc.M708844200 PubMed
Boerboom D, Brown KA, Vaillancourt D, Poitras P, Goff AK, Watanabe K, Dore M, Sirois J (2004) Expression of key prostaglandin synthases in equine endometrium during late diestrus and early pregnancy. Biol Reprod 70:391–399. doi:10.1095/biolreprod.103.020800 PubMed
Bowen JA, Bazer FW, Burghardt RC (1996) Spatial and temporal analyses of integrin and Muc-1 expression in porcine uterine epithelium and trophectoderm in vivo. Biol Reprod 55:1098–1106PubMed
Budik S, Lussy H, Aurich C (2010) Quantification of different type I interferon transcripts in equine embryos at days 10 to 16 of gestation. Anim Reprod Sci 121:307–308. doi: http://dx.doi.org/10.1016/j.anireprosci.2010.04.058
Catchpole HR, Cole HH, Pearson PB (1935) Studies on the rate of disappearance and fate of mare gonadotropic hormone following intravenous injection. Amer. J. Phy8iol. 112: 21–26
Cavanagh AC (1996) Identification of early pregnancy factor as chaperonin 10: implications for understanding its role. Rev Reprod 1:28–32PubMed
Chakraborti S (2003) Phospholipase A(2) isoforms: a perspective. Cell Signal 15:637–665PubMed
Choi I, Collante WR, Simmen RC, Simmen FA (1997a) A developmental switch in expression from blastocyst to endometrial/placental-type cytochrome P450 aromatase genes in the pig and horse. Biol Reprod 56:688–696PubMed
Choi SJ, Anderson GB, Roser JF (1997b) Production of free estrogens and estrogen conjugates by the preimplantation equine embryo. Theriogenology 47:457–466PubMed
Cochet M, Vaiman D, Lefevre F (2009) Novel interferon delta genes in mammals: cloning of one gene from the sheep, two genes expressed by the horse conceptus and discovery of related sequences in several taxa by genomic database screening. Gene 433:88–99. doi:10.1016/j.gene.2008.11.026 PubMed
Cole HH, Howell CE, Hart GH (1931) The changes occurring in the ovary of the mare during pregnancy. Anat Rec 49:199–209. doi:10.1002/ar.1090490305
Coleman RA, Smith WL, Narumiya S (1994) International Union of Pharmacology classification of prostanoid receptors: properties, distribution, and structure of the receptors and their subtypes. Pharmacol Rev 46:205–229PubMed
Cooke RG, Ahmad N (1998) Delayed luteolysis after intra-uterine infusions of nordihydroguaiaretic acid in the ewe. Anim Reprod Sci 52:113–121PubMed
Crossett B, Allen WR, Stewart F (1996) A 19 kDa protein secreted by the endometrium of the mare is a novel member of the lipocalin family. Biochem J 320(Pt 1):137–143PubMedCentralPubMed
Crossett B, Suire S, Herrler A, Allen WR, Stewart F (1998) Transfer of a uterine lipocalin from the endometrium of the mare to the developing equine conceptus. Biol Reprod 59:483–490PubMed
de Ruijter-Villani M, van Boxtel PR, Stout TA (2013) Fibroblast growth factor-2 expression in the preimplantation equine conceptus and endometrium of pregnant and cyclic mares. Theriogenology 80:979–989. doi:10.1016/j.theriogenology.2013.07.024 PubMed
de Ruijter-Villani M, van Tol HT, Stout TA (2014) Effect of pregnancy on endometrial expression of luteolytic pathway components in the mare. Reprod Fertil Dev. doi:10.1071/RD13381 PubMed
Diel de Amorim M, Nielsen K, Card C (2014) 113 Preliminary characterization of oxytocinase in equine serum. Reprod Fertil Dev 27:149. doi: http://dx.doi.org/10.1071/RDv27n1Ab113
Donaldson WL, Zhang CH, Oriol JG, Antczak DF (1990) Invasive equine trophoblast expresses conventional class I major histocompatibility complex antigens. Development 110:63–71PubMed
Douglas RH, Ginther OJ (1972) Effect of prostaglandin F2alpha on length of diestrus in mares. Prostaglandins 2:265–268PubMed
Douglas RH, Ginther OJ (1976) Concentration of prostaglandins F in uterine venous plasma of anesthetized mares during the estrous cycle and early pregnancy. Prostaglandins 11:251–260PubMed
Ealy AD, Eroh ML, Sharp DC 3rd (2010) Prostaglandin H synthase Type 2 is differentially expressed in endometrium based on pregnancy status in pony mares and responds to oxytocin and conceptus secretions in explant culture. Anim Reprod Sci 117:99–105. doi:10.1016/j.anireprosci.2009.03.014 PubMed
Enders AC, Liu IK (1991) Trophoblast-uterine interactions during equine chorionic girdle cell maturation, migration, and transformation. Am J Anat 192:366–381. doi:10.1002/aja.1001920405 PubMed
Evans HM, Gustus EL, Simpson ME (1933) Concentration of the gonadotropic hormone in pregnant mare’s serum. J Exp Med 58:569–574PubMedCentralPubMed
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree