The complication rates following surgery of the breast and axilla are considered low. The mortality after these procedures is less than 1%; most complications are wound related (ie, surgical site infections and seroma). These complications may lead to cosmetic compromise and psychological distress to the patient; result in increased costs, prolonged hospital stays, and frequent outpatient visits; and potentially delay important adjuvant therapies. In this chapter, the following topics are addressed:
- Early systemic complications (ie, cardiovascular complications, thromboembolic events, and allergic reactions)
- Early wound-related complications, including hematomas, wound infections, and seromas
- Long-term complications such as chronic breast cellulitis, lymphedema, and chronic pain
- Future directions for scientific study
In a prospective review of 3107 patients using National Surgical Quality Improvement Program (NSQIP) data, El-Tamer et al reported a 30-day mortality rate of 0.128% after surgery for breast cancer. The mortality rate was significantly increased in patients after mastectomy when compared with lumpectomy (0.24% vs 0.0%), but death remained a rare postoperative event. Breast cancer operations are commonly performed on an elective basis, enabling thorough preoperative evaluation and potential optimization of patients deemed to be high risk. In addition, patients with breast cancer are relatively healthy overall, as most (>85%) patients are classified as ASA 1 or 2.1
Furthermore, breast and axillary procedures are classified as clean, soft tissue operations, with no violation of the thoracic or abdominal cavity. As a result, serious infections are rare. As most patients are considered low risk, the incidence of cardiac complications after breast surgery is also low; the incidence of cardiovascular complications is 0.06% in mastectomy patients. The incidence of stroke is 0.1%. Pulmonary complications are also rare, even with the use of general anesthesia.1
It is well established that patients with a diagnosis of cancer are at an increased risk of thrombotic events. The estimated annual incidence of thrombotic events in the general population is 0.117%; cancer patients have a 4-fold increase in risk.2 The pathogenesis of the hypercoagulable state in cancer is multifactorial: activation of the coagulation and fibrinolytic cascades, change in the vascular endothelium, generation of acute phase reactants, and hemodynamic compromise.3
Patients undergoing surgery for breast cancer often exhibit multiple factors that put them at increased risk of thrombotic events: advanced age, diagnosis of malignancy, use of previous chemotherapy or hormonal therapy, and indwelling central venous catheters.4 The risk of thrombosis is patients with breast cancer is somewhat lower when compared with other malignancies (brain, pancreas, primary liver). The MD Anderson Cancer Center reported their results in a series of 3898 patients who underwent 4416 breast surgical procedures. Seven women developed venous thrombosis within 60 days of surgery, for a rate of 0.16% per procedure.2
There is little consensus regarding the optimal strategy for thromboembolic prophylaxis in the breast cancer population. Randomized trials in the general surgical, orthopedic, and gynecologic literature have demonstrated a substantial reduction in risk of thrombosis with use of prophylactic measures, with only a minimal increase in bleeding complications.5 However, little data exist on the use of prophylactic measures in those patients undergoing surgery for breast cancer. Breast surgical procedures are increasingly performed on an ambulatory, outpatient basis and tend not to be prolonged cases, questioning the need for prophylaxis.6 Surgeons are fearful that bleeding complications may result from preoperative anticoagulation.
A survey of 137 breast surgeons in the United Kingdom demonstrated wide variation both in the perceived risk of deep venous thrombosis (DVT) in the patient population and prophylaxis practices. Thirty percent of surgeons surveyed did not routinely prescribe thromboprophylaxis. In those who did, 9 different regimens were reported; the most common was subcutaneous heparin alone, followed by subcutaneous heparin with compressive stockings. Sixty-three percent of the surgeons who did not use routine prophylaxis considered their patient population “low risk” for venous thrombosis; 75.7% of the surgeons who frequently prescribed prophylaxis considered their patients “high risk.”7
Friis et al compared the use of low-molecular-weight heparin (LMWH) to compression hose in their series of 425 Danish women undergoing breast cancer surgery. No patient developed clinical evidence of DVT. The incidence of hematoma in women who received LMWH was 18.7%, compared with 6.8% in those treated with hose alone. In both groups, almost half of patients with hematoma required reoperation. In multivariate analysis, LMWH prophylaxis was independently associated with postoperative hematoma formation when compared with compressive stockings (OR 3.13, 95% CI 1.38-7.13).5 Additionally, in a recent series published in the British Journal of Surgery, Hardy et al found the use of LMWH to be associated with a 4-fold increase in hemorrhagic complications when compared with unfractionated heparin. No patient in either group developed a clinically evident DVT or pulmonary embolism (PE) within 30 days of surgery; the overall incidence of hematoma requiring reoperation was less than 2% in both groups.8 The incidence of DVT in these studies may be underrepresented, as up to 25% of patients may have no clinical manifestations.5
The National Comprehensive Cancer Network has attempted to address the issue of venous thrombosis in the cancer population. According to the most recent guidelines released in 2008, it is recommended that all hospitalized patients with cancer should undergo mechanical prophylaxis with pneumatic compression devices. Prophylactic anticoagulation with LMWH or unfractionated heparins should be considered in conjunction with pneumatic compression devices for those patients with multiple risk factors and no contraindications to anticoagulation.9 There is no consensus as to whether or not women with early stage breast cancer should routinely receive anticoagulants prior to outpatient or ambulatory breast surgery.
Lymphatic mapping with sentinel lymph node biopsy (SLNB) has become the standard of care for staging the axillary lymph nodes in women undergoing primary surgery for early stage breast cancer. Most surgeons use a combination of radiolabeled sulfur colloid and blue dye, with overall identification rates ranging from 65.5% to 94%.10 Adverse reactions have been reported in 1% to 4% of cases, most of which were minor in severity, including interference with pulse oximetry, discoloration of body fluids, and skin rashes.11 With widespread clinical adaptation of SLNB, isosulfan blue has been shown to be associated with life-threatening reactions.
Multiple authors have reported their experience with isosulfan blue dye in lymphatic mapping for breast cancer. In the largest series of 2392 patients who underwent SLNB for breast cancer at Memorial Sloan-Kettering Cancer Center using isosulfan blue dye, 1.6% patients had an allergic reaction. Sixty-nine percent presented with urticaria, pruritus, or skin reaction. Only 0.5% patients developed a severe episode of hypotension; the mean interval from injection to development of reaction was 44 minutes.12 Similar findings were reported by the authors at MD Anderson Cancer Center in their review of 1835 patients who had SLNB procedures for various types of cancer. The overall incidence of adverse events that could be attributed to isosulfan blue dye was 1.5%. In the subset of 583 patients undergoing breast surgical procedures, incidence was 2.0%. Most of these reactions (75%) were skin reactions, but half were associated with hypotensive episodes.13
Risk factors for the development of adverse reactions to isosulfan blue are unknown. Isosulfan blue is a triphenylmethane dye that is present in medications, cosmetics, and textiles; common use of these dyes may lead to sensitization by the general population.9 Patient allergies to other medications and environmental factors have not been associated with development of adverse reactions.13 Isosulfan blue contains 2 sulfur atoms, but no correlation has been made between a history of sulfa allergy and adverse events. Montgomery et al noted a 2.6% incidence of allergic reactions to isosulfan blue in patients with sulfa allergy, but this was not statistically different from those without.12 Chronic use of beta blockade and corticosteroids are unrelated to adverse events. Daley et al found the only significant risk factor for the development of an adverse reaction was chronic use of angiotensin-converting enzyme (ACE) inhibitor and angiotensin-receptor blocker medications prior to surgery. The exact mechanism of this relationship is unknown; those patients may be more susceptible to hypotension, but skin reactions were also noted, suggesting other factors.13
Concern regarding incidence of life-threatening anaphylaxis and a recent national shortage has prompted use of methylene blue as a substitute for isosulfan blue. Methylene blue has been shown to have equivalent identification rates (99.1% vs 98.9%) and lower cost profiles ($31 per patient vs $191 per patient), without the associated risk of severe adverse reactions.14 However, methylene blue, especially when injected in the intradermal plane, may be associated with severe skin reactions in up to 21% of patients. In a series of 24 patients who received methylene blue injections during SLNB for breast cancer, 5 presented with skin lesions ranging from superficial ulceration to necrosis. Skin toxicity is thought to result from oxidizing properties of methylene blue leading to local inflammatory reactions and vasospasm.15 Therefore, intraparenchymal injection is the preferred route when using methylene blue.9
Regardless of the type of blue dye used, intradermal injection can lead to prolonged staining of the skin. In a cohort of 33 patients who underwent SLNB with intradermal injection of 1 mL isosulfan blue, 64% reported blue staining of the skin at the injection site after 6 months, and 41% reported persistent staining after 1 year. None of the women considered it to be a psychological or cosmetic issue; however, it is important to warn patients of this finding.16
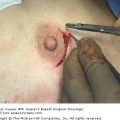
The common use of electrocautery in breast surgical procedures has reduced the risk of bleeding complications, but the incidence of hematoma has been reported in up to 11% of patients.17 Mastectomy patients have a wide surface area of dissection; lumpectomy often requires fracture of breast parenchyma without dissection of avascular planes. Postoperative bleeding complications are associated with increased risk of postoperative wound infection18 and can lead to poor cosmesis and discomfort for the patient. Small hematomas will be absorbed over time, but larger hematomas can expand quickly and may require reoperation for evacuation, irrigation, and closure.6
Use of medications with antiplatelet activity, such as aspirin, other nonsteroidal anti-inflammatories, and herbal medications, in the perioperative period can increase the risk of bleeding complications.6 Intravenous ketorolac has been increasingly used as an adjunct for the acute management of surgical pain; it provides effective pain relief in the postoperative period while reducing the need for narcotics.19 Some surgeons have expressed concern with the use of ketorolac, as it inhibits platelet aggregation and can increase bleeding time. The only data evaluating bleeding complications with the use of ketorolac after breast surgical procedures were reported by Sharma et al, who published a series of 215 patients who underwent transverse rectus abdominis musculocutaneous (TRAM) flap flap reconstruction. The incidence of hematoma in those patients who received ketorolac was 1.5%; this was statistically similar to those who did not.20
Surgical site infections are the third most common nosocomial infections and the most common infection in surgical patients.21 Overall wound infection rates after breast surgery vary between reports, from 3% to 19%. Most wound infections are superficial, occur as a result of skin flora (most commonly Staphylococcus aureus and Streptococcus epidermidis), and can be managed with a short course of antibiotics.6
Patient-related risk factors for the development of wound infections have been widely studied. Age is consistently identified as a risk factor, with older patients more likely to develop postoperative wound infections.18 Obesity has also been associated with increased morbidity and risk of wound infection in patients undergoing breast surgery.1 Patients on chronic steroids preoperatively are at 1.4 times higher risk of surgical site infections (95% CI 1.18-1.63); smokers and diabetic patients are also at higher risk.18 Smoking causes a reduction in tissue blood flow and oxygen tension, and as shown by Sorensen et al, smokers have a 3-fold increased risk of wound infections and 9-fold increase in skin flap necrosis after breast surgery.22,23 A recent review of 260 mastectomy patients showed that any blood glucose value over 150 mg/dL during the perioperative period increased the risk of postoperative wound infection 3-fold (95% CI 1.2-6.2).24 In a review of 425 patients undergoing breast cancer surgery, diabetes was the single most significant factor for the development of wound infection (multivariate OR 4.22, 95% CI 1.10-16.2).22 El-Tamer et al also found the preoperative albumin and hematocrit to be independent predictors of postoperative wound infection, likely related to overall nutritional state and immunologic defense.1
Treatment-related factors have also been correlated with the risk of wound infections in this patient population. As reported by NSQIP data, patients undergoing mastectomy are more likely to have postoperative wound infections when compared with lumpectomy (2.8% vs 1.4%, p = .007); however, other studies have been somewhat inconsistent.1 In patients undergoing SLNB for staging of their breast cancer, wound infection rate was 1.0%, as shown by data from the American College of Surgeons Oncology Group (ACOSOG) Z0010 trial. This is significantly lower than the accepted rates of infection (6%-19%) after axillary dissection (ALND).25 Furthermore, patients who undergo chemotherapy prior to their definitive breast surgery have a 2.8 times higher risk of surgical site infection (95% CI 1.4-5.8).24
The use of preoperative antibiotic coverage has been evaluated in multiple randomized trials; a meta-analysis by Platt et al noted a 38% reduction in wound infection rates with the use of antimicrobial prophylaxis.26 Other studies have reported equivocal results.27,28 As a result, some clinicians have chosen to limit preoperative antibiotics to those patients at high risk for postoperative wound infection and those involving foreign bodies, such as guidewire placement or implant reconstruction.6 Weber et al concluded that the timing of antimicrobial prophylaxis was also a significant risk factor for surgical site infection; lowest rates were seen when antibiotics were administered 30 to 74 minutes before surgery.23
Seromas are accumulations of fluid under the skin resulting from transected lymphatic channels that occur during surgical dissection. They may occur in the dead spaces of lumpectomy cavities, axillary wounds, and the anterior chest wall.6 Histologic evaluation of the fluid shows that it also contains components associated with an acute inflammatory exudate (ie, immune globulins, granulocytes, and leukocytes).29 Seromas are commonly reported complications after breast and axillary surgery, and may be considered side effects rather than complications. The incidence varies significantly throughout the literature (2.5%-72%). Seromas can be managed with percutaneous aspiration, which is required in 10% to 80% of patients, as reported by Pogson et al.30 Aspiration is fairly well tolerated, as the axillary and mastectomy incisions tend to be insensate. Persistent seromas can be a source of significant physical and psychological morbidity for patients, with increased costs, frequent visits, and potential delay of wound healing and initiation of adjuvant therapies.
The data are inconclusive regarding patient factors associated with seroma formation. Lumachi et al reported a higher incidence of seroma in older patients after ALND (60.0 vs 53.8 years, p = .02); however, others found no association.31,32 Increased body mass index (BMI) is also a risk factor for seroma formation, as shown in the same study.31
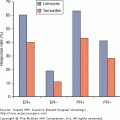
It has been suggested that more extensive surgical dissection is associated with risk of seroma formation. As reported by Kuroi et al in a systematic review, 6 studies comparing breast-conserving therapy with mastectomy have shown equivocal data regarding seroma formation. Immediate reconstruction at time of mastectomy is associated with lower incidence of seroma, while extended radical mastectomy is associated with higher rates of seroma when compared with simple mastectomy.32 Regarding ALND, 4 studies have shown no association with the number of nodes removed and seroma formation.31 However, it has been demonstrated that the use of SLNB has significantly reduced seroma rates when compared with ALND. Guiliano et al reported the presence of a seroma in 1.5% of patients after SLNB, compared with 15% in women who underwent SLNB followed by ALND, despite external drainage in ALND patients.33
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree