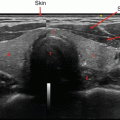
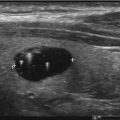
Fig. 12.1
High suspicion thyroid nodule with hypoechoic appearance and microcalcifications
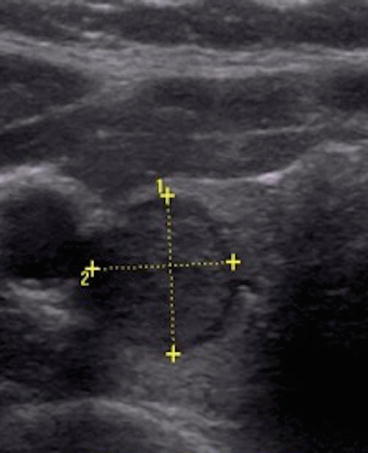
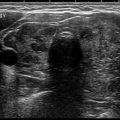
Fig. 12.2
Intermediate suspicion thyroid nodule with hypoechoic appearance and smooth borders
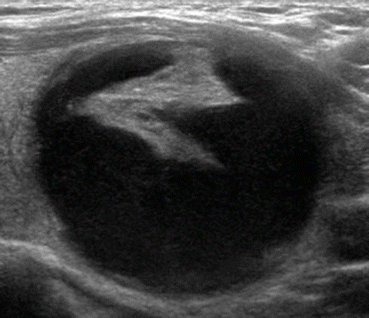
Fig. 12.3
Low suspicion thyroid nodule with partially cystic appearance with an eccentric solid component
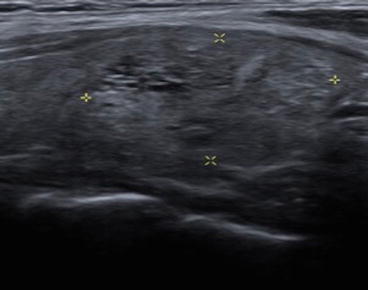
Fig. 12.4
Very low suspicion thyroid nodule with spongiform appearance
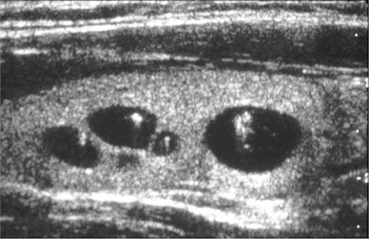
Fig. 12.5
Benign cystic thyroid nodule with comet tail artifact from colloid
For patients with known thyroid cancer or cytology and/or molecular findings suspicious for malignancy, lymph node mapping with ultrasound should be performed of the central and lateral neck compartments to evaluate for lymph node metastases. An FNA should be performed on lymph nodes ≥8 mm with any imaging features that are suspicious for malignancy, which include microcalcifications, cystic component, peripheral vascularity, round shape, and hyper-echogenecity (Fig. 12.6). Thyroglobulin washout of the FNA aspirate should be performed for indeterminate cytology to improve the accuracy of FNA for differentiated thyroid cancer. A thyroglobulin concentration <1 ng/mL is reassuring for benign disease, but optimal values for the identification of malignancy are less clear. A meta-analysis by Pak et al. pooling eight studies evaluating the optimal cutoff value for thyroglobulin washout determined that the optimal cutoff was 32 ng/mL for discriminating between benign and malignant lymph nodes [16]. Cross-sectional imaging with CT is not indicated except for goiters that are suspected to extend into the mediastinum or retropharyngeal space where ultrasound is limited or for locally advanced cancers with posterior extension or associated with bulky lymphadenopathy (Fig. 12.7). In these situations, intravenous contrast is essential to evaluate for invasion into the aerodigestive tract, nerves, and major blood vessels and to facilitate operative planning.
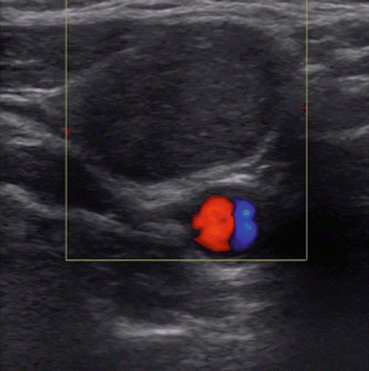
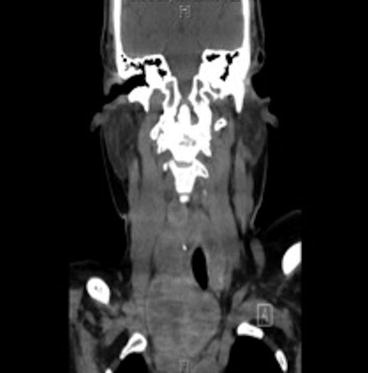

Fig. 12.6
Ultrasound of enlarged hyperechoic lateral compartment lymph node suspicious for metastases

Fig. 12.7
Computed tomography demonstrating substernal thyroid extension
All patients with subjective or objective voice changes, a prior history of anterior cervical or chest surgery, or locally advanced cancer should be evaluated with laryngoscopy to assess vocal cord function . A paralyzed vocal cord significantly increases the risk of surgery and may alter surgical planning. Surgery on the side contralateral to the paralyzed vocal cord should be undertaken with caution because the patient is at increased risk for bilateral vocal cord paralysis and subsequent tracheostomy.
Fine Needle Aspiration/Molecular Testing
The management of thyroid nodules is largely directed by the cytologic findings from FNA; however, these results must be considered in the context of ultrasound and clinical findings. Results from FNA should be reported using the Bethesda classification system (Table 12.1), which recognizes six different categories and provides an estimation of malignancy for each category [17]. The risk of malignancy for each Bethesda category may vary between institutions, such that clinicians should be aware of the risk of malignancy at their own institutions for each cytologic category in order to make the most informed decisions for treatment [18]. Generally, the majority of thyroid nodules have benign (Bethesda category II) cytology (75%), a small proportion have malignant (Bethesda category VI) cytology (2–5%), while the remainder (20–30%) have indeterminate cytology. Indeterminate cytology includes atypia of undetermined significance (AUS)/follicular lesion of undetermined significance (FLUS) (Bethesda category III), follicular neoplasm (FN) (Bethesda category IV), and suspicious for malignancy (SM) (Bethesda category V).
Table 12.1
Bethesda system for reporting thyroid cytology
Diagnostic criteria | Estimated risk of malignancy (%) | |
|---|---|---|
I | Nondiagnostic | 1–4 |
II | Benign | 0–3 |
III | Atypia of undetermined significance of follicular lesion of undetermined significance | 5–15 |
IV | Follicular lesion or suspicious for a follicular lesion | 15–30 |
V | Suspicious for malignancy | 60–75 |
VI | Diagnostic for malignancy | 97–99 |
Molecular testing may be performed for indeterminate nodules to supplement clinical and sonographic findings and provide further risk stratification. One option for molecular testing is the use of a gene expression classifier that analyzes 167 genes from an indeterminate nodule and compares them to the gene signatures of benign and malignant lesions using a proprietary algorithm to classify the nodule as either benign or suspicious. The gene expression classifier test was validated for 265 nodules with indeterminate cytology [19]. The negative predictive value (NPV) for AUS/FLUS and FN was 95% and 94%, respectively, which is similar to benign cytology results; therefore, many argue that it is sufficient to “rule out” malignancy. On the other hand, the NPV for Bethesda V lesions was only 85%, which is thought to be inadequate to exclude malignancy. The positive predictive value (PPV) for suspicious results was only 38% for AUS/FLUS and 37% for FN, making it insufficient to “rule in” malignancy. Another molecular approach is to evaluate the FNA aspirate for a panel of seven gene mutations or rearrangements (including BRAF, RAS, RET/PTC, and PAX8/PPARG translocation) associated with thyroid cancer. A single-center prospective study by Nikiforov et al. [20] analyzed 513 cytologically indeterminate nodes with the seven gene mutation panel and definitive histopathological assessment. The BRAF, RET/PTC, and PAX8/PPARG mutations were associated with 100% risk of malignancy, while the RAS mutation carried an 85% risk of cancer. The nonmalignant RAS-positive nodules were all follicular adenomas. Based on the high likelihood of malignancy, the authors suggested that patients with a positive BRAF, RET/PTC, and/or PAX8/PPARG mutation should undergo definitive thyroid cancer treatment and could avoid a diagnostic lobectomy and two-stage thyroidectomy (lobectomy followed by completion thyroidectomy). However, the low sensitivity for malignancy of the seven gene panel is felt to be inadequate to support surveillance for negative results. More recently, next-generation DNA sequencing for an expanded panel of genes has shown promising results, with a 90% sensitivity and 92% specificity for FN [21] and 91% sensitivity and 92% specificity for AUS/FLUS [22], but this has not yet been validated.
Surgical Management of Papillary Thyroid Cancer
There has been a long-standing controversy regarding the extent of surgery for patients with PTC and whether thyroid lobectomy or total thyroidectomy is superior (Table 12.2). Proponents of thyroid lobectomy argue that PTC is an indolent disease with an excellent prognosis, and the higher risks for recurrent laryngeal nerve injury and hypoparathyroidism associated with thyroidectomy are not justified since there is not a clear survival benefit [23, 24]. In many patients, the need for lifelong thyroid hormone replacement may be avoided with lobectomy. Advocates for total thyroidectomy argue that surgery can be performed safely, and complete resection of the thyroid addresses the risks of multifocal and bilateral disease, facilitates treatment with radioactive iodine, and simplifies surveillance [25]. The 2015 ATA Management Guidelines for Adult Patients with Thyroid Nodules and Differentiated Thyroid Cancer [11] recommend that patients with PTC measuring between 1 and 4 cm may be treated with either thyroid lobectomy or total thyroidectomy. Prior versions of these ATA guidelines [26] had recommended total thyroidectomy for PTC measuring >1 cm. This recommendation was based primarily on a study by Bilimoria et al. [27] using the National Cancer Database (NCDB) that included 52,173 patients with PTC demonstrating a slightly higher 10-year survival for total thyroidectomy vs thyroid lobectomy (98.4% vs 97.1%, respectively, p < 0.05). Several factors that could impact survival, including extrathyroidal extension, completeness of resection, and patient comorbidities, were not available for this study. A more contemporary NCDB study by Adam et al. [28] that included 61,775 patients with PTC accounted for these and other risk factors and found no survival advantage associated with total thyroidectomy compared to thyroid lobectomy for patients with tumors 1–4 cm in size, suggesting that thyroid lobectomy is an effective treatment for low-risk differentiated thyroid cancer. Another study using the Surveillance, Epidemiology, and End Results (SEER) database that included 22,724 patients with PTC also revealed no difference in survival between thyroid lobectomy and total thyroidectomy [29]. Since PTC is frequently multifocal, some studies have reported a lower risk of locoregional recurrence after total thyroidectomy [30]. However, in a retrospective study by Vaisman et al. [31] of 289 patients treated with thyroid lobectomy (n = 72) or total thyroidectomy (n = 217), there was no difference in structural recurrence between the lobectomy and total thyroidectomy groups (4.2% vs 2.3%, respectively; p > 0.05). There were no patients who died from thyroid cancer. Importantly, 88% of patients who recurred were rendered free of disease with additional therapy, suggesting that locoregional recurrence rates are low after lobectomy with proper patient selection and that recurrences can be treated without detriment to survival.
Table 12.2
Extent of surgery: reasons for/against total thyroidectomy for papillary thyroid cancer
For | Against |
|---|---|
Regional or distant metastases | Localized tumor |
Addresses multifocal and/or bilateral disease | Increased risk of RLN injury, hypoparathyroidism |
Facilitates surveillance with Tg and/or imaging | Indolent disease that does not imply a significant risk of mortality, recurrence |
Need for postoperative RAI | Requires lifelong thyroid hormone replacement |
Based on these studies, the 2015 ATA guidelines recommended a more individualized approach to treatment, taking into consideration tumor features, patient characteristics, patient preference, and surgeon experience. The ATA has devised a risk stratification scheme that categorizes patients into low-, intermediate-, and high-risk categories for recurrence that should be incorporated into treatment decisions [11] (Table 12.3). Patients with intermediate- or high-risk tumors (tumors that are >4 cm or with gross extrathyroidal extension, clinically apparent lymph node metastases, distant metastases, aggressive histological variants, and vascular invasion) should undergo a total thyroidectomy to enable radioactive iodine treatment and reduce the risk for recurrence and facilitate surveillance [32]. Regardless of tumor size, patients with a history of radiation exposure to the head and neck or familial thyroid cancer, or with bilateral thyroid nodules, should be considered for total thyroidectomy, since they are at risk for multifocal/bilateral cancer [11, 33]. Patients who have tumors between 1 and 4 cm without intermediate- or high-risk features are at low risk for recurrence and unlikely to need treatment with radioactive iodine; therefore, they may be treated with either thyroid lobectomy or total thyroidectomy. The decision for extent of surgery in these patients should be based on the overall treatment plan ideally formulated by a multidisciplinary team taking into consideration the individual risks/benefits of surgery, overall treatment goals, and patient preference. If thyroid lobectomy is selected, the patient should be informed (and consented) that if high-risk tumor features are identified during surgery (i.e., macroscopic extrathyroidal extension or metastatic lymph nodes), total thyroidectomy may be performed. Similarly, if high-risk tumor features are identified on final pathology, completion thyroidectomy should be performed.
Table 12.3
ATA 2015 risk stratification system
ATA low risk | Papillary thyroid cancer with all of the following: |
– No distant metastases | |
– No tumor invasion into local structures | |
– No aggressive histology (tall cell, columnar, hobnail) | |
– No vascular invasion | |
– Clinical N0 or ≤5 lymph node micrometastases (<0.2 cm) | |
Intrathyroidal encapsulated follicular variant of PTC | |
Intrathyroidal well-differentiated FTC with capsular invasion and no/minimal vascular invasion (≤4 foci) | |
Intrathyroidal papillary microcarcinoma, unifocal or multifocal | |
ATA intermediate risk | Microscopic invasion of tumor into perithyroidal soft tissue |
PTC with vascular invasion | |
Clinical N1 or >5 pathologic lymph node metastases <3 cm | |
Multifocal papillary microcarcinoma with ETE and BRAF mutated | |
Aggressive histology (tall cell, columnar, hobnail) | |
ATA high risk | Macroscopic invasion of tumor into perithyroidal soft tissue |
Incomplete tumor resection | |
Distant metastases | |
Postoperative serum thyroglobulin suggestive of distant metastases | |
Pathologic lymph node metastases ≥3 cm | |
FTC with extensive vascular invasion (>4 foci) |
If a FNA is suspicious for PTC (Bethesda V), there is a 60–75% risk of malignancy. Due to the high risk, these patients should be treated similarly to patients with a FNA diagnostic of malignancy (Bethesda VI), with either thyroid lobectomy or total thyroidectomy. Alternatively, a diagnostic lobectomy with frozen section/touch prep may be performed to determine if the tumor is malignant. If the intraoperative assessment confirms malignancy, a definitive oncological operation may be performed with either lobectomy or total thyroidectomy. If frozen section/touch prep is not definitive for malignancy, the operation should be terminated and the decision for completion thyroidectomy based on final pathology results.
For tumors <1 cm without lymph node metastases or other high-risk features, thyroid lobectomy is recommended, except if there are other indications for total thyroidectomy (history of radiation exposure to the head and neck, history of familial thyroid cancer, or the presence of bilateral thyroid nodules). Alternatively, active surveillance can be considered for these very low-risk tumors. A study from Japan by Ito et al. [34] evaluated nonoperative management among 1235 patients with papillary microcarcinoma with a mean follow-up of 75 months. At 10 years, only 6.8% of patients progressed to develop clinically significant disease, which was defined as an increase in tumor size to 12 mm or development of lymph node metastases. Disease progression at 10 years was observed in 22.5%, 4.5%, and 2.5% of patients who were aged <40, 40–60, and >60 years, respectively. None of the patients in the study developed distant metastases or died of PTC. Nonoperative management of sub-centimeter PTC with surveillance may be considered as an alternative to surgery for those patients at high risk for surgery due to severe comorbidities, those who have limited life expectancy, or those who are of advanced age.
The relationship between surgeon volume and patient outcomes after thyroidectomy has been extensively studied. Published studies have consistently demonstrated that surgeons who perform more thyroidectomies have superior outcomes, on average, with fewer complications and lower costs. Sosa et al. [35] analyzed the effect of surgeon volume on patient outcomes in 5860 patients who underwent total thyroidectomy in Maryland from 1991 to 1996. After adjustment for case mix and hospital volume, surgeon volume of >100 cases was associated with lower complication rates, shorter hospital stay, and lower cost. In a national study of 16,954 patients who underwent total thyroidectomy, Adam et al. [36] looked for a surgeon volume threshold using multivariate modeling and restrictive cubic spines. The study demonstrated that the likelihood of experiencing a complication decreased with increasing surgeon volume until surgeons performed >25 thyroidectomies per year. Complication rates were on average 51% higher when surgery was performed by low-volume surgeons. Based on these results, the authors defined high-volume thyroid surgeons as those who perform >25 total thyroidectomies per year, which has important implications for quality improvement and identification of criteria for referral. In areas where referral to high-volume surgeons is problematic, a surgeon could be identified to perform all the thyroidectomies in order to concentrate surgeon experience and facilitate improved patient outcomes.
A therapeutic central (Level VI) lymph node dissection (CLND) is recommended for patients with PTC who have clinically involved lymph nodes identified on either preoperative imaging or physical examination or intraoperatively. The ATA published a Consensus Statement on the Terminology and Classification of Central Neck Dissection in 2009 to standardize the surgical approach to the central neck [37]. A complete CLND should extirpate all lymph nodes within the central compartment, for which the anatomic boundaries are the hyoid superiorly, innominate artery inferiorly, and carotid arteries laterally (Fig. 12.8). There is no role for “berry picking” of clinically involved lymph nodes, since recurrence after non-compartment-oriented dissection is increased, along with the likelihood of requiring a higher-risk, remedial operation. The role of prophylactic CLND (pCLND) for patients without clinically evident lymph node metastases is controversial. Prophylactic CLND has been suggested to decrease locoregional recurrence and postoperative thyroglobulin levels, provide more accurate staging, and inform radioactive iodine use [38]. However, pCLND has not been shown to be associated with improved survival, and it is associated with an increased chance of temporary recurrent laryngeal nerve injury and hypoparathyroidism [39]. A meta-analysis by Wang et al. [40] of studies evaluating pCLND revealed no difference in long-term complications or recurrence between patients who underwent total thyroidectomy alone or total thyroidectomy with pCLND. There was a trend toward lower recurrence among patients who underwent pCLND; however, 31 patients would have to be treated with pCLND in order to avoid one recurrence, suggesting that any benefit of pCLND would be small and observed only in high-volume surgical practices. The 2015 ATA guidelines for differentiated thyroid cancer recommend that prophylactic CLND be considered for advanced primary tumors (T3 or T4) , or for tumors with clinically involved lateral compartment lymph nodes, and that thyroidectomy without prophylactic CLND dissection is appropriate for low-risk (T1 and T2) tumors [11]. If suspicious lateral compartment lymph nodes are identified on preoperative ultrasound, an FNA is recommended, and if the FNA is positive for malignancy, a lateral compartment lymph node dissection of levels IIa, III, IV, and Vb (Fig. 12.8) should be performed with preservation of the internal jugular vein, sternocleidomastoid muscle, and spinal accessory nerve. The ATA published a Consensus Review and Statement Regarding the Anatomy, Terminology, and Rationale for Lateral Neck Dissection in Differentiated Thyroid Cancer in 2012 [41]. There is no role for prophylactic lateral compartment lymph node dissection for PTC.
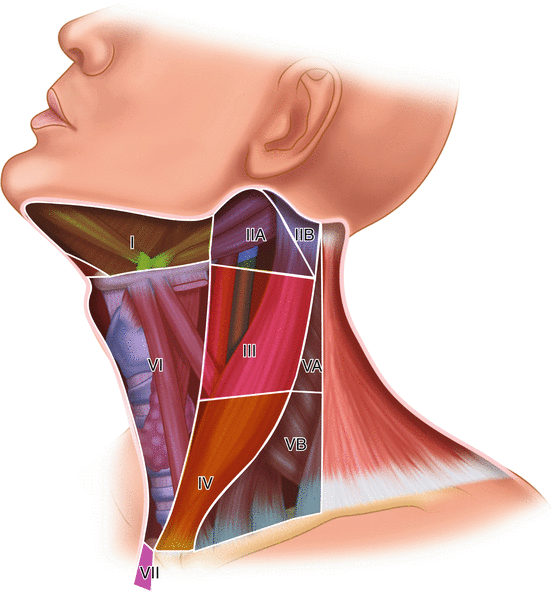

Fig. 12.8
Lymph node compartments in the neck
Indeterminate Nodules
The goal of thyroid surgery for indeterminate thyroid nodules is to establish a definitive diagnosis while minimizing the risk of surgery and, if possible, perform the appropriate oncological operation up front if a diagnosis of malignancy can be established. Patients should be consented for a possible total thyroidectomy if a definitive diagnosis of malignancy is established during surgery and indications for total thyroidectomy are present, including such high-risk tumor features as extrathyroidal extension or lymph node metastases. An up-front total thyroidectomy should be performed for indeterminate lesions if the patient has a prior history of radiation exposure to the thyroid or a history concerning for familial thyroid cancer and if clinically significant nodules are present in the contralateral thyroid lobe. Intraoperative frozen section/touch prep analyses are not routinely recommended for indeterminate nodules as they are seldom helpful. In a study by Chen et al. [42], 120 patients with follicular neoplasms on FNA were treated with thyroid lobectomy with intraoperative frozen section. Frozen section only identified malignancy in 4 patients (3.3%), nondiagnostic in 104 patients (87%), and falsely positive in 6 patients (5%). A cost analysis by Zanocco et al. [43] comparing intraoperative frozen section for follicular neoplasms vs thyroid lobectomy alone revealed that intraoperative frozen section was not cost-effective. Frozen section/touch prep is unable to evaluate for capsular or vascular invasion to diagnose follicular/Hurthle cell cancer so should be reserved for situations when there are clinical features that are highly suspicious for malignancy, such as extrathyroidal extension or lymph node metastases.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree