Abstract
Cancer-induced back pain (CIBP) may cause significant morbidity in patients with metastatic or primary neoplastic spinal disease. Physiatrists practicing in outpatient settings may be the initial providers consulted for the evaluation and management of CIBP. Increasingly, physiatrists are part of the multidisciplinary cancer team working alongside spinal surgeons, radiation oncologists, medical oncologists, pain management and palliative care physicians, as well as physical therapists, occupational therapists, and speech/language pathologists. Proper management of patients with CIBP requires integrating clinical information from all involved specialists which physiatrists are well suited to do. Nuances from the patient’s history and physical examination along with corroborative imaging findings and evidence-based medical and surgical management guidelines need to be appreciated and understood. Ultimately, the physiatrist may play a role in the discovery phase of the patient’s illness, the treatment phase, or the convalescence. As medical and surgical therapies improve and patients enjoy longer disease-free survival, physiatrists will inevitably take on a larger role in cancer patients’ care.
Keywords
Cancer, Pain, Rehabilitation, Spine
Introduction
As physiatrists continue to fill the role of “ gate keepers ” in many outpatient- and hospital-based orthopedic and neurosurgical practices, our surgical colleagues increasingly rely upon us to quickly identify those patients presenting with spinal pain, constitutional symptoms (i.e., red flags), and abnormal imaging findings indicative of metastatic spinal disease or primary spinal tumors. There is a need to further educate physiatrists practicing in these settings on the relevant patient histories, radiologic evaluations, management strategies, and disposition of patients presenting with cancer-induced back pain (CIBP). This chapter will review some of the more commonly seen benign and malignant spinal lesions that physiatrists may encounter in clinical practice by way of brief case histories and expected imaging findings. It is the authors’ hope that the information provided within will lessen any anxieties surrounding these patients, especially related to their imaging and treatment.
History
The term “red flag” was first used by the Clinical Standards Advisory Group in 1994 to identify signs and symptoms associated with cancer or an active malignancy. Red flags including age over 50, night pain, unexplained weight loss, fever or chills, progressive neurologic deficit, bladder dysfunction, saddle anesthesia or any history of cancer, immune suppression, or environmental or toxin exposure should be elicited in the patient’s review of symptoms. Furthermore, the patient’s smoking history and alcohol intake should be documented in the social history. A family history of cancer should be established, particularly in first-degree relatives, such as parents or siblings.
Constitutional or systemic symptoms are often nonspecific and may be encountered in a variety of disease states including certain infectious (i.e., pyogenic discitis or Pott’s disease), hematologic (i.e., sickle cell or Gaucher’s disease), rheumatologic (i.e., rheumatoid arthritis or systemic lupus erythematosus), and neurologic conditions (i.e., multiple sclerosis). It is therefore imperative for physiatrists to screen for them while developing an appropriate differential diagnosis.
Any unexplained weight loss of more than 5% within a 4-week period should alert the physiatrist to the possibility of an underlying malignancy and prompt further investigation. Other nonspecific signs and symptoms associated with malignancy in a patient presenting with spinal pain include feeling generally unwell, malaise, loss of appetite, and decreased exercise tolerance.
The age of the patient presenting with back pain should be kept in mind as vertebral metastases are much more frequent in individuals older than 50 years. Conversely, spinal pain in children, teenagers, and young adults should raise concern for more benign tumors such as osteoid osteomas or osteoblastomas. Certain malignant cancers, however, including sarcomas, lymphomas, and leukemias may be encountered in this patient population, and corroboration from the medical record should be sought.
Further in the review of systems, the physiatrist should query the patient on the quality and character of the pain experienced. Chronic cancer–related pain can arise from visceral or neural structures, but is most commonly related to bony metastases. Pain that does not vary within a 24-h period; pain in the thoracic spine; severe, nonmechanical or night pain; or abdominal pain with a change in bowel habits should alert the physiatrist to the possibility of an underlying malignancy. A change is bowel habits is a red flag for metastases to the cauda equina in the appropriate clinical setting (i.e., invasion of the sacral roots by lymphoma).
Approximately 90% of all spinal (i.e., spinal cord + vertebral body) neoplasms are metastatic in origin and vertebral metastases are already present in 10% of newly diagnosed cancers. Tumors that commonly metastasize to bone can be remembered using the mnemonic “lead kettle” spelled PBKTL (where lead is Pb on the Periodic Table of Elements) for prostate, breast, kidney, thyroid, and lung. The axial skeleton (i.e., ribs, pelvis, and spine) is generally involved earlier than the appendicular skeleton (i.e., long bones) given the persistence of red bone marrow there. In women, breast and lung cancers represent nearly 80% of tumors metastasizing to the skeleton, whereas in males, prostate and lung cancers make up 80% of cancers metastasizing to bone. The other 20% of tumors affecting both sexes include kidney, thyroid, and gastrointestinal (GI) tract cancers.
Historically, metastases to the spine were thought to occur primarily through Batson’s venous plexus, a valveless plexus from the chest and pelvis, allowing malignant cells to enter the vertebral circulation without first passing through the lungs during periods of increased intra-abdominal pressure. Malignant cell survival was favorable in this low-pressure venous system where repeated reversals of blood flow allowed cancer cells to lodge in the vertebral bodies. Alternatively, the “seed and soil” hypothesis was proposed whereby tumors were thought to metastasize through the arterial circulation rather than through venous routes. Other malignancies including lymphomas and multiple myeloma may affect the spine leading to vertebral compression fractures (VCFs) and possibly spinal canal compromise.
In patients with an established cancer diagnosis, the appropriate medical records should be requested to ascertain the behavior of the cancer, its response to treatment, and any future planned medical or surgical interventions. In patients who are receiving or have undergone adjuvant chemotherapy (CTX) or radiation therapy (RTX), the physiatrist should perform a detailed medication reconciliation as various chemotherapeutic agents can cause myalgias and arthralgias (i.e., aromatase inhibitors), which may present as CIBP, in addition to the numbness, tingling, and burning sensations associated with chemotherapy-induced peripheral polyneuropathies. ISODOSE color wash maps can be requested to see how much of an individual’s total radiation dose was given to nearby spinal structures which could predispose to painful VCFs. Any patient with a recent history of malignancy who presents with spinal pain to the office or clinic should raise a high suspicion for metastatic disease to the spine or iatrogenic injury from adjuvant therapies (i.e., RTX).
In reviewing the patient’s past medical history, the physiatrist should also inquire about any history of immune suppression (i.e., HIV/AIDS), conditions requiring prolonged corticosteroid or immune modulating treatment (i.e., organ transplantation), or treatment of childhood cancers that may predispose to secondary cancers as an adult (i.e., leukemia).
Physical Examination
A complete physical examination should be performed with special attention paid to the neuromusculoskeletal examination. Specifically, focal motor deficits, asymmetric muscle stretch reflexes, and sensory loss should be looked for. Depending upon the location of the tumor, the patient may present with prominent upper motor neuron findings such as spastic weakness, hyperreflexia, and neurogenic bowel and bladder dysfunction. In these patients, a digital examination assessing for anal sphincter tone may be indicated. Caution is advised in patients who are neutropenic or severely thrombocytopenic from recent chemotherapy or who have undergone RTX to the pelvis as the rectal mucosa may be friable and prone to bleeding.
Further inspection of the patient can uncover areas of muscle atrophy, skin fibrosis, radiation burns or dermatitis, scars, joint contractures, and other deformities. Restricted spinal range of motion, pelvic muscle imbalance, or scoliosis should be noted if present.
In certain patients, percussion over the spinous processes of the thoracolumbar spine may elicit pain, which should alert the physiatrist to the possibility of a VCF with involvement of the posterior elements. Pain with passive lumbar extension may indicate involvement of the pars interarticularis (i.e., spondylolysis) or the zygapophyseal joints from tumor invasion. Bilateral pars defects, however, with resulting spondylolisthesis, are more commonly seen in younger patients without a history of malignancy.
A positive dural tension test (i.e., seated slump test or Lasegue’s test), normally implying nerve root impingement from a herniated nucleus pulposus (HNP), may also be positive in patients with nerve root sheath tumors (i.e., schwannomas or neurofibromas) as the mass lesion prevents sliding of the nerve root in its dural sleeve from compression against the pedicle. As the physical examination alone cannot differentiate between these two spinal pathologies, the physiatrist must rely upon the appropriate imaging studies to make the diagnosis.
A Lhermitte’s sign may be elicited in patients who are myelopathic or those who have received radiation to the head and neck. It is thought to be caused by transient demyelination of the posterior columns of the spinal cord. Lhermitte’s sign has been described as an electric shock-like sensation travelling down the spine when the examiner flexes the patient’s neck.
Gait and balance testing should be included in the physical examination as loss of proprioception due to an underlying diabetic, carcinomatous, or chemotherapy-induced polyneuropathy can alter patients’ postural stability and predisposition to falls. In patients with severe neoplastic spinal cord compression, one of the earliest presenting symptoms may be gait disturbance, which should prompt the physiatrist to refer for neurosurgical evaluation.
Imaging
As the triaging spine physician, it is imperative that the physiatrist attempt to correlate the patient’s physical examination findings with their imaging studies. If the patient’s physical examination findings do not correlate well with their imaging, then an alternate diagnosis should be considered or a new imaging study requested to help clarify the diagnosis.
Despite recent guidelines recommending against the routine use of spinal radiographs for patients presenting with lower back pain, physiatrists often need to screen new patients with a suspicion or history of malignancy. Plain radiographs are widely available and relatively low cost. X-rays are useful for evaluating large masses and pathologic fractures and detecting the presence of lytic or blastic lesions. Radiographs are, however, considered a relatively insensitive means of screening for asymptomatic metastases. When evaluating for osteolytic lesions from patients with primary lung, GI, renal, or multiple myeloma, for example, 30%–50% of bone mineral loss needs to occur before these lesions become prominent on plain radiographic studies. Other modalities, discussed later in the chapter, are able to detect these lesions earlier.
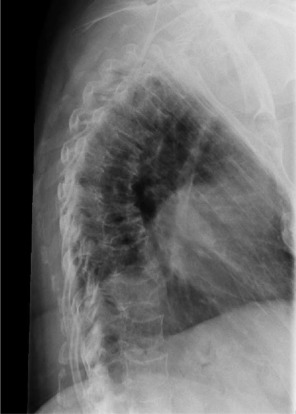
Our first case is of a middle-aged female with no significant past medical history who presents with a breast lump and thoracic back pain with the following radiograph ( Fig. 13.1 ).
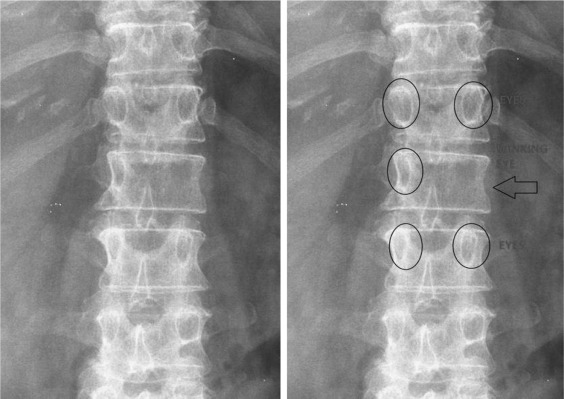
Anteroposterior X-ray of the thoracolumbar spine shows an absent left L1 pedicle with the so-called “winking owl” sign. Early destruction of a pedicle is a classic sign of metastatic disease (differentiating it from multiple myeloma, for example), but in the presence of extensive disease, tissue or laboratory diagnosis is often needed. In anticipation of a metastatic workup, the physiatrist should obtain an MRI scan of the patient’s thoracolumbar spine with and without contrast and discuss the findings with the patient’s primary care physician.
An elderly patient referred by her primary care physician for worsening lower back pain for 6 months with a history of metastatic cancer presents with the belowmentioned MRI ( Fig. 13.2 ). The images reveal multiple areas of abnormal signal involving the vertebral bodies and posterior elements along with prominent degenerative changes and spinal stenosis from L2-3 through L4-5. Incidentally, there is a left laminectomy defect at T11 ( Fig. 13.2D ). The areas of low signal on T1, high signal on T2 (excluding endplate regions), and contrast enhancement (excluding surgical bed) represent bony metastases to the spine with primary lung, prostate, renal, GI, and melanoma high on the differential. This case illustrates the difficulty differentiating tumor involvement from advanced degenerative disc disease given the latter’s high prevalence in outpatient practice. Similar difficulty is encountered when an infectious etiology is considered in the differential diagnosis of patients with widespread degenerative disease; although, pyogenic discitis often crosses the involved disc space, whereas the signal change related to degenerative disc disease does not. Prompt discussion of the MRI findings with the patient’s primary care physician is paramount to avoid further morbidity and mortality.
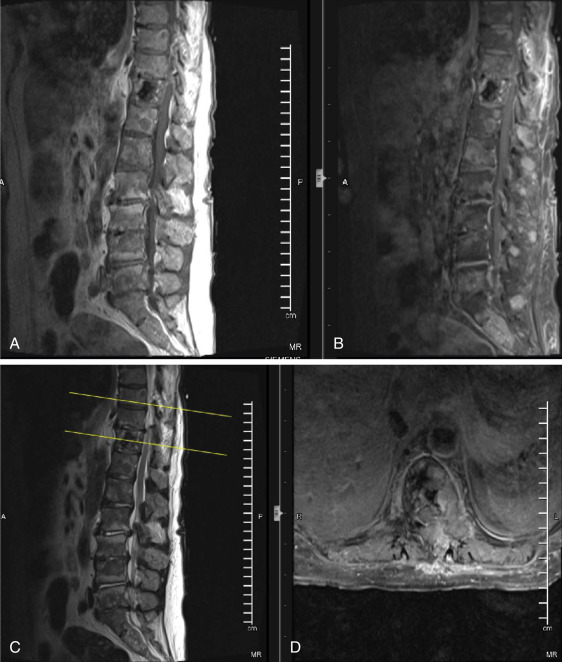
As a brief review of neuroimaging for the physiatrist, normal T1 imaging of bone marrow reveals high signal intensity due to its high fat content. Osseous metastases, on the other hand, have low T1 signal intensity due to remodeling and replacement of normal marrow fat with tumor. On T2-weighted images, metastases appear hyperintense secondary to their elevated water content. After gadolinium contrast administration, there is enhancement secondary to the increased vascularity associated with malignant cells. Of note, gadolinium is a paramagnetic contrast agent that also crosses disrupted blood-brain and blood-nerve barriers. MRI does not show cortical bone very well, as such, bones with low marrow volume (i.e., ribs) are better evaluated with CT scan.
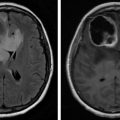
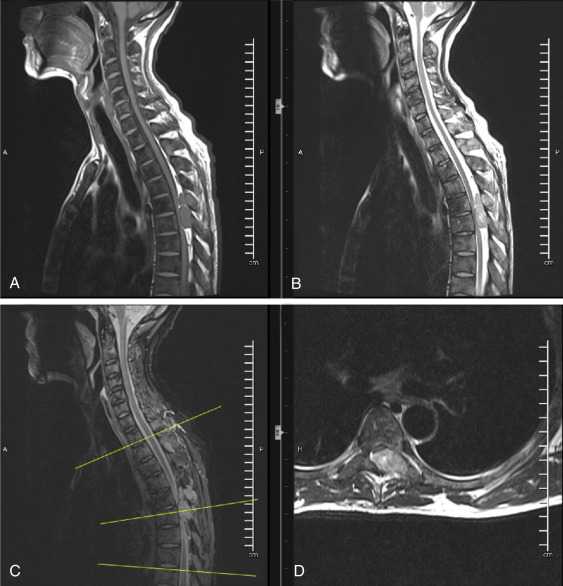
In patients with a history of cancer who present with new neurologic deficits, the physiatrist is obliged to image the entire neural axis from brain to sacrum with and without contrast. Fig. 13.3 is from a middle-aged male with metastatic prostate cancer. MRI findings are significant for diffuse predominantly low signal intensity osseous metastases on all pulse sequences typical for sclerotic metastases. At T5-6, there is high-grade epidural spinal cord compression (ESCC) arising from the left posterior elements. These findings could help explain the patient’s leg weakness and bladder dysfunction.
The physiatrist may encounter the rare but concerning patient presenting with a combination of pain, weakness, sensory changes, gait disturbance, and bowel or bladder dysfunction. These individuals often report a gradual decline in physical functioning and a long history of symptoms prior to a proper diagnosis being made. The MRI in Fig. 13.4 is from a patient with an intramedullary spinal tumor extending from C2-3 to the midpoint of C4. Upon close inspection of the images, one sees low signal on T1 (with areas of high T1 signal consistent with hemorrhage), high signal on T2, and significant contrast enhancement on axial images. Vasogenic edema is seen extending above and below the lesion.
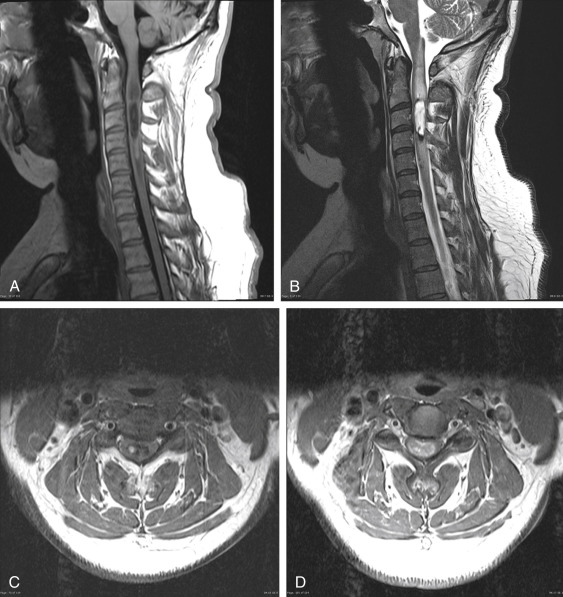
Intramedullary spinal tumors are relatively rare, representing 4%–10% of all central nervous system (CNS) tumors and less than 10% of all pediatric CNS neoplasms. The differential diagnosis includes both glial and nonglial neoplasms, vascular lesions (i.e., cavernomas, arteriovenous fistulas), inflammatory lesions (i.e., multiple sclerosis, transverse myelitis), infectious lesions (i.e., spinal cord abscess), and spinal cord infarctions. As the majority of intramedullary spinal tumors are of glial origin, the differential diagnosis shrinks to either an ependymoma (60%), astrocytoma (33%), or ganglioglioma (1%).
The pathology and MRI characteristics in this case were most consistent with a spinal ependymoma. The physiatrist may recall the association of intramedullary spinal tumors with a history of neurofibromatosis, which may aid in making the correct diagnosis.
In older patients with relapsing and remitting back pain who suffer exacerbations after minimal exertion or trauma, the physiatrist should have a lower trigger for ordering imaging studies. Fig. 13.5 is from an elderly patient whose X-ray reveals multiple thoracic VCFs causing an exaggeration of the thoracic kyphosis. These features are consistent with multiple myeloma; however, metastatic disease (i.e., breast and prostate), lymphoma, and leukemia remain in the differential as contiguous tumor involvement in the spine is common in these cancers.
MRI is a more sensitive tool for detecting focal bone marrow lesions than CT or positron emission tomography (PET) scans. It is also superior when evaluating disease progression and can detect changes in lesions before they become lytic. If there is suspicion of extramedullary disease outside of the spine, whole body PET scanning is recommended as shown in Fig. 13.6 .
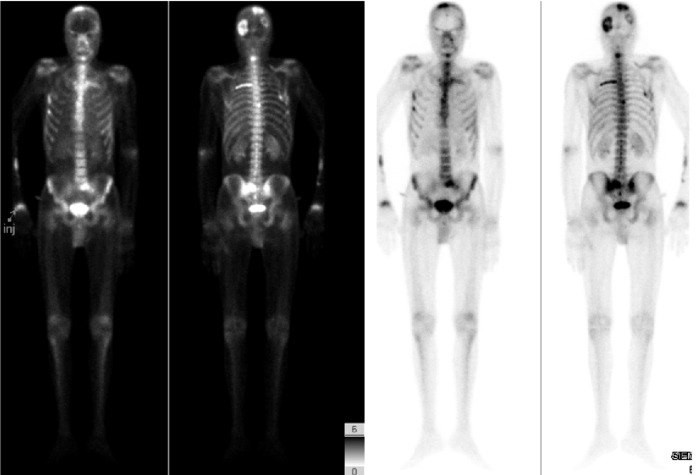
It should be remembered that the distribution of multiple myeloma deposits mirrors that of red marrow in older individuals with the axial skeleton and proximal appendicular skeleton most commonly involved. It is the most common bone neoplasm in adults, often presenting with pathologic fractures (i.e., VCFs, proximal long bone fractures), anemia, and renal failure.
A 50-year-old male with a past medical history of biliary atresia and liver transplantation presents to the office with complaints of chronic lower back pain. Additional history reveals that the patient has been maintained on tacrolimus since 1998 without any episodes of rejection. The MRI scan in Fig. 13.7 was obtained revealing a pathologic crush fracture of L4 with complete replacement of the normal fatty marrow with low T1 signal soft tissue which enhances following contrast administration. At L4-5, there is moderate-to-severe central canal stenosis. An additional pathologic deposit is seen at T12. An old, nonpathologic crush fracture of L1 is also present with normal bone marrow signal.
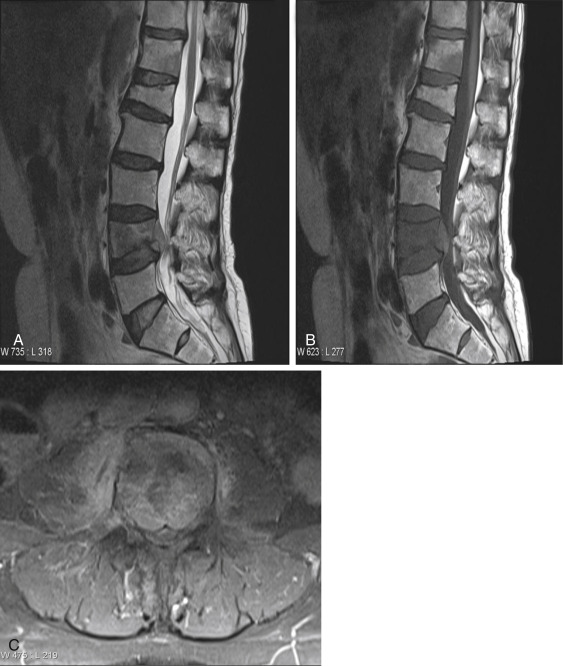
The MRI findings are consistent with lymphoma, but multiple myeloma and metastatic disease are also in the differential. Pathology revealed diffuse large B cell lymphoma. This case illustrates the importance of taking a complete patient history and recognizing that chronic immune suppression may place patients at risk for the development of secondary cancers, which may preferentially affect the spine.
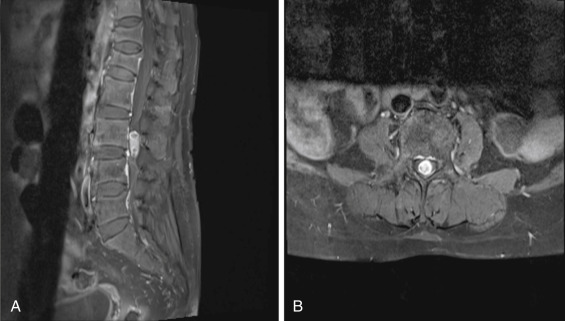
Another challenging problem arises when a patient presents without back pain but with predominantly painful radicular symptoms with a noncontrast-enhanced MRI. Fig. 13.8 is from a patient with a chronic right L4 radiculopathy. The images reveal an intradural, extramedullary mass. The differential at this point includes an HNP, schwannoma, neurofibroma, or a metastasis.
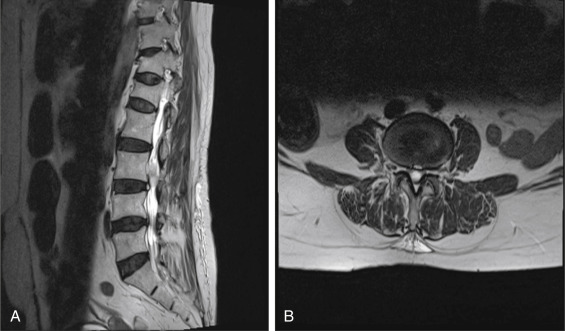
Without the administration of contrast, one may mistake this lesion for an atypical presentation of an HNP given its close proximity to the annular tear represented by the high-intensity zone seen along the posterior border of the L3-4 intervertebral disc.
Following the administration of contrast, however, as seen in Fig. 13.9 , there is avid enhancement with small cystic areas of degeneration within, consistent with a schwannoma. Schwannomas are the most common intradural, extramedullary spinal tumors, accounting for 30% of such lesions. They are most frequently seen in the cervical and lumbar spine, arising from the dorsal sensory nerve root sheaths.