Abstract
Central nervous system (CNS) tumors account for 25% of all cancers in childhood. Owing to multimodal treatment regimens coupled with aggressive supportive care regimens, overall pediatric survival rates continue to rise. The pediatric physiatrist plays an important role in the rehabilitation of a child with CNS cancer from the time of diagnosis through survivorship or end of life. The patient-family-centered approach allows rehabilitation specialists and allied health professionals to coordinate efforts with the child and family to maximize short-term and long-term medical, rehabilitation, and survivorship outcomes.
Keywords
Cancer, CNS, Palliative care, Pediatric, Rehabilitation, Survivorship, Tumor
Introduction
Central nervous system (CNS) tumors account for 25% of all cancers in childhood. Primary central nervous system tumors constitute the second most common tumor and most common solid tumor in children. Tumors of the CNS include both nonmalignant and malignant tumors of the brain and spinal cord and are the leading cause of pediatric cancer–related mortality, as well as significant morbidity. Pediatric tumors of the brain and spinal cord differ from those occurring in adulthood in their relative incidences, histologic features, sites of origin, and responsiveness to therapy. Establishing accurate incidence rates for pediatric CNS tumors (PCNST) is a challenge not only because they are a very heterogeneous group with more than 100 distinct pathologic entities but also because of the variations in cancer registry requirements, classification changes, and improvements in diagnostic techniques over time. Brain metastases in children occur in 1%–20% of cases of pediatric cancer, compared with 20%–40% in adults.
The Central Brain Tumor Registry of the United States (CBTRUS) estimates that approximately 4300 US children are diagnosed yearly. Primary brain tumors are significantly more common and account for 98%–99% of childhood CNS tumors, whereas primary spinal cord tumors account for the remaining 1%–2%. Classification of primary CNS tumors is based on histopathology, although tumor location and extent of spread are important to consider for treatment and prognosis.
This chapter will highlight aspects of CNS cancer in the pediatric population. The chapter will not detail emerging and experimental treatments, although this knowledge is also important when planning a pediatric cancer rehabilitation program.
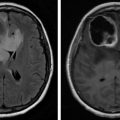
Primary malignant CNS tumors are the second most common childhood malignancies, after hematologic malignancies, and they are the most common pediatric solid organ tumor. Although significant progress has been made with earlier tumor diagnosis and more effective therapies in the treatment of childhood cancers, significant mortality and morbidity are still associated with malignant brain tumors, more so than with leukemia. Incidence rates were initially increasing due to improvement in diagnostics after the introduction of computerized tomography (CT) and magnetic resonance imaging (MRI) in the 1970s and 1980s. This was followed by a decrease in incidence rates in all age groups by approximately 0.2% annually over the past 15 years due to advanced technologies and better treatment.
Overall, childhood cancers are rare, but their importance in the pediatric population is highlighted by the fact that PCNST are now the leading cause of cancer-related death in individuals between 1 and 19 years of age in the United States. Acknowledging the differences between pediatric and adult CNS cancer patients is important due to the important implications on future research, treatment, and prognostic factors. For example, specific histologic types are more common in children than in adults; significant differences exist in the molecular biology and tumor behavior of pediatric and adult glial tumors. A higher percentage of pediatric primary brain tumors are malignant. Owing to multimodal treatment regimens with chemotherapy, radiation therapy, and neurosurgery, coupled with aggressive supportive care regimens, overall pediatric survival rates continue to rise. Cure from cancer, however, is not without its complications. Every organ system can be affected by previous cancer therapy. Therefore, comprehensive long-term follow-up (LTFU) care is essential for this high-risk pediatric population, especially during their developmental maturation and as adult survivors. Pediatric cancer survivorship is associated with neurocognitive, musculoskeletal, and endocrine disorders, as well as a decreased quality of life (QoL) and quality of survivorship. The Children’s Oncology Group (COG, https://www.survivorshipguidelines.org ) has developed evidence-based screening recommendations based on previous therapeutic exposure to be followed in LTFU care of physical late effects for pediatric survivors in survivorship care plans (SCPs). SCPs are used to guide optimal, individualized interdisciplinary care in oncology clinic settings and for transitioning to a primary care physician after young adulthood. SCPs include essential surveillance and screening recommendations to prevent and reduce late effects from exposure to cancer treatment. Based on a 2003 Institute of Medicine report entitled, “ Childhood Cancer Survivorship: Improving Care and Quality of Life ,” approximately 25% of survivors will experience a late effect that is life-threatening or severe. It is essential for pediatric cancer survivors to receive annual, comprehensive LTFU care throughout childhood and into adulthood. LTFU care should be interdisciplinary in approach to ensure that patient-family-centered care is delivered.
Tumors
Most cases of brain tumors in children are sporadic, although 5% are associated with hereditary syndrome with increased risk of various cancers. In neurofibromatosis type 1, neurofibromas predominantly affect the peripheral nervous system. In neurofibromatosis type 2, the associated tumors are schwannomas, meningiomas, and glial hamartomas. Children affected by tuberous sclerosis complex can have CNS tumors, including cortical hamartomas, subcortical glioneuronal hamartomas, subependymal glial nodules, and subependymal giant cell astrocytomas (most common). Cowden disease is associated with dysplastic gangliocytoma of the cerebellum, a benign tumor. Von Hippel-Lindau disease has associated hemangioblastomas of the nervous system and retina. Turcot syndrome is associated with glioblastoma. Li-Fraumeni syndrome is characterized by multiple primary neoplasms in children and young adults, including low-grade astrocytomas, anaplastic astrocytomas, and glioblastomas, medulloblastomas, and primitive neuroectodermal tumor. Nevoid basal-cell carcinoma syndrome is also associated with medulloblastoma. In rhabdoid tumor predisposition syndrome, atypical teratoid rhabdoid tumor is a highly malignant CNS tumor.
Environmental exposure from ionizing radiation, such as previous radiotherapy (due to recurrent diagnostic imaging or treatment), has been implicated in meningiomas, gliomas, and nerve sheath tumors. No convincing evidence has demonstrated a link with trauma, diet, or electromagnetic fields.
Histologically benign tumors can be life-threatening because of the space-occupying effects within the skull, local infiltration, and for some, a risk for malignant transformation. Significant morbidity from both the disease and treatment results in decreased health-related quality of life (HRQoL).
Primary brain tumors arise from one of many different cell types in the CNS. Diagnosis, treatment, and prognosis depend on the tumor cell of origin, pattern of growth, and location.
Brain tumors are categorized as hemispheric tumors, middle fossa tumors, or posterior fossa tumors. Spinal cord tumors are classified as intradural or extradural. Intradural tumors can be intramedullary or, more commonly, extramedullary.
Astrocytomas can arise anywhere in the CNS. Presenting symptoms depend on tumor location, rate of tumor growth, tumor size, and chronologic/developmental age of the child. Low-grade astrocytomas presenting in the hypothalamus may result in diencephalic syndrome, which results in failure to thrive in an emaciated and seemingly euphoric child with little other neurologic findings. Diagnosis is often limited to MRI of the brain or spine. Classification is based on World Health Organization (WHO) histologic grade (grade I and II are low, whereas grade III and IV are high). More than 80% of astrocytomas located in the cerebellum are low grade; malignant tumors are rare in this location. High-grade astrocytomas are locally invasive, extensive, and tend to occur above the tentorium in the cerebrum. Astrocytomas arising in the brainstem may be of any grade, although tumors exclusively involving the pons tend to be high grade, and those outside the pons tend to be low grade.
Prognosis for low-grade astrocytomas is generally favorable. Unfavorable prognostic features include young age, diencephalic syndrome, inability to obtain complete resection, intracranial hypertension at initial presentation, certain oncomutations, and presence of metastases. High-grade astrocytomas carry a poor prognosis.
About two-thirds of childhood ependymomas arise in the posterior fossa. The most common subtype is EPN-PFA. EPN-PFA has a high rate of disease recurrence (33% progression-free survival at 5 years) and low survival rates compared with other subtypes (68% at 5 years). The EPN-PFB subtype is less common but carries a more favorable prognosis: 73% progression-free survival at 5 years and 100% overall survival at 5 years. Spinal cord ependymomas make up about 13% of all ependymomas, 30% of which occur in the cervical spinal cord.
Infratentorial (posterior fossa) ependymomas may present with signs and symptoms of obstructive hydrocephalus (due to obstruction of the fourth ventricle), ataxia, neck pain, or cranial nerve palsies. Supratentorial ependymomas may present with seizures, headaches, or focal neurologic deficits based on location of the tumor. Spinal cord ependymomas may present with back pain, lower extremity weakness, or bowel/bladder dysfunction. MRI and cerebrospinal fluid (CSF) cytology are useful for diagnostic evaluation. Treatment generally involves surgery with or without adjuvant radiation therapy. Chemotherapy does not play a role in treatment, except in some cases for children under 3 years of age.
Embryonal tumors comprise 20%–25% of primary CNS tumors in children, clustered early in life (11 cases per million at the ages under 5, 7 cases per million at ages 5–9, and 3–4 cases per million at ages 10–19). Medulloblastomas comprise the majority of pediatric embryonal tumors; these arise in the posterior fossa and account for 40% of all posterior fossa tumors. Other forms of embryonal tumors each make up 2% (or less) of all childhood brain tumors.
Clinical features depend on tumor location and age at the time of diagnosis. These tumors are fast growing and usually diagnosed within 3 months of symptom onset. In about 80% of children, medulloblastomas arise in the fourth ventricle, with symptoms related to CSF blockage (hydrocephalus): headaches, nausea, vomiting, lethargy, ataxia, nystagmus, or papilledema. A total of 20% of patients will not have symptoms of hydrocephalus initially and may instead present with cerebellar deficits.
Nonmedulloblastoma embryonal tumors generally present rapidly. Supratentorial embryonal tumors can result in focal neurologic deficits, and pineoblastomas can result in Parinaud syndrome. Diagnosis is made by MRI or CT (MRI preferred), with CSF evaluation if safe. Treatment includes surgery with adjuvant chemo- and radiation therapy.
Atypical teratoid/rhabdoid tumor (AT/RT) is a clinically aggressive and rare tumor that most often affects children 3 years of age or younger. One half of AT/RT tumors arise in the posterior fossa. Diagnosis is made with MRI of the brain/spine and CSF evaluation. There is no current standard of treatment; multimodal management with chemo- and radiation therapy, as well as surgery, is under current evaluation. Exact incidence is unknown, as this tumor has been widely recognized only in the last two decades. The Austrian Brain Tumor Registry (1996–2006) has shown that AT/RTs represent the sixth most common malignant brain tumor, with a peak incidence during the first 2 years of life. Clinical presentation typically includes only a short history (days to weeks) due to rapid growth of the tumor. Signs and symptoms are dependent on tumor location, typically the posterior fossa. Patients often present with symptoms related to hydrocephalus, such as vomiting, lethargy, and early morning headaches.
Prognostic factors are not fully delineated at this time. Factors associated with poor outcome include age under 2 years, metastases at time of diagnosis, subtotal resection of tumor, and germline mutation.
Craniopharyngiomas are relatively uncommon, accounting for up to 10% of all intracranial tumors in children. They occur in the region of the pituitary gland, so endocrine function can be affected. Proximity to the optic chiasm can result in visual deficits. Obstructive hydrocephalus due to tumor growth can also occur. Diagnosis is made based on CT or MRI findings. Prognosis is good, with a long-term event-free survival of ∼65% in children, with 5- and 10-year survival rates over 90%.
CNS germ cell tumors (GCTs) are a heterogeneous group that make up 0.5% of all primary brain tumors; 90% of all cases are diagnosed before the patient’s 20th birthday. CNS GCTs arise twice as frequently in the pineal region compared with the suprasellar region of the brain. A total of 5%–10% of patients have involvement of both regions at the time of diagnosis. Tumors arising in the pineal region have a shorter history of signs and symptoms (weeks to months) that include raised intracranial pressure, diplopia, Parinaud syndrome, headache, nausea, and vomiting. Tumors arising in the suprasellar region present subtly with symptoms spanning months to years, including diabetes insipidus, visual deficits, and hormonal symptoms. Diagnosis is made using clinical signs and symptoms, tumor markers (AFP and β-HCG), neuroimaging (MRI brain and spine with gadolinium), and lumbar CSF.
Treatment
The general goal of treatment mirrors that of adults. Surgical resection is often first-line treatment, although location of tumor can restrict success. Often, with low-grade tumors, removal of tumor and observation is sufficient. Adjuvant treatment (chemotherapy and radiation therapy) is usually reserved for higher grade and recurrent tumors. Owing to the well-known debilitating effects of radiation on growth and neurologic development, especially in younger children, chemotherapy may play a larger role than radiation therapy for children, depending on type of tumor.
For low-grade gliomas/astrocytomas, surgical resection is primary treatment, although observation has played a role in some incidentally found cases or asymptomatic masses. Lesions in the cerebellum, cerebrum, optic nerve, hypothalamus, thalamus, brainstem, and spinal cord are generally well resected, but optic nerve lesions may result in blindness and the midline structures may result in more significant neurologic sequelae. About 50% of cases that have less than gross total resection remain progression free at 5–8 years. Chemotherapy for low-grade gliomas include carboplatin with or without vincristine or a combination of thioguanine, procarbazine, lomustine, and vincristine. In optic pathway gliomas, targeted therapy with bevacizumab plus irinotecan can result in visual improvement.
For high-grade gliomas/astrocytomas, the extent of tumor resection correlates with prognosis. Chemotherapy should be considered first as an adjuvant therapy, which can obviate the need for radiation therapy. Commonly used chemotherapy agents include lomustine, vincristine, and temozolomide. For AT/RT tumors, surgical resection with or without adjuvant therapy is also the primary treatment. However, in AT/RT tumors, radiation therapy is more effective and can play a more prominent role. For CNS GCTs, surgical resection with or without adjuvant therapy is the primary treatment protocol for teratomas, although chemoradiation therapy can avoid the need for surgery in germinomas as they are highly sensitive to chemoradiation. Craniopharyngiomas treated with surgery alone or with adjuvant radiation therapy carry an excellent survival rate without the need for chemotherapy. Treatment of ependymomas generally involves surgery with or without adjuvant radiation therapy; chemotherapy does not play a role in treatment except in some cases for children under 3 years of age. In embryonal tumors, treatment consists of surgery with or without adjuvant chemotherapy and radiation therapy; for children age 3 years or younger, radiation therapy is avoided. Chemotherapy regimens include the combination of cisplatin, lomustine, and vincristine or the combination of cisplatin, cyclophosphamide, and vincristine; additionally, etoposide, methotrexate, mafosfamide may also be used for high-risk medulloblastomas.
Outcomes and Prognosis
Overall prognosis depends on the likelihood of a cure. Functional outcomes depend on many factors including type and location of cancer, age at the time of diagnosis, function/deficits at the time of diagnosis, expected neurologic recovery or deterioration, and treatment. Deficits from treatment, including those related to surgery, chemotherapy, and radiation, will vary depending on type of tumor, location, extent of resection, type of chemotherapy, and RT. Intraoperative monitoring with somatosensory evoked potentials and transcranial motor evoked potentials can help predict postoperative deficits. Brain mapping improves outcomes by aiding in surgical planning and reducing surgery-related morbidity. Boys have higher cancer mortality than girls.
More than 50% of PCNST are in the posterior fossa. Following surgery, the incidence of posterior fossa syndrome has been reported as 28%. Children may present with cerebellar mutism, dysarthria, slow speech, stuttering, or nasal-sounding speech. Swallow and feeding may also be impacted. Other findings may include neurobehavioral and emotional problems, as well as decreased initiation of voluntary movements. Factors related to the development of cerebellar mutism may include tumor type, with a higher incidence in children with medulloblastoma, midline location, and brainstem involvement.
Other CNS deficits, as seen in children with brain injury, include dysphagia, auditory impairment, hemiplegia (in up to 21% of survivors), weakness, cognitive impairment, vision deficits, and cranial nerve palsies.
Surgical resection is often recommended for pediatric intramedullary spinal cord tumors and is associated with significant morbidity, including weakness/paralysis, bowel and bladder dysfunction, spinal instability, and spinal deformity.
Short-term side effects of radiation therapy depend on age of the child, location, and dose. Radiation over the spinal cord or the vertebral column can lead to bony effects and radiation myelopathy.
Short-term impairments from chemotherapy include neuropathy, bowel and bladder dysfunction, fatigue, myositis, swallowing difficulties, hearing loss, memory loss, and impairment of executive function.
Late effects are “health problems caused by cancer disease or treatment… [They] can occur during treatment or even decades after treatment is completed.” Over time, the prevalence of late effects increases. The severity of late effects is variable, from chronic conditions to life-threatening health problems. Survivors may not be aware of the risk for late effects and may not have any symptoms at the time of transition to long-term care. It is important that these patients are not lost to follow-up. The risk of late effects can be minimized by long-term care from a provider who has detailed knowledge of the patient’s cancer history and understanding of appropriate screening and surveillance.
Discussion about late effects should be initiated early, delivered in a balanced and patient-centered manner, and include discussion of physical and psychosocial late effects, routine screening schedule, and lifestyle education. Physical late effects can be grouped into various categories: problems with growth and development, organ dysfunction, reproductive health, and risk of secondary cancers. Late effects can be particularly challenging to pediatric cancer survivors, who may still be young and find it difficult to conceptualize future risk when they currently have no symptoms. Some centers provide survivors and their families with educational “portfolios” that include information about late effects, long-term risks, and necessary follow-up care. This can help greatly with transition to LTFU care and patient self-advocacy. Although late effects can be anticipated, and risks minimized, the prevalence and manifestation of these effects can vary, depending on patient factors (age, genetics, lifestyle, comorbidities), follow-up times, and means of risk assessment.
There has been an overall decrease in pediatric cancer mortality; however, compared with matched cohorts, survivors have increased morbidity and mortality beyond their 40s, with females undergoing a steeper age-dependent decline than males. Pediatric cancer survivors have higher rates of late onset health problems than their siblings or the age-matched general population.
Specific late effects that will be discussed in more detail are neurocognitive deficits, hearing loss, endocrine morbidity, reproductive dysfunction, and fatigue. St Jude Lifetime Cohort (SJLIFE) should be mentioned by name as generating significant research regarding outcomes for pediatric cancer survivors.
Platinum-based chemotherapy increases the risk of developing sensorineural hearing loss. This risk is increased in patients who received platinum-based chemotherapy aged less than 5 years and/or those who received high-dose treatment (>400 mg/m 2 ). Hearing loss can also present after a latent period in patients who received cranial irradiation. Survivors associate serious hearing loss with negative impact on education and vocation. Although not statistically significant, survivors affected by hearing loss also report deleterious impact on their social functioning.
Alkylating chemotherapy is associated with ovarian dysfunction. Pediatric cancer survivors are at 13 × higher risk of undergoing premature menopause than their siblings. Infertility as a late effect is associated with reduced QoL in survivors.
Chemotherapy and radiotherapy are both gonadotoxic. Patients may have mild early puberty (due to premature GnRH activation) and then experience premature ovarian failure or azoospermia. In other patients, surgery and irradiation may lead to arrest of pubertal development. Pituitary growth hormone (GH) deficit can be due to brain injury from cancer or from treatment such as surgery, chemotherapy, and/or radiotherapy. GH deficit can be treated with replacement, although skeletal damage is only partially reversible.
Pediatric brain tumor survivors (PBTS) are at risk of academic/intellectual deficits. They are less likely to graduate from high school than their unaffected classmates, which can have a major impact on life trajectory. They have lower achievement in spelling, reading, and arithmetic. Difficulties with reading and arithmetic are associated with chemotherapy and whole brain irradiation. Difficulties with reading and spelling are more likely to be seen in PBTS who were diagnosed with cancer at a younger age. Over time, PBTS will experience age-referenced intellectual and academic decline. Reading deficits in PBTS may be improved with remedial training and emphasis on phonologic skills.
Other areas of neurocognitive deficit in PBTS include reduced attention, processing speed, and executive function. Deficits can be seen in children who have had surgery only, even without chemo- or radiotherapy. Survivors of CNS tumor are at greater risk than pediatric leukemia survivors. Cognitive training programs are safe interventions that carry no adverse effects. The literature has shown that attention is a good target for cognitive remediation. Working memory and mathematics also show potential. Executive function can be more difficult to target for intervention. Exercise is also a low-risk intervention which can have benefits for cognition, as it may help to promote neuroplasticity. The size and/or severity of the cancer insult is not always proportional to the neurocognitive deficits; some neural circuitry can be more resilient than other networks. There is ongoing research looking at modafinil for brain tumor survivors, and metformin for cognition in pediatric populations. Overall, PBTS should be assessed for neurocognitive deficits, if patient, family, caregivers, or educators express concerns. Tools include NIH Toolbox (<2 h to administer) and a brief computer-based battery Cogstate. A multi-approach intervention plan may be of most benefit.
Fatigue is a prevalent late effect in pediatric cancer survivors, which can be very distressing. It is important to address pain management, sleep hygiene, and mood when trying to reduce fatigue. Behavioral strategies (e.g., relaxation and lifestyle changes) and psychosocial interventions (e.g., cognitive behavioral therapy and self-coping strategies) can both be used to manage fatigue. The literature shows that exercise provides moderate improvement in cancer-related fatigue and should be encouraged.
Pain can be vague and nonspecific, especially in pediatric patients; however, it is important to manage to improve QoL. Of note, pain is the most common presenting symptom of spinal cord tumors. When CNS tumors are treated, pain often improves; however, certain types of pain may persist, e.g., neuropathic pain. Nociceptive pain can be managed as per the WHO analgesic ladder, starting with simple analgesics and escalating accordingly. Neuropathic pain may respond to certain anticonvulsant or antidepressant medications. In the pediatric population, attention must be paid to weight-adjusted dosing. If pain is difficult to control, it is beneficial to consult a pain team or pediatric palliative care (PPC) team.
Other symptoms that may need management in pediatric CNS cancer cases include agitation, insomnia, spasms, seizures, vomiting, constipation, dyspnea, and respiratory secretions. Complementary care for pain and symptom management can include music therapy, mind/body therapy (including meditation), dance and movement therapy, yoga, and touch/massage therapy.
Rehabilitation and The Physiatrist’s Role
A total of 500,000 adult survivors of childhood cancer are expected to be living in the United States by 2020. A 5-year relative survival rate for children with cancer of the CNS varies between 20% (glioblastoma) up to 95% (low-grade astrocytoma). Along with increased survival comes the impact of persistent disability, QoL impact, and premature mortality. The interdisciplinary approach to cancer care should include rehabilitation to optimize function, along with support services to include QoL and pain management. The multidisciplinary approach in the rehabilitation of a child with brain and spinal cord cancer includes the child and family, physicians, physical, occupational, and speech therapists, along with psychosocial support and involvement of the school. Goals of rehabilitation for a child with cancer of the brain or spinal cord, like those of brain or spinal cord injury, can include regaining skills that were affected by the tumor or because of treatment, improving and maintaining function to the maximum extent possible, developing new, age-appropriate skills, and optimizing QoL. Motor deficits and mobility impairment, deficits in cognition (memory, thinking, attention, problem-solving, reasoning), ADLs, speech, feeding, and play skills all need to be addressed, as well as management of learning, behavior, and emotional wellbeing.
Various approaches have been reported to address the provision of rehabilitation to cancer patients throughout the continuum from diagnosis through end of life. Dietz defined four categories of rehabilitation intervention: preventive to lessen effects, restorative to return to prior level of function, supportive to accommodate change, and palliative for increasing level of disability and advanced disease. The cancer care trajectory created a framework to integrate palliative, psychosocial, and rehabilitative care across the continuum through cancer-directed treatment, survivorship, and end of life.
Rehabilitation should start once the child is well enough and stabilized in the acute care setting, and it can continue through survivorship or end of life. The specific goals are determined based on individual needs and expected deficits/outcomes and should incorporate the family and child’s plan. The rehabilitation team provides communication and management of expectations at initial consult and throughout provision of services, continued review of the treatment plan, updating of goals and level of service based on medical condition (fatigue, treatment effects, disease progression), and support to family and child.
Pediatric physiatrists play an important role as experts in implementing and coordinating cancer rehabilitation programs via an interdisciplinary team approach. They can optimize the rehabilitation of a cancer patient by identifying and treating neuromusculoskeletal impairments, spasticity, pain, fatigue, bladder/bowel dysfunction, neuro-arousal, and sleep hygiene. Pediatric physiatrists can address barriers in therapy for better participation, coordinate therapies with appropriate dissemination of medical histories to therapists, and define medical therapy precautions and appropriate modalities based on individualized clinical status. In addition, they can prescribe prosthetics, orthotics, and equipment as needed and coordinate an interdisciplinary team approach with a patient- and family-centered focus.
Depending on the needs of the child and other factors, therapy can be provided in the acute hospital setting, an inpatient rehabilitation setting (either acute or subacute), outpatient, or home. School-age children will have access to educationally based rehabilitation services in the home or school setting.
In the period after diagnosis and during early treatment, the rehabilitation specialist can formulate a program to address bed mobility, ADLs, maintenance of strength, and prevention of effects of immobility related to prolonged bed rest or cancer-related motor deficits. Specific attention is paid to pain management, skin integrity, and family education. Based on the treatment plan, functional deficits, and child/family plan, the appropriate venue for ongoing service provision, whether restorative or supportive, can be determined. The child may require further assessment and intervention for deficits related to surgery, chemotherapy, or RT, as well evaluation of the home for modifications, equipment needs, and need for support services.
During outpatient cancer treatment, the physiatrist can aid in anticipating further weakness or deconditioning from ongoing treatment, treat neuropathy, manage cognitive impairments, spasticity, ataxia, and pain, as well as determine the need for equipment, bracing, and educational support. Support of the child and their family should take into consideration their own views of rehabilitation needs.
With advancing delivery models of care for childhood cancer survivors, there will be an increasing need for pediatric physiatry specialists as part of the medical team to use their neuromusculoskeletal expertise and interdisciplinary team skills approach in SCPs. Lifetime surveillance of these high-risk survivors is needed to monitor late effects and their impact on affected organ systems. Pediatric physiatrists are uniquely placed to coordinate comprehensive LTFU with various other pediatric specialists, such as oncologists, cardiologists, gastroenterologists, neurosurgeons, endocrinologists, neurologists, and neuropsychologists.
In children who have entered surveillance, it is necessary to monitor for signs of recurrence and late effects and to provide physician input for appropriate rehabilitation, education, and social services. For progressive or recurrent cancer, review of the palliative care plan of child and family will assist with a shift in rehabilitation goals.
Pediatric rehabilitation specialists play an important role in palliation, especially in progressive or recurrent cancer cases. Goals are set to maintain mobility and function where possible, provide comfort care and pain management, manage new onset conditions from tumor or treatment, review and adjust equipment and home needs, provide anticipatory guidance, optimize fall/injury prevention, and maintain skin integrity.
Rehabilitation treatment modalities can be applied to pediatric CNS cancer patients. Exercise interventions have been studied in adult cancer patients. There is limited literature on exercise interventions in the pediatric oncology population, most of which is conducted with acute lymphoblastic leukemia patients. A systematic review found exercise to be feasible with benefits in managing disease and treatment-related side effects of strength, fatigue, and QoL. Small studies and case reports for treatment of deficits in children with PCNST include constraint-induced movement therapy (CIMT) and treadmill training. A pilot study of CIMT in children with hemiplegia resulting from brain tumors, 1–10 years out from diagnosis, showed improved quality and use of the affected upper extremity, while maintaining HRQoL. Treadmill training can be incorporated into a rehabilitation program after resection of spinal cord tumors.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree