30.1
Introduction
The cells of bone influence its structure by means of four processes: growth, repair, modeling, and remodeling, the last being the basis of bone tissue turnover in the adult skeleton. The purposes of growth and repair are obvious. Modeling serves to adapt bones to changes in mechanical loading , and remodeling serves to thicken trabeculae in the growing skeleton , processes that are most effective during adolescence . But why does a tissue that can survive for thousands of years after death need to be maintained by periodic replacement during life? Most of those interested in bone, whether as physicians, as clinical investigators, or as basic scientists, show remarkably little interest in this fundamental question. Many articles and book chapters discuss the regulation of bone remodeling, but regulation, at least in the physiologic sense, implies a target . The target value of any regulatory process in biology has been optimized by natural selection. Mechanisms have evolved which ensure that deviations from the target are detected and that corrective measures to restore the target value are carried out. In this sense, body temperature, extracellular fluid osmolality, tissue oxygen tension, and countless other physiologic quantities are regulated, but the mechanisms of regulation could not be determined until the existence of the target had been recognized and its precise nature defined. Is there a target for bone remodeling or for some characteristic of bone that is influenced by remodeling?
The piecemeal, quantal nature of bone remodeling is well known. The process is carried out by temporary anatomic structures known as basic multicellular units (BMUs) , which excavate and replace tunnels through cortical bone (osteonal remodeling) or trenches across the surface of cancellous bone (hemiosteonal remodeling). Each BMU includes two teams of executive cells (osteoclasts and osteoblasts), supported by blood vessels, nerves, and loose connective tissue. The life span of the BMU is measured in months, but the life span of osteoblasts while they are making bone is measured in weeks, and the life span of osteoclast nuclei is measured in days. During progression of the BMU through or across the surface of bone, the spatial and temporal relationships between its components are maintained by the continued growth of the central capillary in cortical bone , and extension of the remodeling compartment in cancellous bone , together with recruitment of new cells . These cells, like the formed elements of the blood, originate from stem cells in the bone marrow except that in the peripheral skeleton osteoblasts are derived from local precursors . For blood cells, as for other short-lived cells, control of cell production and survival is more important than control of differentiated cell function; although the details are less clear, the same applies also to bone cells .
Each type of blood cell is normally produced at a basal rate that is sufficient for ordinary purposes but that can be increased when needed . For each cell type, the circumstances under which demand is increased are well known, and are related to the function of the particular cell, although the cell types differ with respect to the time scale of this response, its specificity, the relative importance of reactive and anticipatory homeostasis , and the extent to which the control mechanisms have been elucidated. The importance of these relationships between supply and demand, and between demand and function, applies also to bone cells. For osteoblasts in the adult nongrowing skeleton, the demand is created by bone resorption, since the function of osteoblasts is to replace the bone removed by osteoclasts. However, the circumstances that create a demand for osteoclasts are much less well defined, since these circumstances are dictated by the purposes of bone remodeling. Indeed, the questions “What are the purposes of bone remodeling and how are they achieved?” are essentially equivalent to the questions “Where and when are osteoclasts needed, and how is this need recognized and satisfied?” The answers to these questions are different in different types of bone and in different regions of the skeleton.
30.2
Skeletal heterogeneity
30.2.1
Structure and function
The structural differences between cortical bone, in which porosity and surface-to-volume ratio are low, and cancellous bone, in which these geometric quantities are high , are now widely recognized. All intermediate values for these quantities can occur, but they are infrequent, implying that transitional structures tend to be temporary and short-lived . Less often noted are the differences between the axial and appendicular subdivisions of the skeleton ( Table 30.1 ); the pelvis, defined anatomically as appendicular, behaves functionally as part of the axial skeleton, so that it is more accurate to contrast central with peripheral regions. This distinction is important because the different functions of the skeleton are divided differently between the central and peripheral components. The primary function is load-bearing to support posture, permit movement (including locomotion), and provide protection for the soft tissues. Subsidiary functions are to participate in mineral homeostasis and to provide a favorable microenvironment for hematopoiesis. For convenience the former functions will be referred to as “mechanical” and the latter as “metabolic” .
| Feature | Central | Peripheral |
|---|---|---|
| Main bone tissue | Cancellous | Cortical |
| Main soft tissue | Viscera | Muscle |
| Main joint type | Various | Synovial |
| Cortices | Thin | Thick |
| Marrow | Hematopoietic | Fatty |
| Turnover | High | Low |
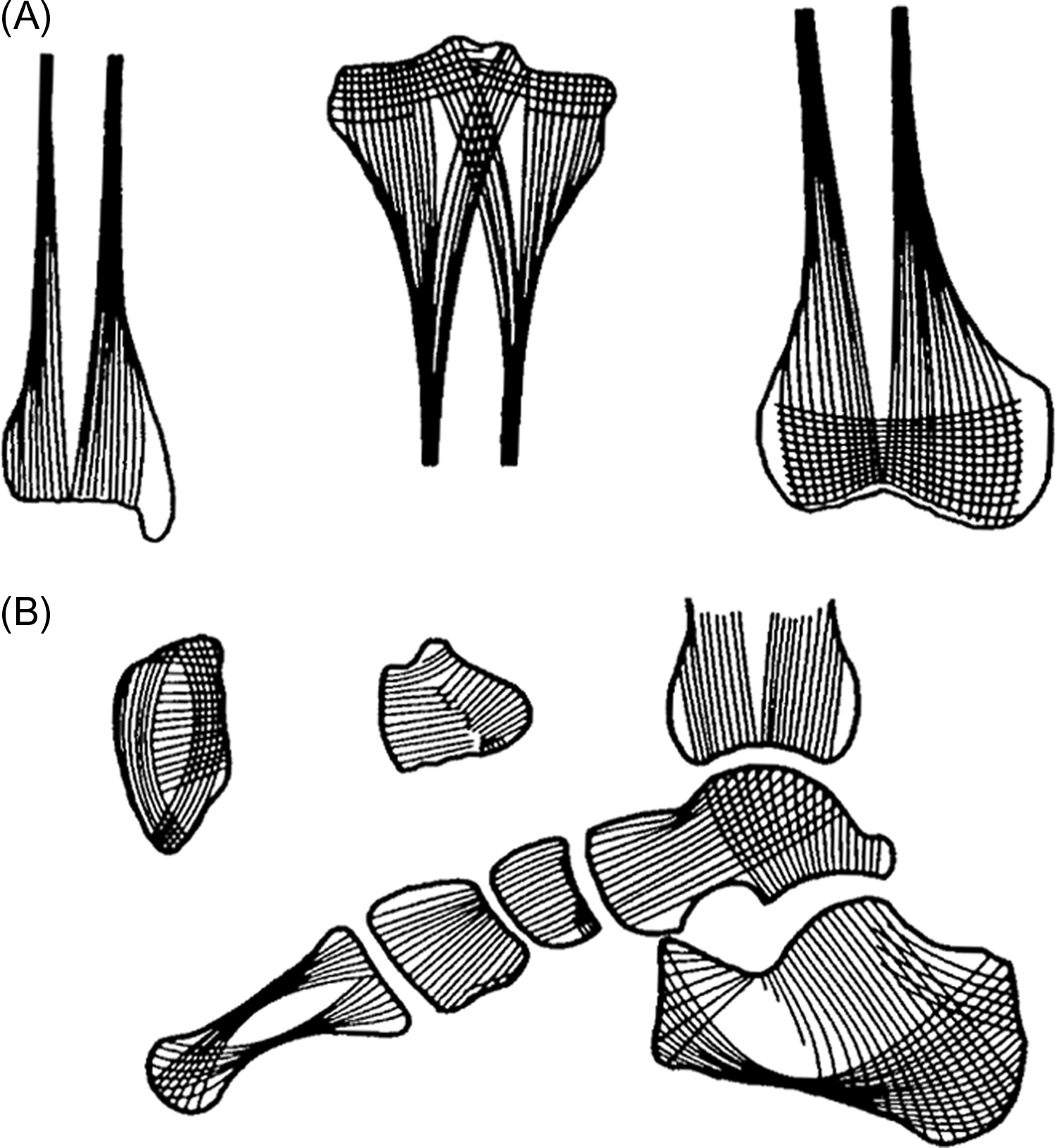
It is commonly believed that the mechanical functions are carried out mainly by cortical bone and the metabolic functions mainly by cancellous bone, regardless of their central or peripheral locations. In fact, the functions of the peripheral skeleton, cancellous as well as cortical, are mainly mechanical, whereas the central skeleton, cortical as well as cancellous, in addition to its mechanical function, participates to a much greater extent in the metabolic functions of bone. This revision in functional attribution is most striking for peripheral cancellous bone, such as in the metaphyses of the long bones . As is evident from the orientation of the trabeculae ( Fig. 30.1A ), metaphyseal cancellous bone transmits loads from the joint surfaces to diaphyseal cortical bone. Indeed, the metaphyses are flared in shape precisely to make such load transmission possible. Similar functional and architectural considerations apply to the cancellous bone in the small bones of the hands and feet ( Fig. 30.1B ). As will subsequently be discussed in detail, there is no evidence that such peripheral cancellous bone participates to a significant extent in the metabolic functions of the skeleton, whether related to mineral homeostasis or to hematopoiesis.
30.3
Remodeling and turnover
The frequent assertion that cancellous bone has higher turnover than cortical bone is usually supported by comparing central cancellous with peripheral cortical bone, but this is to confuse the geometrical and biological factors that influence turnover . The remodeling process occurs only on bone surfaces, and the intensity of remodeling is expressed by the activation frequency, which is the reciprocal of the average time interval between the initiation of consecutive cycles of remodeling at the same surface location, referred to as the regeneration period . Turnover refers to volume replacement, which depends not only on the surface-defined activation frequency but on the surface-to-volume ratio. This geometrical property is about four to five times higher in typical cancellous bone than in typical cortical bone . Consequently, the former could have higher turnover despite a lower intensity of remodeling. Systematic site-specific measurements of turnover in the human skeleton are available only for the rib and for the ilium. In the latter, activation frequency is similar on the cancellous, endocortical, and intracortical subdivisions of the endosteal envelope , so that the difference in turnover between cortical and cancellous bone at this site depends entirely on the difference in surface-to-volume ratio. Turnover depends also on distance from the surface; iliac interstitial bone has much lower turnover than inner cortical bone close to the marrow . Unfortunately, the ilium, although probably representative of the central skeleton in general, and of the vertebral bodies in particular , is quite unrepresentative of the peripheral skeleton .
In peripheral cortical bone, turnover is lower by about half than in the ribs (around 2 vs 4%/year), based on a variety of indirect methods . For peripheral cancellous bone, estimates of turnover are based on fewer data, but they suggest that the central-peripheral difference is greater than for cortical bone. During the treatment of osteomalacia the increase in cancellous bone mineral was about 35% in the ilium, measured histologically, but only 1%–2% in the distal radius, measured by single photon absorptiometry . On the reasonable assumption that unmineralized osteoid tissue accumulates during the evolution of osteomalacia in proportion to the initial rate of turnover, this rate in the cancellous bone of the distal radius is normally only about 2%/year. This is similar to the estimate for peripheral cortical bone; since the surface-to-volume ratio would be higher in cancellous bone, the activation frequency would be even lower than on the intracortical surfaces. Direct measurements of turnover in the beagle confirm a much lower value for peripheral than for central cancellous bone, even though the absolute values for both were higher than in human subjects .
30.4
Relationship to marrow composition
In the embryo, hematopoietic marrow appears first in the yolk sac and subsequently migrates to the liver and spleen, and then to the marrow cavities. At birth, hematopoiesis is active in cancellous bone throughout the skeleton but has virtually ceased at extramedullary sites . During growth, there is gradual conversion of red to yellow marrow, a process that begins in the distal extremities and proceeds centripetally. By 25 years of age, hematopoiesis has disappeared from the peripheral skeleton, except to a limited extent in the upper femora . Macroscopically visible hematopoiesis continues in the central skeleton throughout life, although there is a gradual increase in the number of fat cells at the expense of hematopoietic cells . At any age, a sustained increase in demand can lead to reappearance of hematopoiesis in the extremities . Whether this results from reactivation of dormant local stem cells or from recolonization of fatty marrow by circulating stem cells is not known, but the latter seems more likely since it is now certain that hematopoietic stem cells do circulate .
The data presented, although incomplete, indicate that in the adult human skeleton central cancellous bone has persistent hematopoiesis and high bone turnover (except for interstitial bone), whereas peripheral cancellous bone has absent hematopoiesis and low bone turnover ( Table 30.2 ). Furthermore, based on external radionuclide counting, there is a close correlation between the extent of hematopoiesis and bone blood flow . When different bones sampled at autopsy were compared, there was a good relationship between the proportion of cancellous bone surface in contact with hematopoietic cells and the proportion engaged in bone remodeling . In adult beagles, there is an even more striking correspondence between marrow composition and bone remodeling ( Table 30.3 ). Adjacent to red marrow, there is a 15% higher mineral apposition rate and an almost 10-fold higher bone formation rate than adjacent to yellow marrow, with corresponding differences in the uptake of plutonium . If there are no resident hematopoietic stem cells in yellow marrow, all osteoclasts in the peripheral skeleton, cancellous as well as cortical, must be derived from circulating mononuclear precursor cells . In the central skeleton also, participation of the local microcirculation has now been established for normal bone remodeling . In pathologic bone resorption, as in neoplastic bone disease or osteoprotegerin deficiency , osteoclast precursors produced in much larger numbers than needed might be able to migrate directly to the bone surface.
| Feature | Red marrow | Yellow marrow |
|---|---|---|
| Bone type | Metabolic | Mechanical |
| Location | Central | Peripheral |
| Main functions | Calcium homeostasis | Transmit loads |
| Support hematopoiesis | Absorb energy | |
| Cellularity | High | Low |
| Blood flow | High | Low |
| Turnover | High | Low |
| Site | Marrow | MAR a (μm/day) | BFR b (%/year) |
|---|---|---|---|
| Lumbar vertebra | Red | 1.29±0.10 | 106±9 |
| Proximal humerus | Red | 1.23±0.10 | 89±18 |
| Pelvis | Red | 1.26±0.10 | 83±25 |
| Proximal ulna | Yellow | 0.90±0.06 | 13±6 |
| Distal ulna | Yellow | 0.97±0.07 | 7±3 |
a Mineral apposition rate: n =8.
The relationship between marrow composition and remodeling can be disturbed in pathologic conditions. For example, after ovariectomy, bone turnover and amount of fat in the marrow both increase . No relationship between marrow composition and bone remodeling was found in a single patient with osteoporosis who died from an unrelated cause after administration of tetracycline labels in preparation for bone biopsy . The relationship is also disturbed by proximity to synovial joints; turnover is higher within 1 mm of the articular surface than at more distant locations . Nevertheless, the spatial association between hematopoiesis and active remodeling appears to be characteristic of the healthy skeleton. To most observers, this is simply the expected consequence of the presence or absence of precursor cells in close proximity to the bone surface, but this is a superficial view, since circulating osteoclast precursors can be made available anywhere in the skeleton. Does cancellous bone need to turn over so much faster in some locations than in others, and if so, why?
30.5
The purposes of bone remodeling
There is probably no physiologic function other than bone remodeling that has attracted so much study in the face of so much uncertainty about why it occurs. Many in the field act as if they believed that the only purpose of remodeling was to cause osteoporosis and thus provide employment for scientists and business opportunities for the pharmaceutical industry! In the analysis of this problem, it seems reasonable to make two assumptions. First, periodic replacement of bone serves to maintain its ability to carry out its functions, as previously summarized. Second, since the most obvious difference between the old bone removed and the new bone put in its place is in their ages, excessive age of bone in some way compromises its functional capacity. Bone age must be carefully distinguished from subject age. Different regions of bone have widely different ages, since some bone was made yesterday and some was made decades ago, but mean bone age is the reciprocal of the mean rate of turnover ; if turnover increases with subject age for any reason, mean bone age will be lower in an older than in a younger person. Only at sites where turnover is extremely low such as the auditory ossicles or deep interstitial cancellous bone does bone age necessarily increase with chronological age.
As bone gets older, its true density increases as secondary mineralization progresses and water is displaced; consequently, it becomes more brittle . There are also changes in matrix constituents such as accumulation of products of advanced glycation and increased cross-linking of collagen . Osteocytes have a finite life span, eventually dying by apoptosis leaving an apparently empty lacuna that may eventually become occluded by mineralized debris . As will later be discussed in more detail, fatigue microcracks increase in number with bone age and are spatially associated with missing osteocytes . The adverse effects of increased bone age have been studied mainly in cortical bone, but in central cancellous bone regions of bone of widely different ages are in close proximity . The mean age of surface bone close to the marrow varies from about 0.5 to 4 years, but mean age increases progressively with increasing distance from the surface, and beyond 75 μm the bone is essentially isolated from surface remodeling so that its age is close to the age of the subject . Differences between interstitial and surface bone, due entirely to the difference in age, are listed in Table 30.4 . The remodeling of interstitial cancellous bone carries a high risk for trabecular plate perforation so that at this location excessive bone age is the price that has to be paid for the preservation of cancellous architecture .
| Features | Surface (superficial) | Interstitial (deep) |
|---|---|---|
| Turnover | High | Low |
| Age | Low | High |
| Density | Low | High a |
| Microdamage | Low | High b |
| Osteocytes c | ||
| Density | High | Low |
| Effect of age | No change | Fall |
| Determinant | Initial density | Life span |
b By analogy with cortical bone (Qiu et al., 2005) .
The salutary effects of remodeling in preventing excessive aging of bone will differ in different regions, depending on which effects of age are more important. Since the primary function of bone is mechanical, the primary purpose of remodeling of bone is to maintain its load-bearing capacity. This is accomplished both by preventing the adverse effects of excessive bone age at the microscopic and submicroscopic levels, and by repairing damage after it occurs. The role of remodeling in maintaining the metabolic functions of bone will be considered later, but first the remodeling apparatus and how it behaves over time must be examined in greater detail.
30.6
The life history of a basic multicellular unit
The stages of a BMU are commonly depicted as quiescence, activation, resorption, reversal, formation, and back to quiescence . These terms refer to successive states of the bone surface at a single location; it is the surface that becomes activated. This simple down and up model has been useful in describing temporal relationships and in analyzing the cellular basis of bone loss, but it conceals the three-dimensional reality, already briefly mentioned, that the BMU moves through tissue space, so that it has to begin somewhere and end somewhere else . The beginning of a new BMU, termed origination , occurs in response to the identification of a target—a region of bone that needs to be replaced . The need is recognized by osteocytes, which communicate in some way with the cells that line the nearest bone surface, which in turn communicate with the nearest small blood vessel. In cortical bone the vessel of origin is the central capillary of a Haversian or Volkmann canal from which a new capillary grows ; neoangiogenesis is an essential component of bone remodeling. The new capillary passes between the lining cells, which have digested the thin layer of unmineralized matrix beneath them and then retracted , and progresses through the bone in the direction of the target, in the wake of the cutting cone of new osteoclasts . The relationship between the capillary and the osteoclasts is symbiotic: the capillary cannot advance until room is made for it by resorption, and the osteoclasts cannot continue to advance unless their dying nuclei are replaced by the diapedesis of monocytic osteoclast precursors from the capillary, which requires the right area code to be turned on . In cancellous bone, the new capillary sprout penetrates, in the same manner as in cortical bone, between lining cells, which then form a canopy over the new remodeling site, now in direct contact with a temporary extension of the circulation . The new structure, termed the bone remodeling compartment, represents a form of vasculogenic mimicry .
In order for the new BMU to reach its target, it excavates a tunnel through cortical bone—osteonal remodeling—or a trench across the surface of cancellous bone—hemiosteonal remodeling . Progression of the BMU requires continued access to the circulation and arrival of new osteoclast precursor cells and their replacement from the bone marrow . Directional information must somehow be provided by the osteocytic lacunar–canalicular system; the limited available knowledge will be summarized later. During longitudinal progression of the BMU, successive transverse cycles of remodeling are generated, each new cycle slightly out of step with the one before . The total number of such cycles per unit time is the activation frequency, usually reported for a specific surface or region, but which conceptually can be aggregated for the whole skeleton and estimated rather crudely by biochemical indices of bone turnover. After the target has been reached and replaced by new bone, the BMU will continue to progress for some distance beyond the target because it has acquired some biological momentum, but will eventually come to a stop. Such posttargeted remodeling has been described as “redundant,” “surplus,” “spare,” “nontargeted,” or “stochastic.” Each of these terms has some merit, but “spare” is probably the least inaccurate. Origination is such an intricate process that it could not occur by chance . All remodeling is either targeted, requiring a new BMU, or posttargeted, requiring progression of an existing BMU; the distinction between them will be discussed in greater detail in subsequent sections.
30.7
Fatigue damage and mechanical competence
All structural materials that undergo repetitive cyclical loading are subject to fatigue, a phenomenon that has been most extensively studied in fabricated materials such as steel . After a certain number of load cycles, tiny cracks appear that are detectable at first at the ultramicroscopic level, but were probably preceded by damage at the submicroscopic and molecular levels. If cyclical loading continues, the cracks extend and accumulate into microscopic and then macroscopic damage and eventually into overt structural failure. The essence of fatigue is that in each cycle, the load-induced strain (relative deformation) is far below the instantaneous breaking strain of the intact material. Biological materials such as bone also undergo fatigue damage but differ from synthetic materials in their capacity for self-repair . The occurrence of fatigue damage has been demonstrated unequivocally in cortical bone , and there is compelling evidence that experimentally induced fatigue damage in cortical bone induces repair by remodeling, so that the damaged bone is removed and replaced by new undamaged bone . It is reasonable to assume that the same applies to load-bearing cancellous bone, which also develops fatigue damage with repetitive cyclical loading . Various degrees of microdamage can be identified in human cancellous bone , including microcracks that closely resemble those observed in cortical bone . Unlike those in femoral cortical bone such cracks do not increase significantly with age in the vertebral body and do not increase until after age 60 in the femoral head , so that for the most part they must be repaired by remodeling. However, it is not certain that such lesions are due to fatigue, since identical lesions can be produced experimentally by compression . Microfractures in cancellous bone heal by callus formation rather than by remodeling , and although often called fatigue fractures, most of them (at least in the vertebrae) can be explained, not by fatigue, but by instantaneous overload, leading to failure by buckling .
Evidently, a major function of remodeling is to provide a means for replacing load-bearing bone that has undergone fatigue microdamage; indeed, it is quite possible that all BMU origination events in the peripheral skeleton are triggered by microdamage , and that this mechanism has evolved to allow large long-lived vertebrates to maintain a light skeleton . But repair of microdamage may not be the only way in which remodeling maintains the mechanical competence of bone. The similarity between different members of the same species in the spatial distribution of remodeling activity at different skeletal sites is difficult to explain by a mechanism that is purely reparative. One of the most striking aspects of such remodeling maps is their bilateral symmetry, such that cross-sections at the same level of bones on opposite sides of the body are virtually mirror images of one another . It seems unlikely that such consistent symmetry could be the expression of fatigue damage repair , but it might be an expression of fatigue damage prevention .
Because of bilateral symmetry in local bone geometry and mass, there will be bilateral symmetry in the local strains engendered by mechanical loading. The relationship between strain and remodeling rate is “U” shaped ; increasing strain is accompanied by increased remodeling before the occurrence of strain-induced damage , presumably by prolonging the posttargeted progression of existing BMUs. For material of the same mechanical properties, the major determinants of fatigue damage are the number of load cycles and the average change in strain in each cycle, and for the same level of physical activity, the major determinant of the number of load cycles is the age of the structure. The customary pattern and intensity of physical activity are species specific and so are genetically determined . Consequently, it seems possible that the remodeling map is the expression of a genetic program to prevent bone age from exceeding some critical level, a level that is different in different regions of the skeleton . This would be consistent with the notion that remodeling evolved as a means to prolong the fatigue life of bone .
The contrast between the prevention of fatigue and other forms of damage by keeping bone age below some critical value and the repair of such damage by removal of the bone involved is analogous to the contrast between anticipatory and reactive homeostasis , except that the basis of the anticipation is genetic rather than physiologic. More specifically, it exemplifies the distinction between targeted and posttargeted remodeling, a distinction that establishes an order of priority for different remodeling projects. There is a wide range of turnover rates consistent with skeletal health , and the low rates that occur in hypothyroidism and hypoparathyroidism do not appear to increase fracture risk. Presumably, the reason is that spare remodeling to prevent excessive bone age provides a substantial margin of safety. Consequently, curtailing the posttargeted progression of a particular BMU is unlikely to have any harmful effects. However, targeted remodeling to remove fatigued bone before the damage escalates from microscopic to macroscopic, which requires new BMU origination, must be carried out promptly, or else it will fail in its purpose. The existence of such a temporal hierarchy has an important impact on the therapeutic reduction of bone turnover, a point that will subsequently be discussed in more detail.
The mechanism of targeted remodeling in load-bearing bone is now much clearer. The only cell that is in the right location to detect microscopic damage is the osteocyte. This cell can be activated by mechanically induced strain to increase the synthesis of various proteins and prostaglandins, nitrous oxide, and no doubt other signaling molecules , effects that probably mediate the addition of bone to the nearest bone surface during growth , but microdamage repair requires the origination of a new BMU as previously described. In the adult rat ulna, there is a close relationship, both spatial and temporal, between experimentally induced fatigue damage, osteocytes undergoing deoxyribonucleic acid (DNA) fragmentation during apoptosis, and resorption spaces containing osteoclasts , but osteocyte death is preceded by increased expression of Bax, a pro-apoptotic gene . Osteocytes more than 1–2 mm from the damaged bone show increased expression of Bcl2, an antiapoptotic gene . Osteocytes exert a general suppressant effect on bone remodeling , but BMU origination requires a positive signal, either from dying osteocytes or from surrounding Bcl2-expressing osteocytes, which serves also as a beacon or homing signal for the advancing BMU . Whether the signal is biochemical, electrical, hydraulic, or neural is unknown. Many other factors can influence one or more steps in this complex process, but their role is permissive, not regulatory . In unloaded bone also, osteocyte apoptosis serves as a beacon for osteoclastic removal of bone perceived as no longer needed , but the bone removed for damage repair is completely replaced, whereas the bone removed in response to unloading is replaced incompletely or not at all.
30.8
Metabolic functions of remodeling
The foregoing argument has established three interconnected facts. First, the primary function of metaphyseal cancellous bone in the extremities is mechanical load-bearing. Second, the reason why load-bearing bone must be remodeled is to maintain its mechanical competence. Third, the rate of turnover of load-bearing bone adjacent to fatty marrow, whether cortical or cancellous, is low. Clearly, a low rate of turnover, of the order of 2–5%/year, is sufficient to maintain the mechanical competence of bone, regardless of its location in the skeleton or its geometric features. Consequently, the rate of turnover of axial cancellous bone adjacent to hematopoietic marrow (15–35%/year) is much higher (by a factor of at least five) than is necessary to maintain mechanical competence . Remodeling rates were higher in the past because of changes in nutrition and physical activity , but even the lower rates in preagricultural humans were much higher than needed for maintenance . Unless this mechanically surplus or spare remodeling is simply a form of occupational therapy for cells with nothing better to do, it must serve an entirely different purpose. This conclusion will not surprise the many endocrinologists who have always believed that the main purpose of bone remodeling was to support calcium homeostasis, but the restriction of this function mainly to cancellous bone adjacent to red marrow has not previously been emphasized. The relative importance of the mechanical and metabolic aspects of remodeling, debated inconclusively for many years , is evidently different in different regions of the skeleton, although both are essential to the organism as a whole.
The most important nonmechanical function of bone remodeling concerns the regulation of calcium homeostasis. Bone is involved in both determining the steady-state target value for plasma-free calcium and correcting deviations from the target value . Both of these processes depend on a relatively high rate of bone remodeling, but in quite different ways. Bone mineral also functions as a reservoir for sodium and as a buffer for hydrogen ion regulation. Bone remodeling may also provide biochemical support for hematopoiesis as well as the mechanical support provided by the bone itself. Both the number and the proliferative activity of stem cells are greatest adjacent to the endosteal surface, where they are segregated in microenvironmental niches , and for this reason bone lining cells may need timely replacement. Bone matrix contains growth factors and other regulatory molecules, some of which may act on blood-forming cells rather than on bone cells. For several reasons, it could be advantageous for such molecules to be released into the bone marrow during bone resorption rather than directly from the cells involved in their biosynthesis. Possible reasons include cell polarization, with osteoblasts transporting substances away from, and osteoclasts toward, the marrow; the high proton concentration within the ruffled border of osteoclasts; and a need for intermittent rapid release rather than more continuous slow release. However, this is speculative, and the remainder of the discussion will focus on the relationship between bone remodeling and calcium homeostasis.
Except under conditions of extreme calcium deprivation, the calcium homeostatic function of remodeling is not antagonistic to the mechanical function, since normally calcium homeostasis does not depend on continued net loss of calcium from bone . Steady-state levels of plasma-free calcium can be high, normal, or low, regardless of the directional changes in osteoclastic bone resorption or in calcium balance . Plasma-free calcium is regulated by the joint effects of parathyroid hormone (PTH) on the renal tubular reabsorption of calcium and on the blood–bone equilibrium. This equilibrium is achieved when the inward and outward fluxes of calcium at quiescent bone surfaces are equal, and the calcium level at which this occurs is determined by some effect of PTH on bone lining cells . For this mechanism to be effective, several conditions must be met. First, there must be a high blood flow, which is ensured by the proximity of hematopoietic marrow. Second, the bone at the surface must retain enough water to permit rapid diffusion of minerals, which is ensured by a high rate of remodeling. As previously indicated, as bone ages, secondary mineralization proceeds slowly to completion by crystal enlargement and displacement of water, with a progressive decline in its ability to support the rapid mineral exchanges on which plasma–calcium homeostasis depends . Spare, posttargeted remodeling could prevent excessive aging of surface bone, but as for fatigue damage, from time to time targeted remodeling will be needed to remove bone that has become hypermineralized. The mechanism of targeting is less well understood than for fatigue damage but should be simpler, since the bone to be removed is on rather than beneath the surface. One signal to surface remodeling is loss of osteocytes, a mechanism that serves to maintain osteocyte density, probably in the interests of mineral exchange .
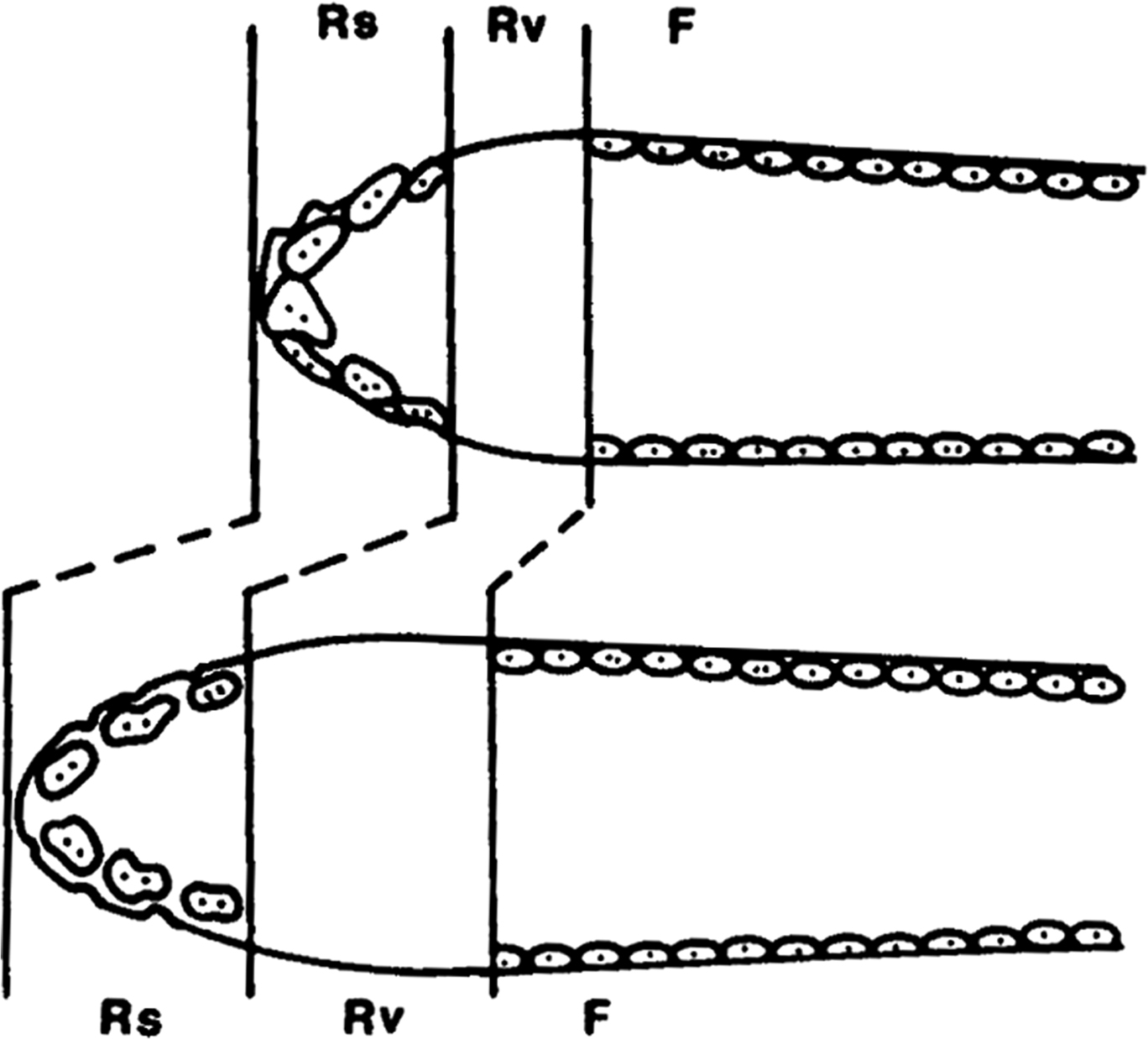
In addition to determining the steady-state target level of plasma free calcium, the bone also participates in the correction of deviations from the target value. A fall in plasma free calcium stimulates PTH secretion, which increases the outflow of calcium from bone, not only by shifting the balance of exchange at quiescent bone surfaces but also by increasing the resorptive activity of existing osteoclasts. This acute effect is quite separate from the long-term effect of PTH to increase activation frequency, osteoclast recruitment, and bone turnover in primary and secondary hyperparathyroidism . Obviously, the rapidity of the correction depends on the number of osteoclasts available, which is determined by the number of BMUs present, and by the efficiency of the local circulation. The most important use for this mechanism is to accommodate the circadian changes in the supply of calcium from intestinal absorption, with an approximately 12- to 16-hour period of eating, followed by an 8- to 12-hour period of fasting, during which both PTH secretion and bone resorption increase . In each BMU, the cutting cone (in osteonal remodeling) or hemicone (in hemiosteonal remodeling) advances more rapidly at night and slows down to allow the closing cone (or hemicone) to catch up during the day. This concertina-like action ( Fig. 30.2 ) allows the skeleton to supply calcium at night when it is needed, without affecting the terminal balance of the BMUs and so without causing an irreversible loss of bone.