55.1
Introduction
Osteoporosis and periodontal disease are chronic conditions that disproportionately affect older populations. Both diseases are associated with low systemic bone mineral density (BMD) and they share common risk factors. Osteoporosis is a systemic skeletal disease characterized by decreased BMD and is associated with increased risk for fractures. Periodontal disease is an inflammatory condition caused by the dysbiosis of pathogenic oral bacteria that leads to destructive loss of tissue and alveolar bone surrounding the teeth. In its early stages, periodontal disease is characterized by inflamed oral tissue. As the disease progresses in severity, there is increasing destruction of gingival tissue and bone loss surrounding the teeth. As severity of periodontal disease increases, tooth loss (TL), and edentulism can result.
55.2
Periodontal disease and tooth loss prevalence
Periodontal disease is highly prevalent in US adults. Surveillance data from the 2009–14 National Health and Nutrition Examination Survey (NHANES) show prevalence of mild/moderate and severe periodontal disease increases steeply with age from 25.3% to 4.1% among adults aged 30–34 years to 50.7% and 9.0% among those 65 years and older, respectively ( Table 55.1 ) . If untreated, periodontitis can lead to impaired oral function, poor quality of life, and TL . There also appears to be increased susceptibility to common chronic diseases (diabetes, hypertension, heart disease, cancer, cognition, and osteoporosis) and premature mortality among adults with periodontitis . Periodontal disease also contributes to disability life-years and health-care costs . Given the US population of men and women aged 65 or older is expected to increase from 40 M in 2010 to nearly 80 M in 2050 , the public health burden of periodontal disease and its consequences will be substantial .
| Characteristic | None | Mild/moderate | Severe | Any disease |
|---|---|---|---|---|
| Total | 57.7 (1.4) | 34.4 (1.2) | 7.9 (0.5) | 42.2 (1.4) |
| Age (years) | ||||
| 30–44 | 70.5 (1.5) | 25.3 (1.4) | 4.1 (0.3) | 29.5 (1.5) |
| 45–64 | 53.9 (1.6) | 35.6 (1.4) | 10.5 (0.8) | 46.1 (1.6) |
| 65 and older | 40.2 (2.1) | 50.7 (1.9) | 9.0 (1.0) | 59.8 (2.1) |
| Sex | ||||
| Male | 49.7 (1.4) | 38.7 (1.2) | 11.5 (0.8) | 50.3 (1.4) |
| Female | 65.4 (1.5) | 30.3 (1.3) | 4.3 (0.4) | 34.6 (1.6) |
| Race/ethnicity | ||||
| Mexican Hispanic | 40.3 (1.7) | 46.4 (1.5) | 13.3 (1.4) | 59.7 (1.7) |
| Hispanic (other) | 51.5 (1.6) | 40.7 (1.3) | 7.8 (0.9) | 48.5 (1.6) |
| Non-Hispanic White | 63.0 (1.7) | 31.0 (1.5) | 6.0 (0.6) | 37.0 (1.7) |
| Non-Hispanic Black | 43.4 (1.9) | 42.0 (1.3) | 14.6 (1.1) | 56.6 (1.9) |
| Other race | 53.9 (2.6) | 36.8 (2.3) | 9.3 (1.4) | 46.1 (2.6) |
| Smoking status | ||||
| Nonsmoker | 65.6 (1.4) | 29.5 (1.2) | 4.9 (0.5) | 34.4 (1.4) |
| Former smoker | 54.3 (1.8) | 37.7 (1.8) | 8.0 (0.7) | 45.7 (1.8) |
| Current smoker | 37.6 (1.7) | 45.3 (1.7) | 17.1 (1.3) | 62.4 (1.7) |
| Income category | ||||
| Low ≤130% | 42.4 (1.9) | 44.6 (1.6) | 12.9 (1.0) | 57.6 (1.9) |
| Mid 131%–350% | 54.5 (1.7) | 37.4 (1.7) | 8.1 (0.8) | 45.5 (1.7) |
| High ≥351% | 71.3 (1.4) | 24.7 (1.2) | 4.0 (0.6) | 28.7 (1.4) |
| Body mass index (kg/m 2 ) | ||||
| <25 | 60.6 (1.6) | 31.7 (1.6) | 7.7 (0.6) | 39.4 (1.8) |
| 25–30 | 58.1 (1.4) | 33.8 (1.2) | 8.1 (0.7) | 41.9 (1.4) |
| >30 | 55.4 (1.5) | 36.8 (1.5) | 7.8 (0.7) | 44.6 (1.5) |
| Diabetes mellitus | ||||
| Yes | 40.2 (2.2) | 49.0 (2.4) | 10.8 (1.3) | 59.8 (2.2) |
| Use of dental floss in past 7 days | ||||
| Yes | 62.0 (1.3) | 32.1 (1.2) | 5.8 (0.5) | 38.0 (1.3) |
| Last dental visit (month) | ||||
| ≤6 | 69.6 (1.7) | 26.4 (1.6) | 3.9 (0.6) | 30.4 (1.7) |
| 7–12 | 62.1 (2.4) | 31.6 (2.2) | 6.3 (1.0) | 37.9 (2.4) |
| >12 or never | 45.2 (1.8) | 41.3 (1.7) | 13.5 (1.2) | 54.8 (1.8) |
| Number of teeth missing | ||||
| 0 | 76.0 (1.4) | 21.2 (1.2) | 2.8 (0.5) | 24.0 (1.4) |
| 1–5 | 56.8 (1.4) | 35.9 (1.2) | 7.3 (0.7) | 43.2 (1.4) |
| 6–10 | 34.9 (2.3) | 50.9 (2.1) | 14.2 (1.3) | 65.1 (2.3) |
| >10 | 28.2 (1.8) | 52.4 (2.1) | 19.3 (1.5) | 71.8 (1.8) |
a Weighted to account for the complex sampling design of NHANES and to project the estimates to the entire US population.
TL and edentulism are also very prevalent in the United States and continue to cause a heavy economic burden in the United States and globally. TL affects quality of life and has been associated with many morbidities such as cardiovascular disease, malnutrition , and mortality . The overall prevalence of TL and edentulism in the United States has been decreasing over the past four decades , in part due to changes in health and dental practices over the past century. Factors influencing this change include better nutrition, improved living environments, declines in smoking, improved oral hygiene and dental care and practices, and greater access to dental care .
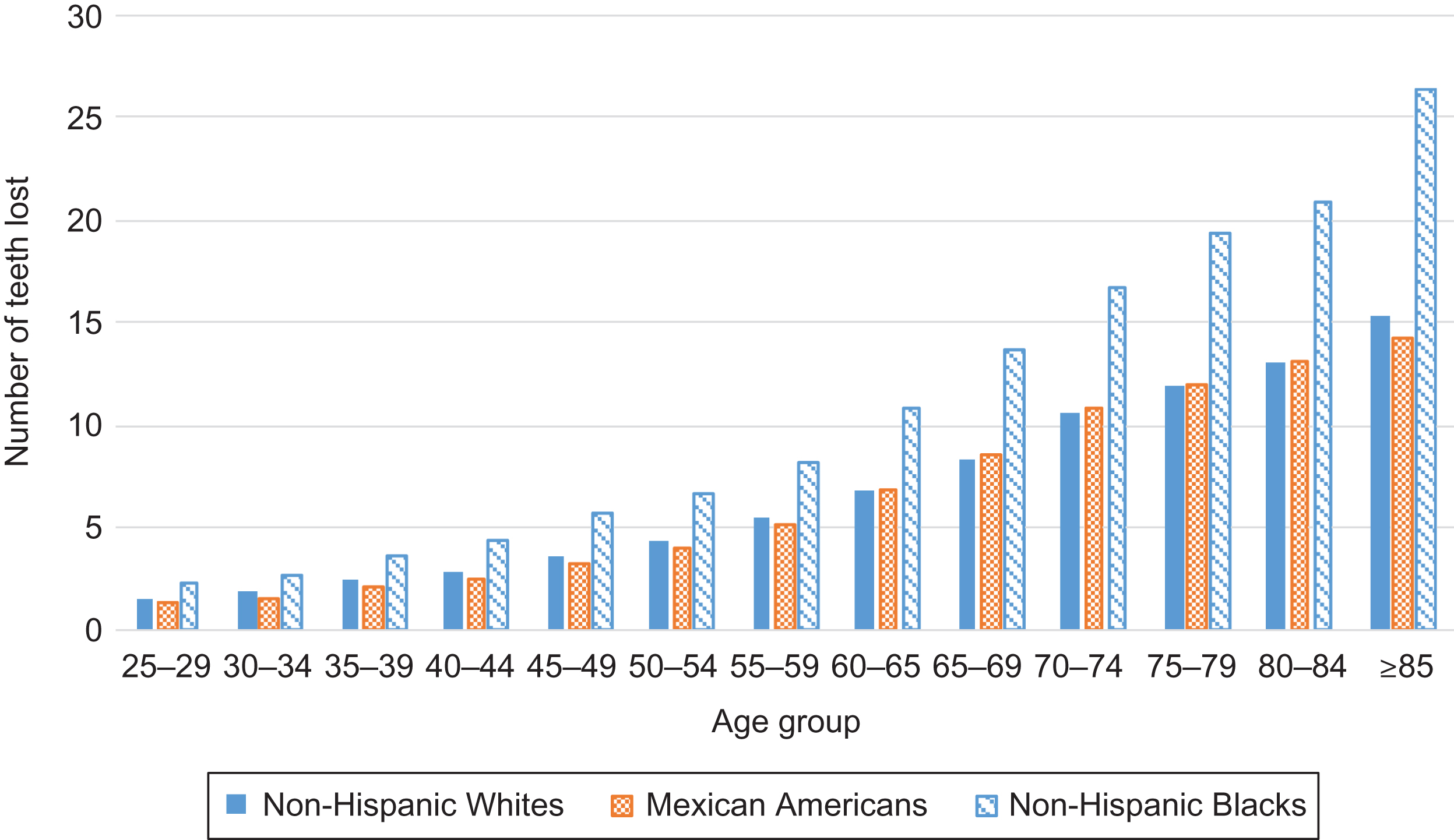
Although the overall prevalence of periodontal disease and TL has been declining, prevalence still remains higher among some groups and the disparities continue to widen. In an analysis by the Centers for Disease Control and Prevention (CDC) a marked disparity in TL (not including mild or moderate disease) by age group was observed for non-Hispanic Blacks compared to non-Hispanic Whites or Mexican Americans ( Fig. 55.1 ). Luo et al. pooled data from 37,609 men and women ≥25 years and older who with at least one permanent tooth participated in NHANES I, II, or III (1971–2012). Using hierarchical age-period-cohort cross-classified random-effects modeling, accounting for time period and birth cohort, found that overall TL increased with age, but non-Hispanic Blacks lost more teeth, earlier, and at a faster rate than non-Hispanic Whites and Mexican Americans .
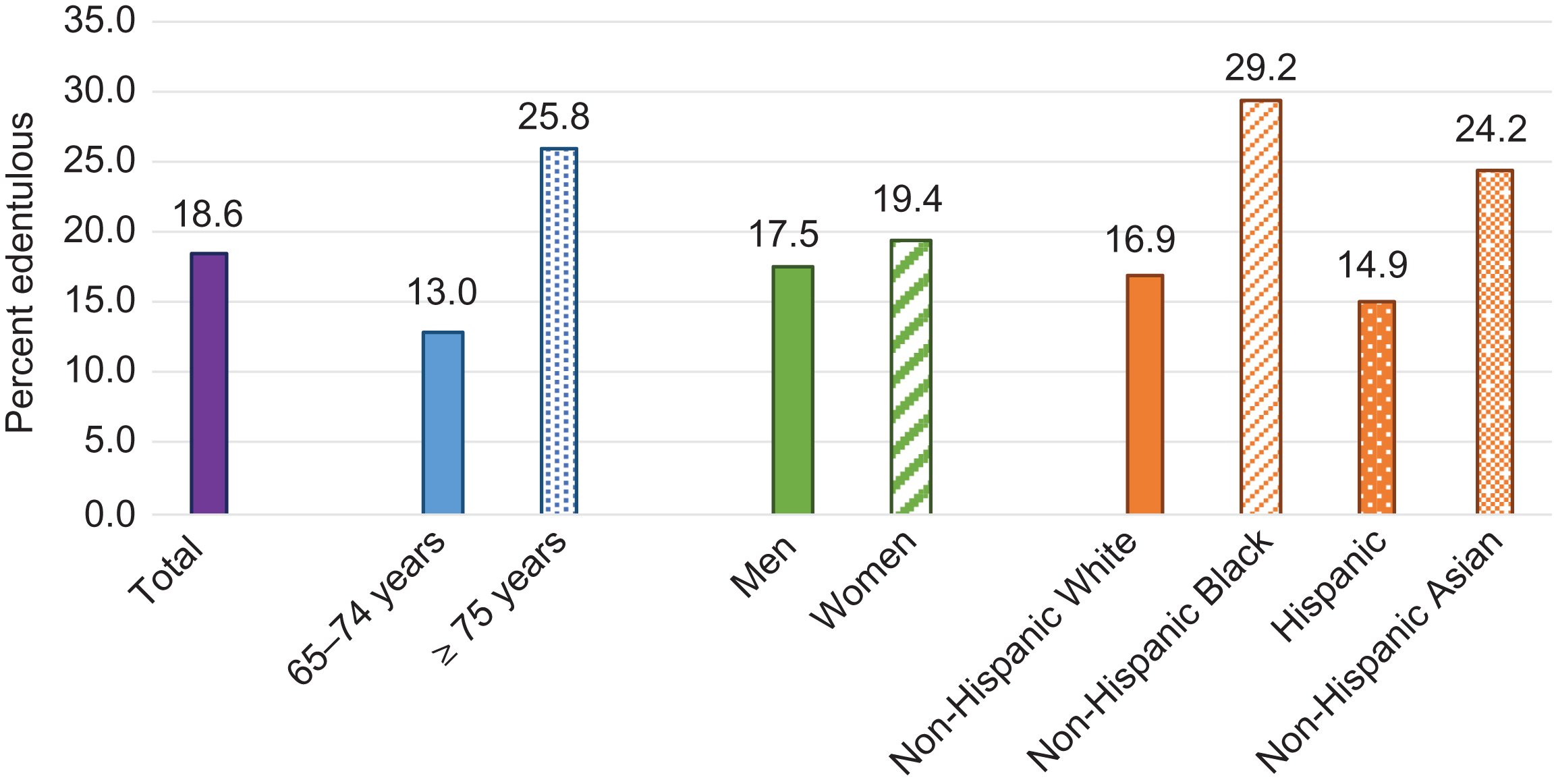
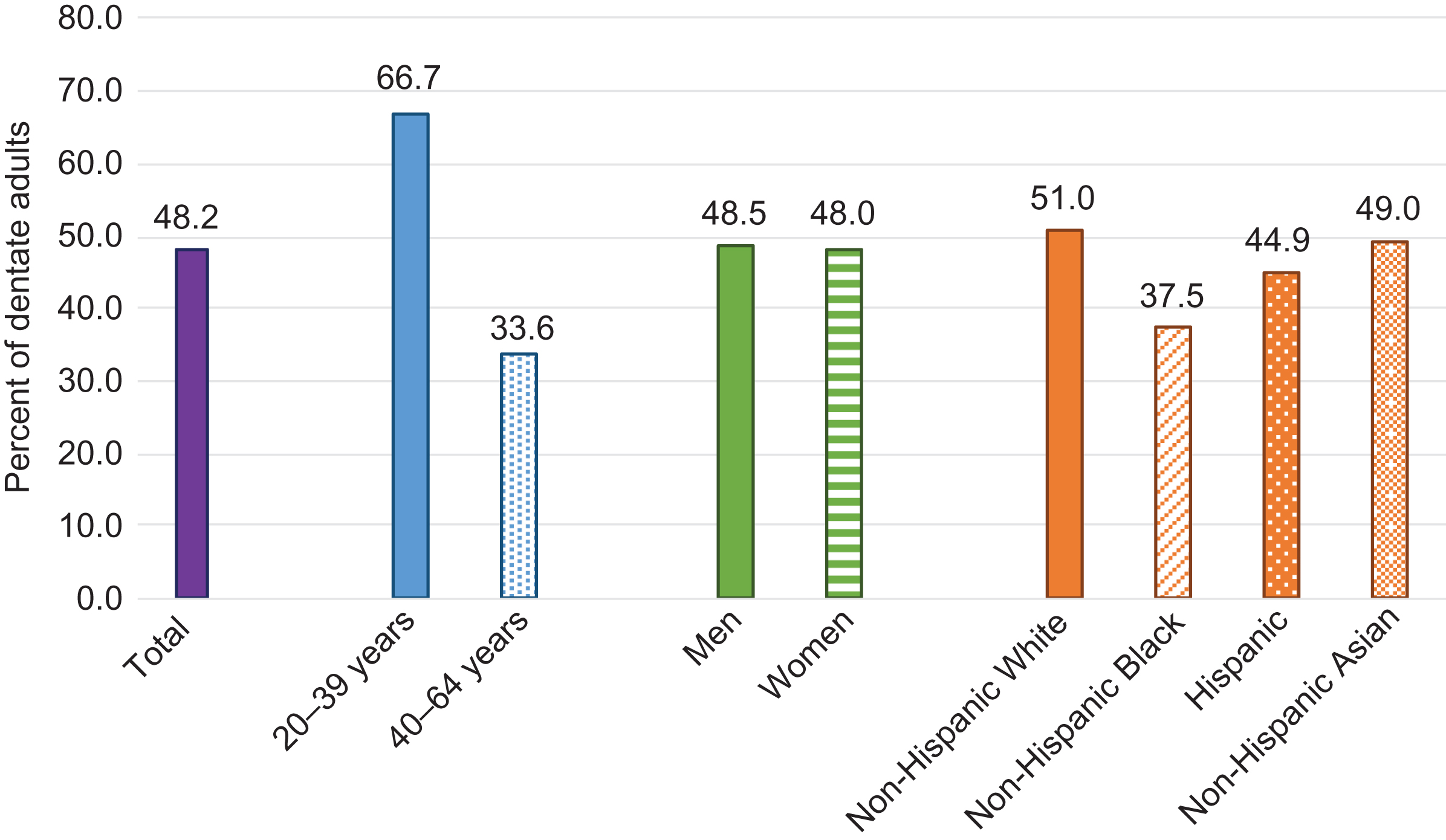
Among participants in 2011–14 NHANES ( Fig. 55.2 ), nearly 18.6% of seniors aged ≥65 years were edentulous, with prevalence highest among non-Hispanic blacks (29.2%) and women (19.4%) . Non-Hispanic blacks between 20 and 64 years of age had less tooth retention (37.5%), than non-Hispanic whites (51%), non-Hispanic Asians (49%), or Hispanics (44%) ( Fig. 55.3 ). Among those 20–64 years old, TL did not vary by sex . Among all adults aged 20 years and older in the 1999–2004 NHANES, edentulism was highest among current smokers and those with lower socioeconomic status (below the federal poverty line and less education) .
55.3
Clinical measures of periodontal disease
Clinical measures that characterize periodontal disease presence and severity are extensive, making interpretation of the associations between osteoporosis, periodontal disease, and TL challenging to summarize, especially over time. Periodontal disease is often based on soft tissue probing measures such as the gingival index or bleeding on probing (BOP), probing pocket depth (PD), and/or clinical attachment level (CAL) . Fewer studies rely on oral radiographs that measure alveolar crestal height (ACH) , residual ridge resorption (RRR) , or mandibular BMD (MBMD) . Some studies have focused on TL or edentulism, not periodontal disease per se . In studies that focus on TL, there is uncertainty as to whether the loss was due to periodontal disease, caries, trauma, or other reasons . In addition, studies have been cross-sectional in nature, conducted in selected cohorts or have small sample sizes, all limiting interpretation. Although not directly a measure of periodontal disease, some studies have focused on measures of poor oral care that can lead to periodontal disease such as extent of plaque or calculus indices .
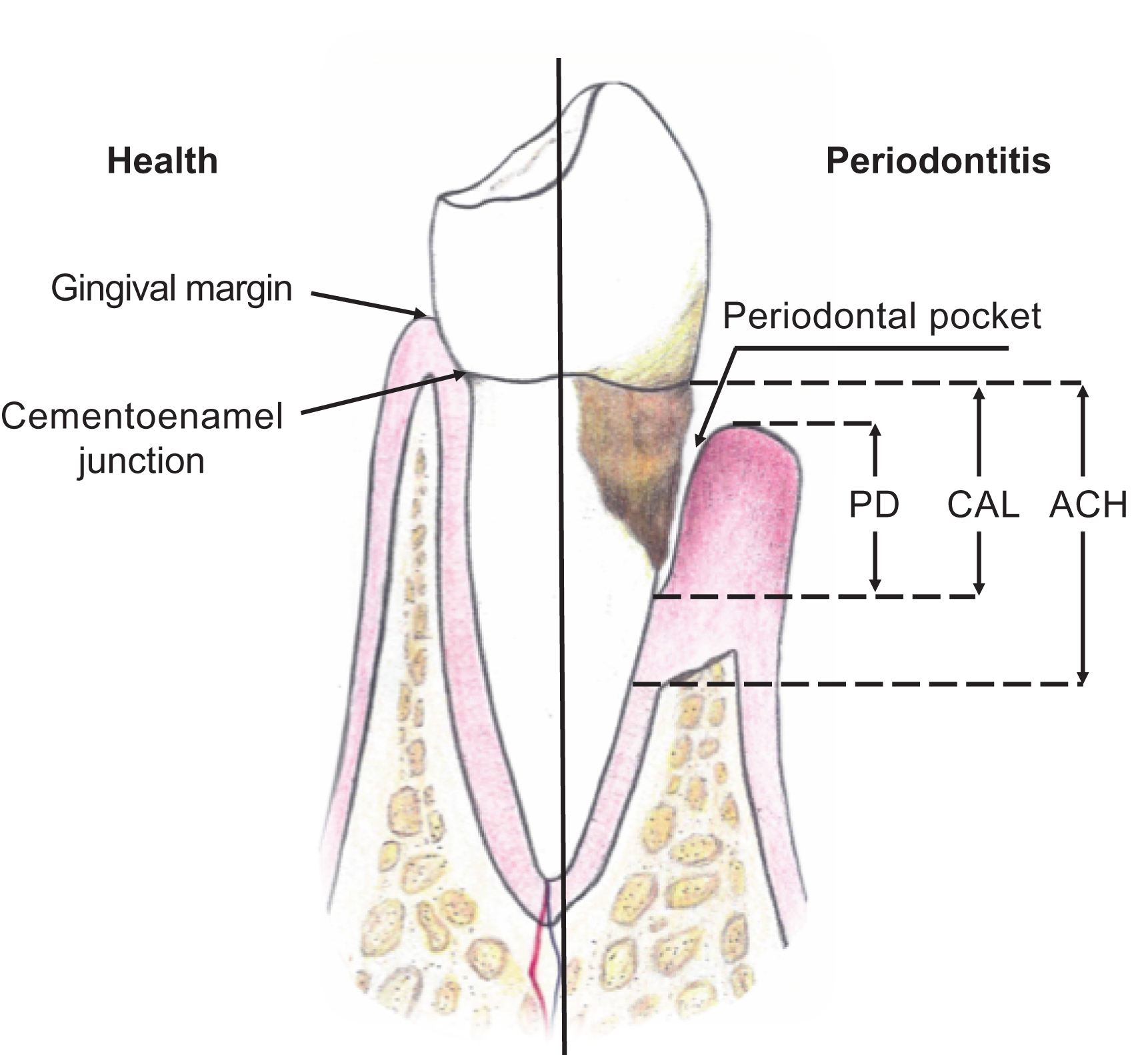
Oral probing measures to characterize periodontal disease include probing PD and CAL . As illustrated in Fig. 55.4 , PD is a measure of the distance in millimeters from the gingival margin of the soft tissue surrounding the tooth to the base of the sulcus/pocket. CAL is the distance in millimeters from the cementoenamel junction (CEJ) on the surface of the tooth to the base of the periodontal pocket. Probing measurements can be assessed as full-mouth measures (excluding third molars) or as partial recording protocols (PRP). Full-mouth measurements are the most accurate, but more time-consuming and costly . PRPs involve assessments on a fixed subset of teeth, or random subsets of teeth or mouth quadrants .
Measurements are typically made on two to six fixed-sites per tooth (distobuccal, midbuccal, mesiobuccal, distolingual, midlingual, and mesiolingual) using either a manual probe or constant force electronic periodontal probes. The specific sites assessed vary in the literature. The National Institute of Dental and Craniofacial Research recommends using only the mesiobuccal and midbuccal sites when using PRPs, whereas the clinical norm for full-mouth assessment is to examine all six sites in all teeth present excluding third molars . A bias analysis study of the various recording protocols found PRP’s underestimate the prevalence of attachment loss compared to full-mouth assessments. Random half-mouth sampling using two or three fixed-sites and fixed-sites full-mouth using two sites underestimated disease severity .
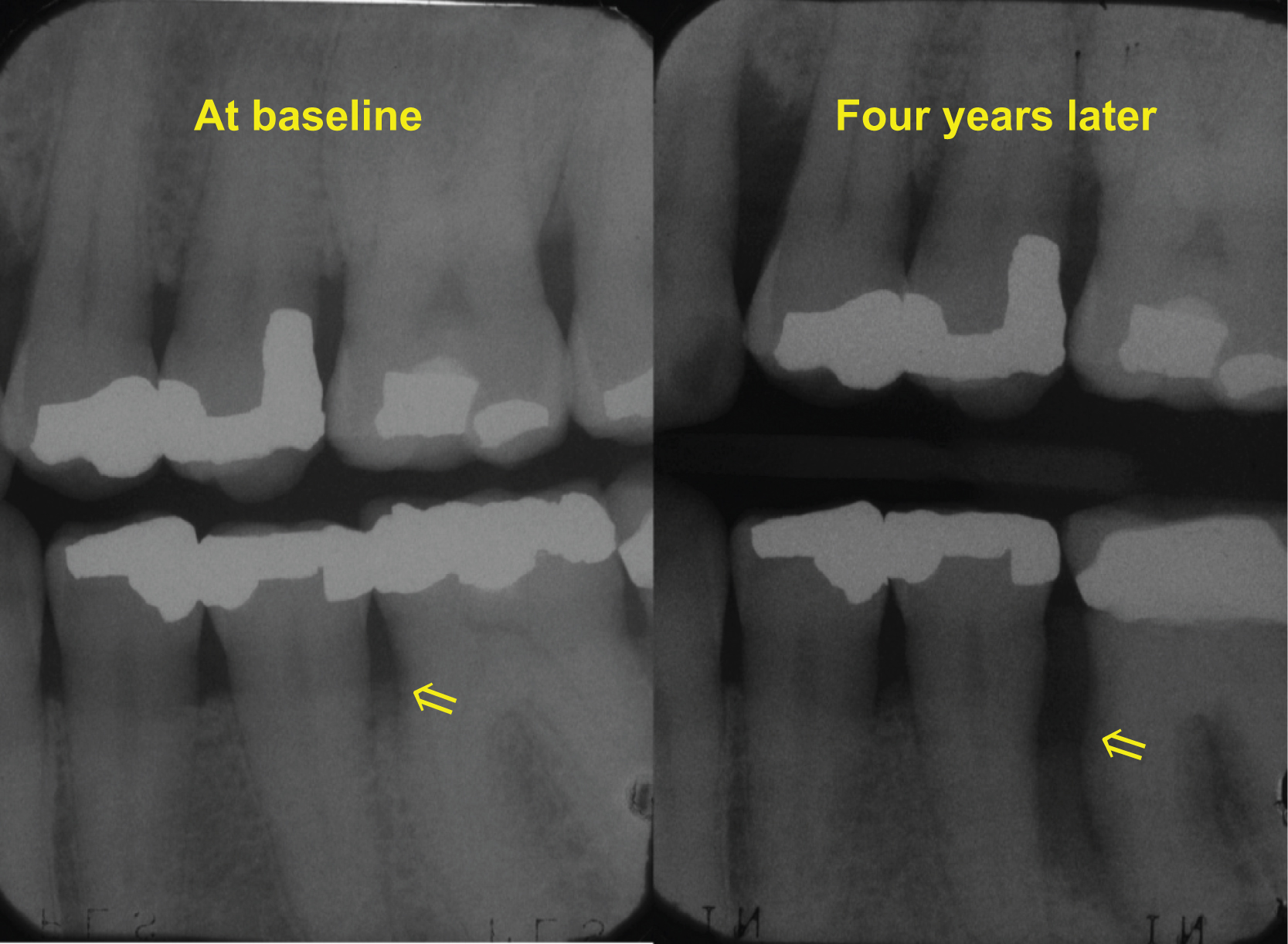
ACH is less commonly used in population-based research studies as it requires radiograph collection. Typically ACH is assessed using a series of standardized intraoral radiographs with loss of bone surrounding the teeth measured as the distance from the CEJ to the most coronal interdental bone next to the tooth. ACH measurements are typically made on two surfaces per tooth (mesial and distal) using techniques described by Hausmann . Radiographs (film/digital), panoramic scans, and CT have all been used in characterizing ACH. Cephalometric head positioners are used to stabilize patient’s heads during imaging. Radiographic techniques have been adapted to assess oral bone density, as well as ACH and density change over time . For change in ACH a side-by-side technique has been established . Fig. 55.5 is a radiograph from one individual measured at baseline and 4 years later, demonstrating loss of bone that can occur with periodontal disease progression. Similar to soft tissue measures, ACH is usually presented as the mean value in all teeth present and measured. Reliability and accuracy of these measure have been reported for ACH, CAL, and PD .
MBMD has been assessed using intraoral radiographs. Early studies have used subtractive radiography techniques to assess MBMD and longitudinal change ; however, these methods have limitations in accuracy, positioning geometry, and limited ability to quantify BMD . Use of densiometric reference wedges of known densities within oral radiograph can allow for quantifiable measurement of mandibular bone mass . The wedge is inserted within the radiographic image, allowing for comparisons of known density of the wedge (via brightness and contrast) to estimate the density oral bone . Aluminum wedges are typically used because the material is homogenous and the properties are similar to human bone , although copper wedges have been used . The step-wedge method has been used for decades, but it is only useful to evaluate small regions, requires specialized equipment for reading the scans, is time-consuming, and has limited use in edentulous individuals .
Mandibular cortical thickness (MCT) is another common technique used to measure MBMD. MCT is the dense outermost layer of the mandible, typically assessed with panoramic X-rays or CT scans . Early studies have also attempted to use dual-photon absorptiometry (DPA) to measure MCT .
TL is determined either by self-report or by visual assessment by a trained dental examiner. Typically, an examiner will use the Decayed, Missing, Filled (DMF) index, a scored assessment of permanent dentition expressed as the total number of teeth or surfaces that are decayed, missing, or filled. Scores typically range from 0–28 (if 3 rd molars are excluded). When the DMF index is specifically applied to teeth rather than teeth surfaces, it is called the DMFT. The DMFT is used to record the present/absent teeth, reasons for missing teeth, the structure and integrity of the crown and root of each tooth, and dental work (bridges, implants, denture use, or fillings) for each tooth . Edentulism is the loss of all existing teeth in the mouth. Reasons for TL are typically characterized as due to periodontal disease, caries, trauma, malposed, or congenitally missing or unerupted .
Definitions of extent of periodontal disease include the CDC and American Academy of Periodontology (AAP) definition , which categorizes disease severity based on combined measures of CAL and PD (see Table 55.2 ). A few studies have used ACH that uses ACH and TL to categorize severity . Often studies report on mean measures of ACH, CAL or PD, or as quantiles that describe severity. Two studies have defined periodontal disease using the British Society of Periodontology criteria that uses PD in addition to presence/severity of calculus, plaque, and bleeding to characterize disease .
| Operational definition | |
|---|---|
| ACH definition | |
| None | Whole-mouth mean ACH <2 mm, no site ≥4 mm, no reported tooth loss due to periodontal disease |
| Mild/moderate | Whole-mouth mean ACH ≥2 to <3 mm, or ≥1 tooth with an ACH ≥4 mm, but no reported tooth loss due to periodontal disease |
| Severe | Whole-mouth mean ACH ≥3 mm or ≥2 teeth with ACH ≥5 mm, or tooth loss (≥1) due to periodontal disease |
| CDC/AAP definition | |
| None/mild | Neither “moderate” nor “severe” periodontitis |
| Moderate | ≥2 Interproximal sites with CAL ≥4 mm (not on same tooth) or ≥2 interproximal sites with PD ≥5 mm (not on same tooth) |
| Severe | ≥2 Interproximal sites with CAL ≥6 mm (not on same tooth) and ≥1 interproximal site with PD ≥5 mm |
55.4
Osteoporosis characterization in studies of periodontal disease and tooth loss
Prevalence, risk factors, and background on osteoporosis, and assessment and diagnostic methodologies to describe osteoporosis are characterized elsewhere in this book. Characterization of osteoporosis or systemic bone loss varies in studies assessing the relationship between osteoporosis and periodontal disease/TL, complicating interpretation and comparison of studies assessing these relationships. Techniques have varied over time; however, dual-energy X-ray absorptiometry (DXA) is the most common method used in recent reports. Most studies have used hip/femoral neck and spine measures in reporting results. Generally these are reported as T-scores or site specific BMD values (g/cm 2 ). Several studies have associated periodontal disease and/or TL with fracture rather than bone density.
55.5
Mechanisms by which osteoporosis and periodontal disease may be associated
Periodontal disease and osteoporosis are prevalent conditions, especially in aging adults. Both conditions are associated with bone loss. Periodontitis is believed to result from pathogenic bacteria and a dysbiosis in the oral cavity that elicits an immune response, signaling the activation of macrophages which promote the secretion of proinflammatory mediators resulting in tissue injury and destruction, loss of collagenous support of the tooth, as well as loss of surrounding alveolar bone . Osteoporosis is a result of loss of BMD throughout the body, including loss that can exist in the oral cavity. Proinflammatory mediators, such as tumor necrosis factor-α and receptor activator of nuclear factor kappa-β ligand, involved in the tissue and bone destruction in periodontitis, promote the activation of osteoclasts and bone resorption . In addition, osteoporosis and periodontal disease share common risk factors including smoking, diabetes, and hormones. As such, periodontal disease and osteoporosis may be related; however, because of the few prospective studies, it is not clear whether there is a causal association between these common conditions.
55.6
Risk factors for periodontal disease
Periodontal disease prevalence, severity, and rate of progression vary according to age and other factors. Severity of disease can also vary within regions of the mouth within a person. Oral hygiene is one of the most significant and influential risk factors in the development and progression of periodontal disease. Poor oral health has been linked to the accumulation of dental plaque and pathogenic oral bacteria, and the development of gingivitis . In addition to oral hygiene, other important risk factors for periodontal disease include age; race/ethnicity and socioeconomic status; sex and hormone-related conditions; lifestyle factors such as smoking, dietary, and nutritional intake; immunologic conditions; obesity, metabolic syndrome, and diabetes; genetics, and stress .
55.7
Studies of the association between periodontal disease and osteoporosis
55.7.1
Methods for reviewing the literature
The revised PRISMA guidelines (Preferred Reporting Items for Systematic Review and Meta-analysis Protocols) were used to guide review of previous literature assessing the association between periodontal disease or TL and osteoporosis or systemic BMD. A MEDLINE (PubMed) search was performed on August 1, 2018, through November 30, 2018 using the following strategy: (“osteoporosis” OR “osteopenia” OR “low bone density” OR “bone density” OR “BMD”) AND [(“periodontal disease” OR “oral bone loss” OR “residual ridge”) OR (“edentulism” OR “TL”)] AND “Humans” [Mesh] AND English [lang]. Additional searches were made by reviewing the reference lists of articles reviewed in the literature review described previously. We also reviewed references from published systematic reviews and performed citation searches between August 1, 2018 and November 30, 2018, for any additional eligible articles. Articles were included for review if they were in English, assessed the association between osteoporosis/osteopenia or systemic BMD and periodontal disease, MBMD, or TL, and used a human sample. Five hundred thirty-one articles were identified through MEDLINE search and an additional 10 were identified through reviewing systematic reviews. A total of 541 records were screened. Four hundred and ten articles were excluded with reasons: title/abstract not meeting inclusion criteria ( n =371), animal studies ( n =35), or duplicates ( n =4). One hundred thirty-one full-text articles were reviewed for eligibility, 26 were review/meta analyses, and 54 were excluded because exposure/outcome assessment did not meet inclusion criteria. A total of 51 clinical and observational studies were included in the qualitative synthesis of the literature in this section.
55.7.2
Clinically defined periodontal disease by probing measures and osteoporosis or bone density
Numerous cross-sectional and case-control studies have investigated the association between osteoporosis/BMD and periodontal disease, with periodontal disease assessed using clinical probing measures: CAL, BOP, and PD ( Table 55.3 ). Most but not all studies observed significant associations between osteoporosis or decreased BMD and worse probing soft tissue periodontal disease measures, while one study only observed associations between systemic BMD and severe but not moderate CAL , another only observed an association with measures of PD but not BOP , and another only observed significant associations in women but not men .









