Chapter 30 Primary Myelofibrosis
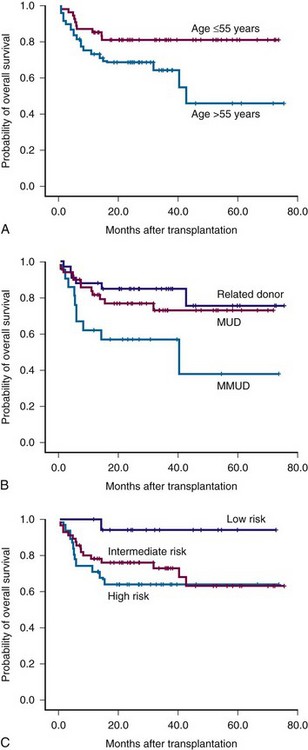
Figure 30-1 Survival of patients with myelofibrosis after reduced-intensity allogeneic stem cell transplantation according to age (A), donor (B), and Lille risk profile (C).
Chapter 30 Primary Myelofibrosis

Figure 30-1 Survival of patients with myelofibrosis after reduced-intensity allogeneic stem cell transplantation according to age (A), donor (B), and Lille risk profile (C).
