##
It is a curious conundrum when adrenal venous sampling (AVS) is performed in a patient with primary aldosteronism (PA) and the aldosterone concentrations from both successfully sampled adrenal veins are low. This clinical circumstance raises the exceedingly rare possibility of an ectopic aldosterone-producing tumor in a gonad or kidney. However, at Mayo Clinic, every time this conundrum on AVS has occurred over the past 30 years, it proved to be due to a technical issue with AVS itself. Herein, we share such a case.
Case Report
The patient was a 34-year-old man who had been hypertensive for 6 years. He had spontaneous hypokalemia for the past 5 years. He was treated with 120 mEq of potassium chloride per day along with four antihypertensive drugs including betaxolol (10 mg daily), amlodipine (10 mg daily), valsartan (320 mg daily), and hydrochlorothiazide (25 mg daily). Blood pressure was poorly controlled and averaged 165/95 mmHg. His lowest serum potassium concentration was 2.2 mEq/L. He had no signs or symptoms of Cushing syndrome. He had a family history of hypertension in his father and brother.
INVESTIGATIONS
The baseline laboratory test results are shown in Table 15.1 . The patient had positive case detection testing for PA with a plasma aldosterone concentration (PAC) >10 ng/dL and plasma renin activity (PRA) <1.0 ng/mL per hour. Although the patient was taking a diuretic when he came to Mayo Clinic, because of the previously documented spontaneous hypokalemia, formal confirmatory testing was not needed. ,
| Biochemical Test | Result | Reference Range |
| SodiumPotassiumCreatinineeGFRAldosteronePlasma renin activity | 1413.40.9>6035<0.6 | 135–1453.6–5.20.8–1.3>60 mL/min per BSA≤21 ng/dL≤0.6–3 ng/mL per hour |
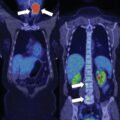
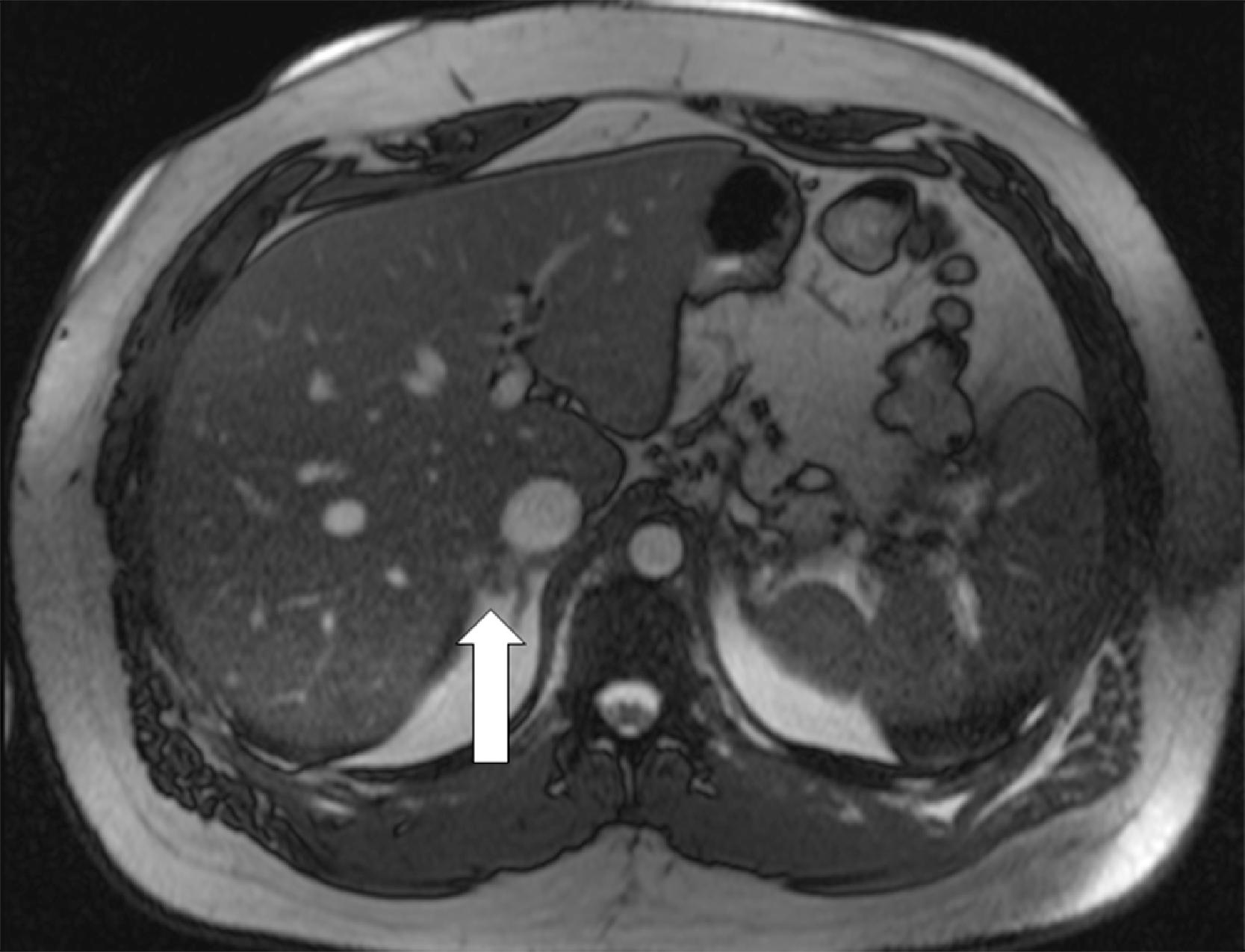
MRI scan of the abdomen demonstrated a 1-cm nodule in the lateral aspect of the right adrenal gland and micronodularity in the left adrenal gland ( Fig. 15.1 ).
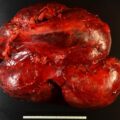
After a thorough discussion with the patient, he was keen to pursue a surgical cure of hypokalemia and either cure or achieve better control of his hypertension on less medication.
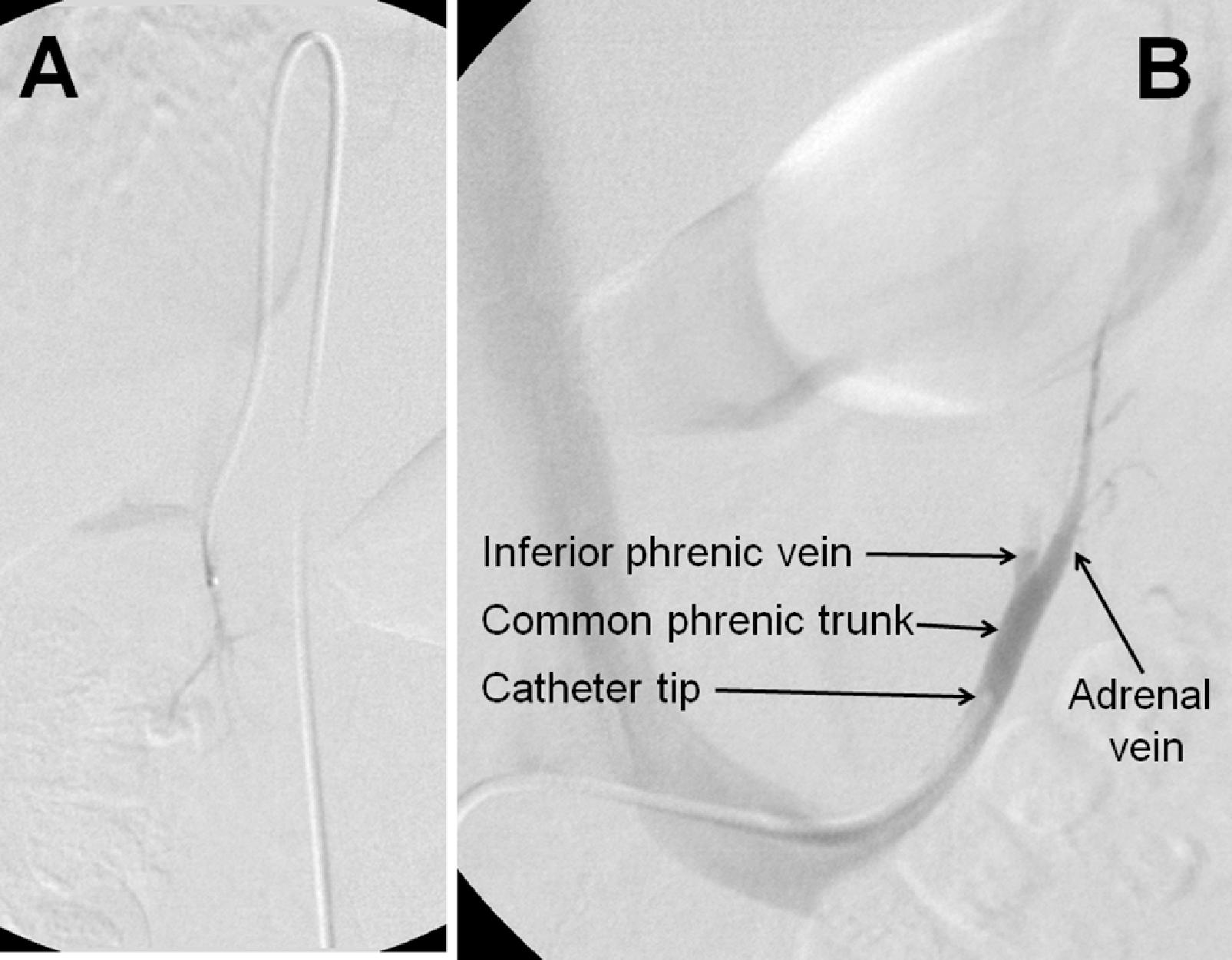
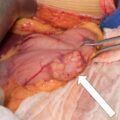
AVS was performed as the next step ( Fig. 15.2 ). AVS was successful based on adrenal vein-to-IVC cortisol gradients of more to 5-to-1 ( Box 15.1 ). With the continuous cosyntropin infusion protocol (cosyntropin 50 mcg/h administered intravenously starting 30 minutes before AVS and continued throughout the procedure), the adrenal vein-to-IVC cortisol gradients are typically well above the 5-to-1 cutoff (in this case, 31.4-to-1 on the right and 23.0-to-1 on the left). Each adrenal vein aldosterone (A) concentration was divided by the respective cortisol (C) concentration for the A/C ratio (see Box 15.1 ). In this case, the A/C ratio from each adrenal was markedly less than the A/C ratio in the IVC (0.17 on the right versus 0.14 on the left versus 3.1 in the IVC). When an A/C ratio from an adrenal vein is >50% less than that in the IVC, it is termed contralateral suppression . In this patient aldosterone secretion from both adrenal glands appeared to be suppressed, raising the possibility of an ectopic aldosterone-producing neoplasm in a kidney or gonad. As a result of these findings, AVS was repeated, and the renal and gonadal veins were also sampled. The IVC aldosterone concentration was 85 ng/dL. The aldosterone concentrations from the right and left renal veins were 68 ng/dL and 75 ng/dL, respectively (thus excluding an ectopic source from the kidneys). The aldosterone concentrations from the right and left gonadal veins were 71 ng/dL and 68 ng/dL, respectively (excluding an ectopic source from the testicles). Taking great care on the right side to have the catheter tip in the proximal right adrenal vein the right adrenal gland proved to be the source of aldosterone hypersecretion ( Fig. 15.3 and Box 15.2 ). Based on cases like this one, a coaxial guide wire-catheter technique was developed to facilitate successful right adrenal venous sampling by positioning the catheter tip at the orifice of the right adrenal vein.