As we have highlighted in the other cases, the clinical management of catecholamine-secreting pheochromocytomas or paragangliomas (PPGLs) can be challenging—both diagnostically and therapeutically. However, when PPGL is diagnosed in the setting of pregnancy, the complexity rises more than 10-fold. Herein we share a case of a patient with a large retroperitoneal paraganglioma (PGL) discovered during pregnancy.
Case Report
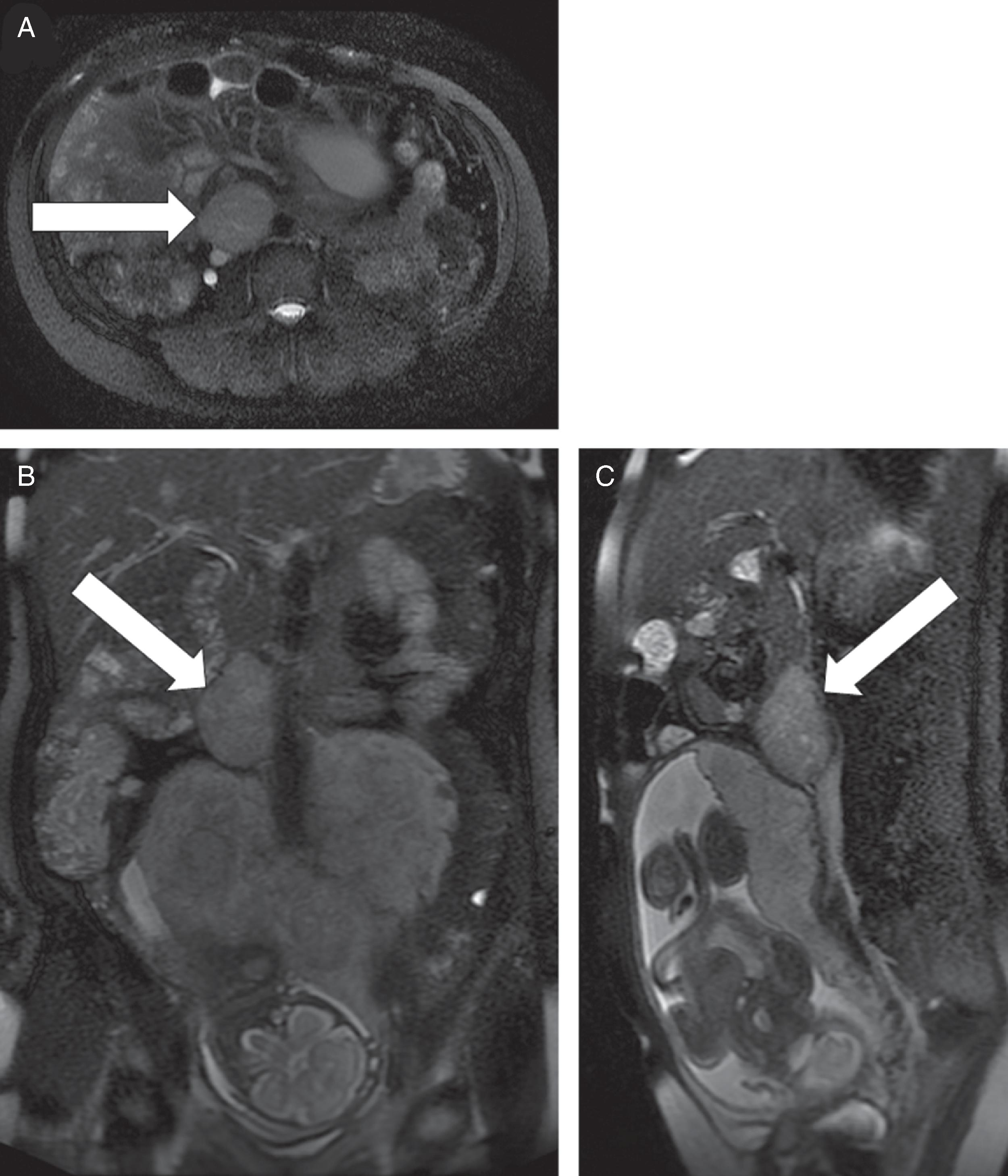
This 35-year-old woman (gravida 2, para 1) presented to the emergency department during her 28th week of pregnancy with palpitations, severe headache, and lightheadedness. Two years previously she had been diagnosed with “reversible cerebral vasoconstriction syndrome” associated with four thunderclap headaches; she was on chronic treatment with metoprolol 25 mg extended release twice daily. In the emergency department her blood pressure was 190/101 mmHg. The elevated blood pressure rapidly returned to normal without intervention. She was admitted to the hospital for observation. She had episodic surges in blood pressure, which appeared to be associated with positional change (e.g., with sitting up, turning to the side). Monitoring during the episodes revealed a transient drop in blood pressure with a subsequent surge in blood pressure and then bradycardia. Forty-five minutes after she received her evening dose of metoprolol, she had sustained systolic blood pressure in the 220s mmHg for 10 minutes that was associated with fetal heart rate decelerations. The metoprolol was discontinued and her episodes seemed to improve. Pheochromocytoma was suspected. Plasma normetanephrine was markedly elevated at 22.4 nmol/L (normal, <0.9) and metanephrine was <0.2 nmol/L (normal, <0.5). Abdominal magnetic resonance imaging (MRI) performed during the 29th week of gestation detected a 3.8 cm × 4.9 cm × 6.8 cm PGL at the inferior aspect of the right kidney flanking the aorta and inferior vena cava ( Fig. 95.1 ). The PGL compressed the inferior vena cava and the posterior-superior aspect of the gravid uterus. There is no family history of PPGL. She has had no prior computed abdominal imaging.
TREATMENT
Based on multidisciplinary team (endocrinology, surgery, obstetrics, neonatology, and anesthesiology) discussion and informed patient decision making, it was decided to treat her with adrenergic blockade for the duration of her pregnancy and resect the PGL with an open operative approach postpartum. Because of the body position impact on blood pressure, she was kept at bedrest for the remainder of the pregnancy. Phenoxybenzamine was titrated to 10 mg three times a day and β-adrenergic blockade was added with propranolol 10 mg three times a day. She was maintained on a high-sodium diet with 1-g sodium chloride tablets—two tablets three times a day with food. With this approach, her seated systolic blood pressure ran 80–90 mm Hg in the morning and 100–120 mmHg in the afternoon. Her paroxysmal symptoms resolved. A preplanned cesarean section (C-section) was performed at 36 weeks of gestation. Except for a slight increase in blood pressure at the time of delivery, maternal hemodynamics remained stable throughout the C-section. The baby boy was healthy and required no medical assisted support.
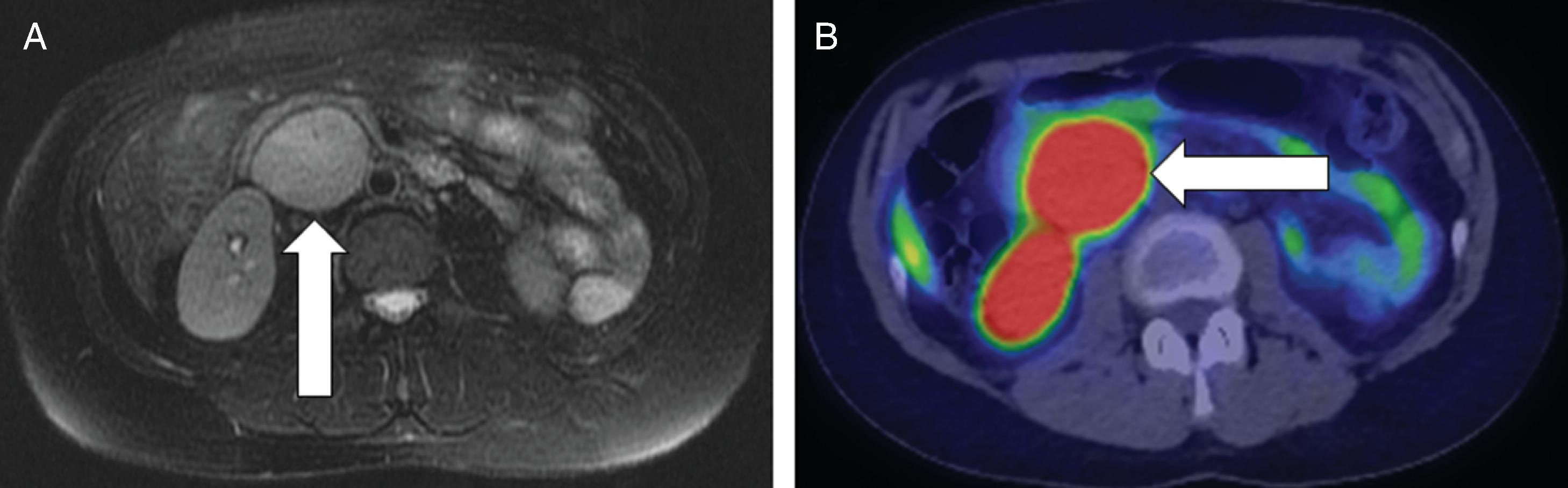
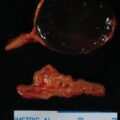
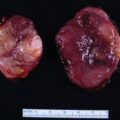
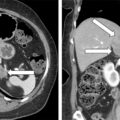
Two weeks postpartum an abdominal MRI with gadolinium redemonstrated the retroperitoneal PGL that was hyperintense on T2-weighted images and had heterogeneous enhancement. Compared to MRI completed during pregnancy, the mass was slightly larger at 4.3 × 5.6 × 6.9 cm ( Fig. 95.2 ). The gallium 68 (Ga-68) 1,4,7,10-tetraazacyclododecane-1,4,7,10-tetraacetic acid (DOTA)-octreotate (DOTATATE)-positron emission tomography (PET)-CT scan showed that the PGL was intensely tracer avid and there was no evidence for metastatic disease or additional PGLs (see Fig. 95.2 ). However, the MRI scan did detect an additional mass along the greater curvature of the stomach near the antrum that measured 2.4 × 2.1 × 1.7 cm ( Fig. 95.3 ). This lesion had no avidity on the Ga-68 DOTATATE PET-CT scan. We suspected that it was most likely a gastrointestinal stromal tumor (GIST), which is associated with PGLs in the setting of succinate dehydrogenase germline pathogenic variants. Although a germline genetic testing panel for pathogenic variants in PPGL susceptibility genes had been obtained, the results were pending.