##
There are six subtypes of primary aldosteronism (PA). The most common subtype of PA is bilateral idiopathic adrenal hyperplasia (IHA) (≈60% of patients) (see Case 8). The second most common subtype of PA, in approximately 30% of patients, is caused by a unilateral aldosterone-producing adenoma (APA) (see Cases 7, 9, 12, 13, 14, and 15). The third most common subtype of PA is unilateral or primary adrenal hyperplasia (referred to as UAH or PAH). The diagnosis of UAH is based on (1) unilateral adrenal localization with adrenal venous sampling (AVS), (2) lack of an adrenal adenoma and the presence of zona glomerulosa hyperplasia on pathology, and (3) long-term cure of PA with unilateral adrenalectomy.
Case Report
The patient was a 65-year-old man who had been hypertensive for 20 years. He had spontaneous hypokalemia for the past 4 years. He was treated with 60 mEq of potassium chloride per day along with five antihypertensive drugs including clonidine (0.3 mg twice daily), doxazosin (8 mg daily), lisinopril (40 mg daily), verapamil (240 mg twice daily), and hydralazine (100 mg three times per day). He had a subarachnoid hemorrhage 6 years previously that left him with left-sided weakness. He had no signs or symptoms of Cushing syndrome.
INVESTIGATIONS
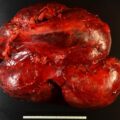
The baseline laboratory test results are shown in Table 10.1 . The patient had positive case detection testing for PA with a plasma aldosterone concentration (PAC) >10 ng/dL and plasma renin activity (PRA) <1.0 ng/mL per hour. Because the patient had spontaneous hypokalemia and a PAC >20 ng/dL, formal confirmatory testing was not needed. , The baseline dehydroepiandrosterone sulfate (DHEA-S) was normal (see Table 10.1 ). An enhanced adrenal-dedicated CT scan showed nodular thickening of the left adrenal gland ( Fig. 10.1 ).
| Biochemical Test | Result | Reference Range |
| SodiumPotassiumCreatinineeGFRAldosteronePlasma renin activityDHEA-S | 1463.81.0>6036<0.6146 | 135–1453.6–5.20.8–1.3>60 mL/min per BSA≤21 ng/dL≤0.6–3 ng/mL per hour12–227 mcg/dL |

After a thorough discussion with him, the patient was keen to pursue a surgical cure of hypokalemia and better control of his hypertension on less medication. He understood that a cure of his hypertension with surgery was not a reasonable goal in view of the duration of hypertension of >10 years. ,
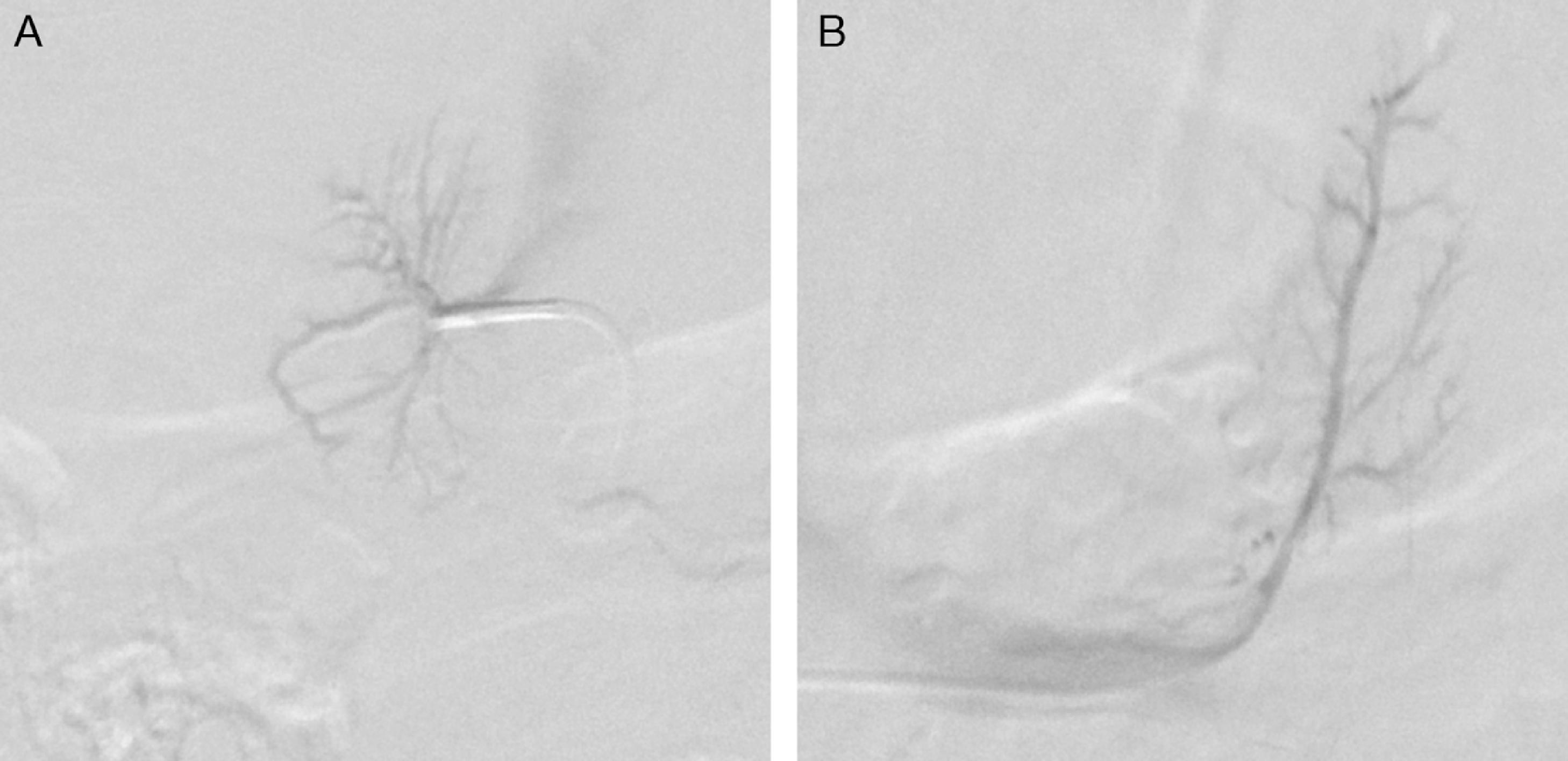
AVS was performed as the next step ( Fig. 10.2 ). AVS was successful based on adrenal vein-to–inferior vena cava (IVC) cortisol gradients of more to 5-to-1 ( Box 10.1 ). , With the continuous cosyntropin infusion protocol (cosyntropin 50 mcg/h administered intravenously starting 30 minutes before AVS and continued throughout the procedure), the adrenal-to-IVC cortisol gradients are typically well above the 5-to-1 cutoff (in this case, 55.5-to-1 on the right and 30-to-1 on the left). Each adrenal vein aldosterone (A) concentration is divided by the respective cortisol (C) concentration for the A/C ratio (see Box 10.1 ). The A/C ratio from the dominant adrenal is divided by the A/C ratio from the nondominant adrenal to determine the aldosterone lateralization ratio. In this case, an A/C ratio of 13.8 on the left is divided by an A/C ratio of 0.5 on the right, yielding an aldosterone lateralization ratio of 25.5-to-1 (left to right). When the aldosterone lateralization ratio is >4-to-1, unilateral adrenalectomy will be curative. , It is also reassuring to confirm relative suppression of aldosterone secretion from the nondominant adrenal by dividing the A/C ratio from the nondominant adrenal by the A/C ratio from the IVC. , In this case, the A/C ratio of 0.2 on the right was divided by 2.4 from the IVC, yielding a value of 0.08. Contralateral adrenal suppression is confirmed when this value is <1.0.