General Overview and Incidence
Incidence varies depending on the institution, patient population, allograph types, and immunosuppression regimes.
Etiology/Histopathology
Clinical settings causing immune dysfunctions, such as viral Epstein-Barr virus (EBV) infection, human viral immunodeficiency, autoimmune disease, and posttransplantation, can cause clonal B- and T-cell lymphoid proliferations, effacing and altering the normal histology architecture and causing symptoms and mass lesions.
Clinical Presentation
- •
Fatigue
- •
Pain
- •
Mass lesions
- •
Ulcers
- •
Headache
- •
Neurologic symptoms
- •
Gastrointestinal symptoms
- •
Dermatologic lesions
- •
Organomegaly: enlarged lymph nodes, spleen, liver
Laboratory Manifestations
- •
EBV serologies
- •
Aberrant CD4/CD8 T-cell subsets
- •
Heavy- and light-chain serum urine immunofixation
- •
Free light chains
- •
Lactate dehydrogenase
- •
Anemia
- •
Thrombocytopenia
- •
Leukopenia
- •
Hypercalcemia
- •
Pancytopenia
- •
Abnormal liver function tests
Diagnostic/Pathologic Entity and Case Examples
- •
Reactive hyperplasia with plasmacytosis ( Fig. 16.1 )

Fig. 16.1
(A) A 57-year-old patient had human immunodeficiency virus/AIDS with retroperitoneal adenopathy. The needle core biopsy specimen shows necrosis and lymphoplasmacytic proliferation at low power. (B) High-power view shows an exuberant plasma cell proliferation; on immunohistochemical study CD138, κ, and λ shows polyclonal plasmacytosis. (C) High-power view shows karyorrhexis, apoptosis, necrosis, and histiocytosis. (D, E) Acute inflammation and histiocytosis favorid a reactive inflammatory process; a diagnosis of lymphoma could not be made.
- •
Infectious mononucleosis like lymphoid hyperplasia ( Figs. 16.2 and 16.3 )
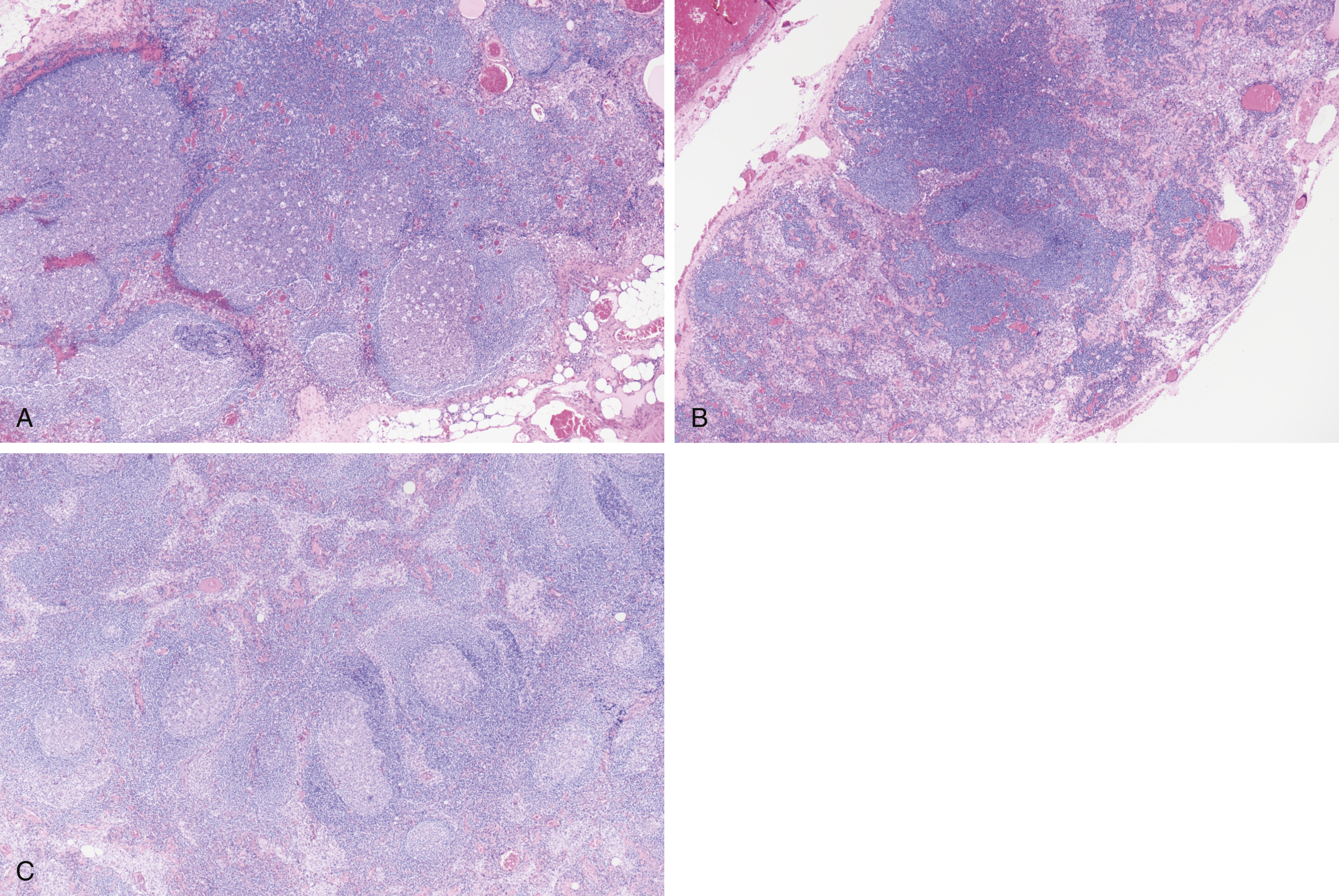
Fig. 16.2
(A) A 36-year-old male presented with prominent external iliac lymph node that showed prominent lymphoid hyperplasia; back to back germinal centers with retained mantle zones and starry sky pattern. (B) Other areas show focal regressed follicle centers, open sinuses, and focal paracortical expansion. (C) Variably sized germinal centers with retained mantle zones, polarized germinal centers with tingible body macrophages or starry sky pattern, and open sinuses. Overall, in the clinical setting of history of renal transplant and human immunodeficiency, the lymph node shows lymphoid hyperplasia, focally florid follicular; other areas show regressed follicles, subtle onion skinning, open sinuses, focal open small vessels, and benign plasmacytosis. Immunohistochemical study shows retained architecture with CD20, CD3, moderate CD138 + polyclonal plasmacytosis with κ and λ light chains, and MIB-1/Ki67 in a reactive pattern. In situ hybridization shows rare small scattered EBER + ; human herpesvirus negative; immunoglobulin M scattered cells positive. Fluorescence in situ hybridization showed disomy (normal copy number) for two tested loci. Cells were BIRC3 − /MALT1 fusion negative. Rearrangements of the MALT1 gene were not detected.
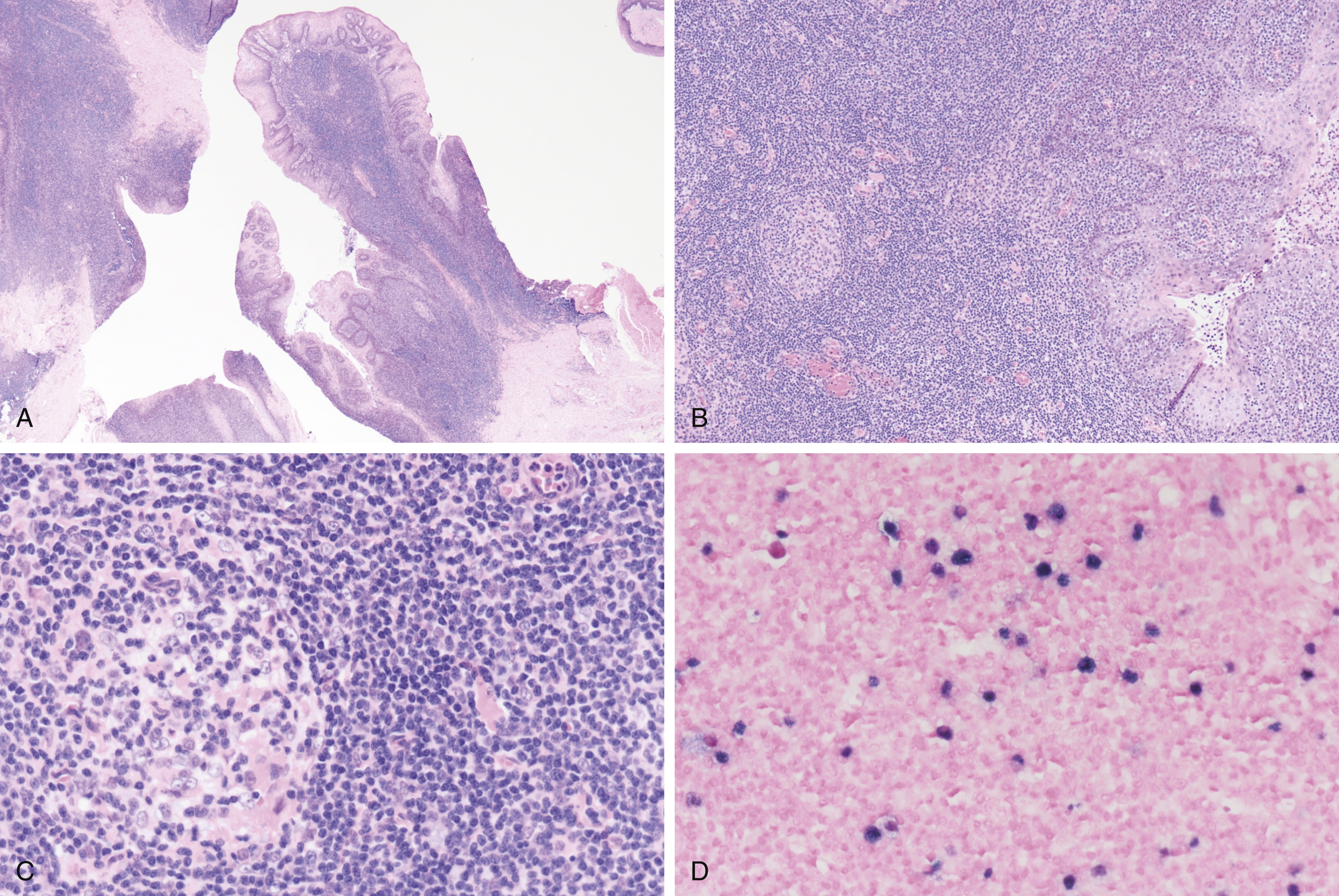
Fig. 16.3
(A) A 19-year-old with a history of kidney transplant (living unrelated donor) showing tonsil biopsy specimen. Tonsillar and adenoidal tissue with B-cell follicles show atretic germinal centers. Results of special stains/immunoperoxidase stains (performed on parts B, C) are as follows: CD2, CD3, CD5, and CD7 are positive in abundant paracortical small T lymphocytes; CD4 + /CD8 + ratio estimated as 3:1. B lymphocytes of primary follicles, secondary follicles with atretic germinal centers, and focally scattered within paracortex, are CD20 weak + , CD79a weak + , PAX5 + . Germinal center B lymphocytes (CD10 + , BCL6 + ) are diminished and do not clearly co-express BCL2. CD23 is strongly positive in follicular dendritic cell meshworks of B-cell follicles (also positive for CD21/35). CD30 is positive in scattered immunoblasts, which are negative for CD15. Scattered CD138 + plasma cells are polytypic by labeling for κ and λ. Proliferation index, overall, is approximately 20% by labeling for Ki67/MIB-1. (B,C) Tonsillar and adenoidal tissue with B-cell follicles showing atretic germinal centers. (D) There are scattered moderately abundant lymphocytes positive for EBER (Epstein-Barr virus–encoded ribonucleic acid).
- •
Polymorphous lymphoproliferative disorder ( Fig. 16.4 )
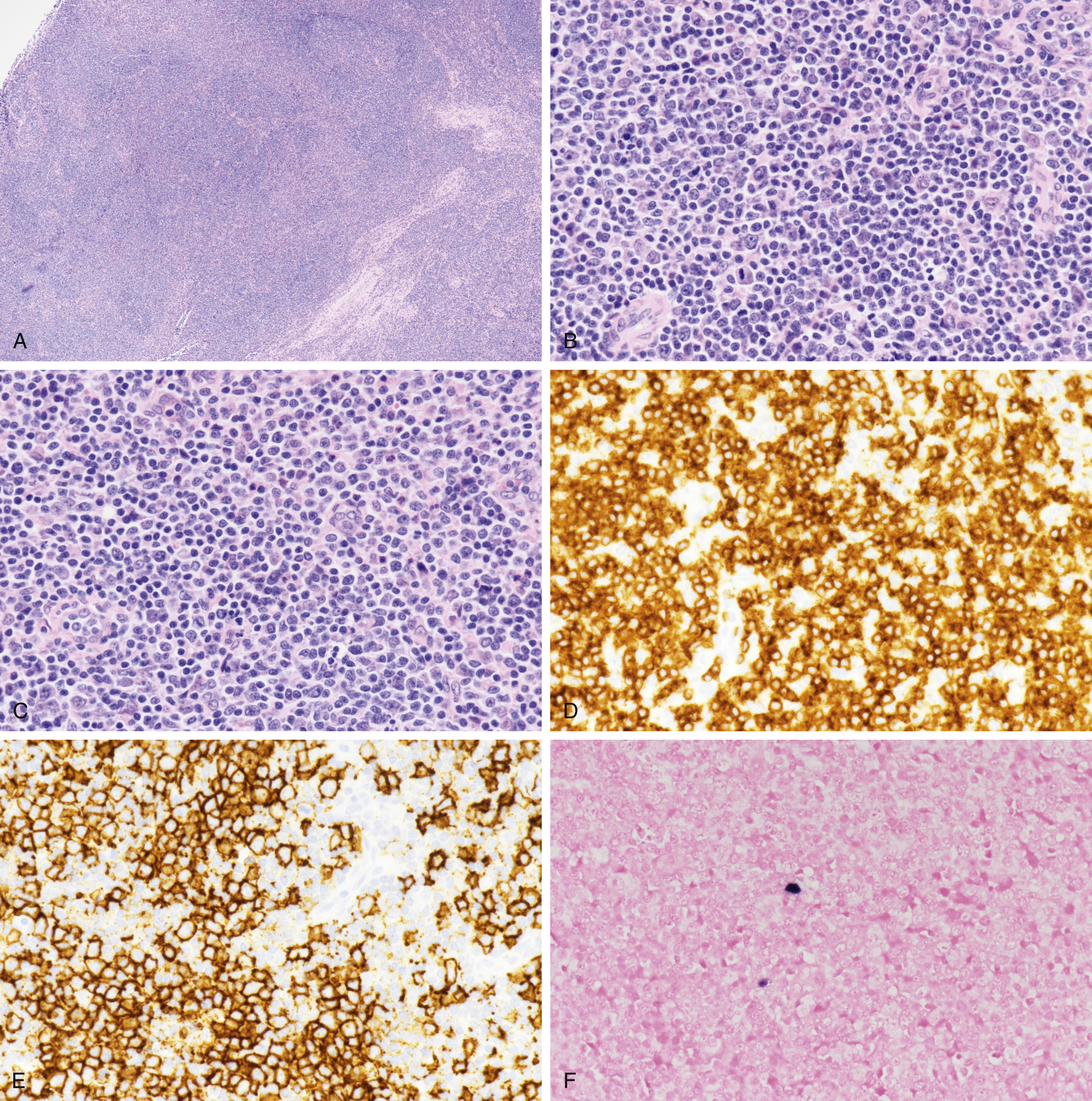
Fig. 16.4
(A) An 11-year-old had polymorphous B-cell lymphoproliferative disorder in the setting of primary immunodeficiency disorder (hyper–immunoglobulin M/combined immunodeficiency), with clonal B cells by flow cytometry findings. Tissue biopsy shows an effaced architecture, with a vague nodular growth pattern, and no open sinuses or few to absent normal germinal centers seen. Lymphoid cells are predominantly small with a few rare larger cells noted. Thickened capsule, scattered mature plasma cells, and focal monocytoid cells were noted. No overt Reed-Sternberg cells or sheets of large cells seen. Ancillary workup by immunohistochemistry showed the following: follicles with germinal centers positive for CD20, CD79a, CD23, and BCL6 ; T cells were positive for PD1, CD43, CD3, CD5, CD4, CD8, BCL2 , CD138, with MUM1-positive plasma cells, polyclonal for κ and λ. B cells by flow cytometry were κ + . Scattered CD30 immunoblasts were positive, MYC rare scattered cells were overall negative. MIB-1/Ki67 were low in paracortical areas and higher in irregular follicles; CD21/CD35 irregular expanded follicular dendritic cell meshworks. Epstein-Barr virus in situ hybridization shows scattered small lymphoid cells positive; human herpesvirus negative. Molecular immunoglobulin (IG) IgH/IgK, TCR κ/λ, TCR γ/δ − . The FoundationOne Heme report showed the following: no genomic alterations; tumor mutation burden low; 2 Muts/Mb; VUS ERBB3 G1271S TYK2 A1004G FANCA A624G FANCE Q95R LRRK2 splice site 347_347+2AGT>G GC MLL2 R755_P763del NCOR2 K876R. (B, C) Higher-power view shows polymorphous lymphocytes. (D) CD3 shows numerous small T cells. (E) CD20 shows B cells, some small, few intermediate to larger in size. (F) In situ hybridization shows rare small EBER-1 + positive lymphocytes.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree