PAPILLARY THYROID CARCINOMA
Part of “CHAPTER 40 – THYROID CANCER“
PREVALENCE
Thyroid carcinoma is classified into four major types, which in decreasing order of frequency are papillary, follicular, medullary, and anaplastic carcinomas. Papillary and follicular carcinomas—often termed differentiated thyroid carcinomas—arise from follicular cells, synthesize thyroglobulin (Tg), and tend to be slow growing. Medullary carcinoma, which originates from thyroidal C cells that secrete calcitonin, may be sporadic or familial. Anaplastic carcinoma usually arises from well-differentiated thyroid tumors and is almost invariably fatal within a brief period (see Chap. 41).
Papillary thyroid carcinoma accounts for ˜80% of all thyroid cancers in the United States,11 including those induced by radiation.12 It is diagnosed in ˜1 in 17,000 people in the United States annually, although occult microscopic papillary thyroidcarcinoma is found in 10% or more of autopsy and surgical thyroid specimens from men and women throughout adult life.13 In contrast, clinically manifest papillary thyroid carcinoma has a peak incidence in the fourth decade and is three times more frequent in women than in men. In children, the sex ratio is nearly equal, which may reflect radiation-induced disease.
FAMILIAL PAPILLARY CARCINOMA
Approximately 5% of papillary carcinomas are familial tumors, which are sometimes inherited as an autosomal dominant trait.14,15,15a Others are inherited as a component of familial adenomatous polyposis (Gardner syndrome), occurring at a young age as bilateral, multicentric tumors with an excellent prognosis, particularly those with ret-PTC activation.16,17 Cowden disease is an autosomal dominant syndrome characterized by multiple mucocutaneous hamartomas, keratoses, fibrocystic breast disease or breast cancer, and well-differentiated thyroid cancer.18,19 Carney complex—a syndrome with spotty skin pigmentation, myxomas, schwannomas, and multiple neoplasia that affects multiple glands—also includes thyroid carcinoma.20
PATHOLOGY
Gross Tumor.
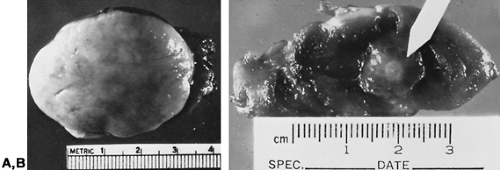
Papillary carcinoma usually is an unencapsulated and invasive tumor with ill-defined margins that is 2 to 3 cm when diagnosed, but this varies widely (Fig. 40-1A).3 Approximately 10% extend through the thyroid capsule into surrounding neck tissues, and another 10% are fully encapsulated.21 They typically are firm and solid, but some develop chronic hemorrhagic necrosis, yielding a thick brownish fluid on needle biopsy that may be mistaken for a benign cyst.22 Small tumors as large as 1.0 cm that often have a stellate appearance, termed microcarcinomas, usually are found by serendipity but rarely are invasive or metastasize (see Fig. 40-1B).23
Architectural Features.
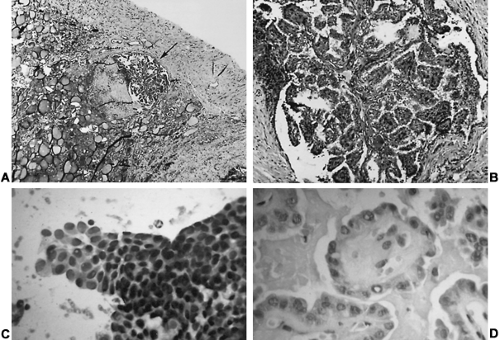
Most papillary carcinomas contain complex branching papillae with a fibrovascular core covered by a single layer of tumor cells, intermingled with follicular structures (Fig. 40-2A), although some have a pure follicular or trabecular appearance.24 The term mixed papillary–follicular carcinoma has no clinical value because the follicular component does not alter 131I uptake or prognosis.25 Moreover, a tumor’s nuclear features are more important than its architectural appearance in establishing a diagnosis of papillary carcinoma. Those with cellular features of papillary carcinoma but a pure follicular pattern are termed follicular variant papillary carcinoma (see Fig. 40-2D), which some argue comprise most tumors diagnosed as follicular carcinoma.26
Cytology Features.
Papillary carcinoma has cellular features that distinguish it from other thyroid neoplasms regardless of its architecture, permitting an accurate diagnosis by fine-needle aspiration (FNA) (see Fig. 40-2C). The cells are large and contain pink to amphophilic finely granular cytoplasm and
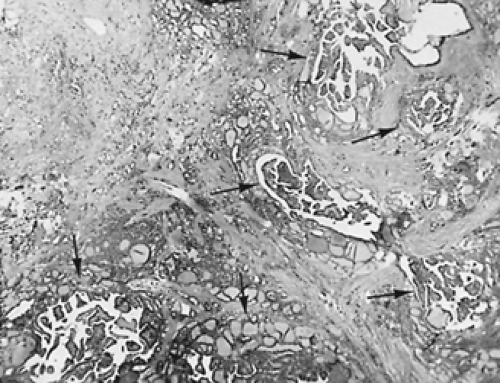
large pale nuclei (“Orphan Annie eye” nuclei) with inclusion bodies and nuclear grooves that identify the tumor as papillary carcinoma (see Fig. 40-2D). Psammoma bodies (Fig. 40-3)—the “ghosts” of infarcted papillae that are virtually pathognomonic of papillary carcinoma—are calcified, concentric lamellated spheres found in approximately half the cases within or near the tumor and lymph nodes or in cytology specimens.27 Lymphocytic infiltration—ranging from focal areas of lymphocytes and plasma cells to classic Hashimoto disease (see Chap. 46)—is often seen in papillary carcinoma. Papillary carcinomas are commonly found in multiple sites within the thyroid gland, which are generally thought to be intraglandular metastases. Multiple microscopic tumor foci are found in as many as 80% of cases when the gland is examined in considerable detail, but in routine clinical practice their frequency is as high as 45%, and they usually are bilateral (Fig. 40-4).11
large pale nuclei (“Orphan Annie eye” nuclei) with inclusion bodies and nuclear grooves that identify the tumor as papillary carcinoma (see Fig. 40-2D). Psammoma bodies (Fig. 40-3)—the “ghosts” of infarcted papillae that are virtually pathognomonic of papillary carcinoma—are calcified, concentric lamellated spheres found in approximately half the cases within or near the tumor and lymph nodes or in cytology specimens.27 Lymphocytic infiltration—ranging from focal areas of lymphocytes and plasma cells to classic Hashimoto disease (see Chap. 46)—is often seen in papillary carcinoma. Papillary carcinomas are commonly found in multiple sites within the thyroid gland, which are generally thought to be intraglandular metastases. Multiple microscopic tumor foci are found in as many as 80% of cases when the gland is examined in considerable detail, but in routine clinical practice their frequency is as high as 45%, and they usually are bilateral (Fig. 40-4).11
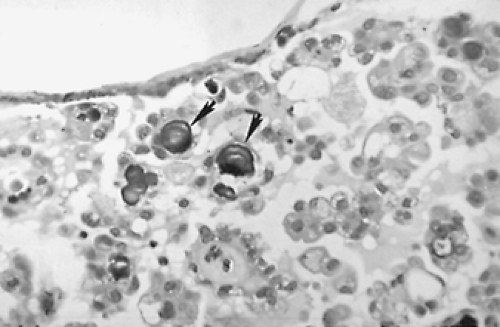 FIGURE 40-3. Psammoma bodies of papillary carcinoma (arrows) are calcified, dark-staining, lamellated spheres that are virtually pathognomonic of papillary thyroid carcinoma. ×400 |
Lymph Node Metastases.
Papillary carcinoma commonly metastasizes to lymph nodes in the lateral neck, central neck compartment, and mediastinum. Gross lymph node metastases are present in approximately half the cases at the time of diagnosis, while even more—as many as 85% in studies from Japan28,29— have microscopic nodal metastases.25 Their number rises as primary tumor size increases. Nodal metastases tend to be bilateral when the isthmus or both thyroid lobes are involved with tumor, or they may extend into the mediastinum or extend beyond the lymph node capsule into soft tissues, which are all poor prognostic signs.25,30
Distant Metastases.
Fewer than 5% of patients have distant metastases at the time of diagnosis, and another 5% develop them later.11 In a review of 1231 patients with distant metastases,
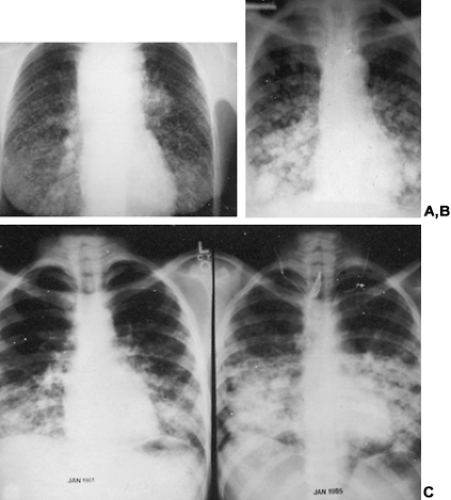
49% were in the lung, 25% were in bone, 15% were in lung and bone, and 12% were in the central nervous system or in multiple organs.11 Pulmonary metastases may be large discrete nodules or may have a “snowflake” appearance caused by diffuse lymphangitic tumor spread (Fig. 40-5A, Fig. 40-5B and Fig. 40-5C). Pulmonary metastases not seen on radiographs may be detected only with whole body scanning done after therapeutic doses of 131I have been administered.31,32,33 and 34
49% were in the lung, 25% were in bone, 15% were in lung and bone, and 12% were in the central nervous system or in multiple organs.11 Pulmonary metastases may be large discrete nodules or may have a “snowflake” appearance caused by diffuse lymphangitic tumor spread (Fig. 40-5A, Fig. 40-5B and Fig. 40-5C). Pulmonary metastases not seen on radiographs may be detected only with whole body scanning done after therapeutic doses of 131I have been administered.31,32,33 and 34
Thyroglossal Duct.
Papillary thyroid carcinoma arising within a thyroglossal duct is almost always small and usually has a benign course.35
Papillary Microcarcinoma.
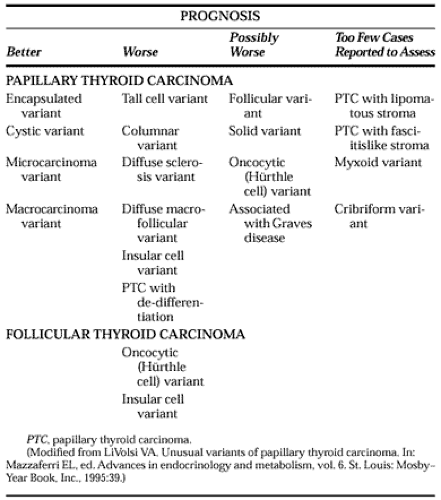
Different histologic variants or subtypes of papillary carcinoma have distinct biologic behaviors (Table 40-1). Five histologic subtypes or variants were recognized in the 1988 World Health Organization classification,24 and at least five others have been described since then.36,37 Papillary microcarcinoma is a tumor 1.0 cm or smaller. Approximately 20% are multifocal, and as many as 60% have cervical lymph node metastases,29 which may be their presenting feature.23 Lung metastases occur rarely with multifocal tumors that have bulky cervical metastases; these are the only micro-carcinomas with significant morbidity and mortality.23,37 Otherwise, the recurrence and cancer-specific mortality rates are near zero.13,23
Encapsulated Papillary Carcinoma.
Encapsulated papillary carcinoma is an otherwise typical tumor completely surrounded by a fibrous capsule. It accounts for ˜10% of papillary carcinomas and is approximately half as likely to metastasize as is typical papillary carcinoma.37 Tumor recurrence is lower than usual, and death that is due to cancer almost never occurs with these tumors.27,37
Follicular Variant Papillary Carcinoma.
Follicular variant papillary carcinoma accounts for ˜10% of papillary carcinomas.37 Usually not encapsulated, its microfollicular architectural pattern is otherwise indistinguishable from follicular carcinoma, but the typical nuclear features of papillary carcinoma identify its true nature, which can be diagnosed by FNA cytology.27 Its metastases often have psammoma bodies and may show the typical features of papillary carcinoma. Some claim that its prognosis is similar to the usual papillary carcinoma, and others suggest that distant metastases are more likely and that long-term outcome may be unfavorable with this tumor.36,37 and 38
Diffuse Macrofollicular Variant Papillary Carcinoma.
Diffuse macrofollicular variant papillary carcinoma is an uncommon tumor that can be confused with goiter or macrofollicular adenoma on frozen section.36 It occurs predominantly in women with goiter, approximately one-third of whom have hyperthyroidism. Most have distant metastases with very high mortality rates.36
Tall Cell Variant.
Approximately 10% of papillary thyroid carcinomas show extensive papillae formation with cells twice as tall as they are wide that comprise at least 30% of the tumor.36 Compared with typical papillary carcinoma, tall cell tumors tend to be diagnosed approximately two decades later (in the mid-50s), are larger tumors associated with significantly more invasion into extrathyroidal soft tissues, and have more distant metastases.18,36,37 The tumor, which can be identified on a cytology specimen from fine-needle biopsy, often expresses the p53 suppressor oncogene, perhaps accounting for its frequent loss of 131I uptake and long-term mortality rates that are two- to threefold those of typical papillary carcinomas.18,37
Columnar Cell Variant.
Columnar cell variant is a rare variant possibly related to tall cell carcinoma. It occurs mainly in men and is composed of rectangular cells with clear cytoplasm.36 More than 90% develop distant metastases, which usually are unresponsive to 131I therapy or chemotherapy and result in death.36,37 When encapsulated, it has a much better prognosis.21
Diffuse Sclerosis Variant.
Approximately 5% of papillary carcinomas are of the diffuse sclerosis variant type, which is even more common in children and adolescents.36,37 Approximately
10% of the tumors in the children of Chernobyl are of this type.39 Usually involving both lobes, diffuse sclerosis variant presents as a goiter with extensive squamous metaplasia, sclerosis, abundant psammoma bodies, and lymphatic invasion involving the whole thyroid gland. Lymph node metastases are almost always present, and ˜25% have lung metastases.36,37 Cytology specimens reveal squamous metaplasia, inflammatory cells, and psammoma bodies, but may be difficult to differentiate from thyroiditis. Although it has a higher incidence of local and pulmonary metastases than typical papillary carcinomas, there is some disagreement about whether its long-term prognosis is worse than usual.36,37
10% of the tumors in the children of Chernobyl are of this type.39 Usually involving both lobes, diffuse sclerosis variant presents as a goiter with extensive squamous metaplasia, sclerosis, abundant psammoma bodies, and lymphatic invasion involving the whole thyroid gland. Lymph node metastases are almost always present, and ˜25% have lung metastases.36,37 Cytology specimens reveal squamous metaplasia, inflammatory cells, and psammoma bodies, but may be difficult to differentiate from thyroiditis. Although it has a higher incidence of local and pulmonary metastases than typical papillary carcinomas, there is some disagreement about whether its long-term prognosis is worse than usual.36,37
Oxyphilic (Hürthle Cell) Variant.
Approximately 2% of papillary carcinomas have nuclei resembling those of Hürthle cell follicular carcinomas.40 Multiple oxyphilic thyroid tumors and a familial occurrence have been noted in some cases.41 This tumor cannot be identified by FNA cytology but is recognized by its papillary architecture on the final histologic sections. Compared with typical papillary carcinomas, oxyphilic carcinomas have fewer neck nodal metastases at diagnosis but have higher rates of recurrence and cause-specific mortality; in this respect, they resemble oxyphilic follicular carcinoma.37,40
Solid or Trabecular Variant.
A tumor with a predominantly (>75%) solid architectural pattern that maintains the typical nuclear features of papillary carcinoma, the solid or trabecular variant has a propensity for extrathyroidal spread and lung metastases.36,37 Some, however, find it to be more common in children and report that its prognosis is the same as with typical papillary carcinoma.42
Insular Carcinoma.
Approximately 5% of all thyroid carcinomas show solid clusters of cells with small follicles that contain Tg but resemble pancreatic islet cells. This is often categorized as a variant of follicular carcinoma, but it may show papillary differentiation, and it has been suggested that it should be considered a separate entity derived from follicular epithelium.43 Insular carcinomas usually are large and highly invasive tumors that grow through the tumor capsule and into tumor blood vessels. Compared with differentiated thyroid carcinoma, insular carcinoma presents at an older age (54 vs. 36 years); with larger tumors (4.7 vs. 2.5 cm); with fewer neck metastases (36% vs. 50%) but more distant metastases (26% vs. 2%); and has a worse 30-year cancer-specific mortality rate (25% vs. 8%).18 Insular carcinoma also displays aggressive behavior in children.44
DIAGNOSIS
History.
In the past, many papillary carcinomas were large and invasive when first diagnosed; however, with the use of FNA, most are small tumors found at an early stage as an asymptomatic thyroid nodule on routine examination or in screening programs of patients with head and neck irradiation.3 The timeliness of diagnosis affects the prognosis (see Prognosis).3 Occasionally, it causes pain, hoarseness, dysphagia, or hemoptysis, or it infiltrates surrounding structures or grows rapidly—findings that are associated with high mortality rates. However, only a few patients have such symptoms. A history of irradiation is important, but only 30% of patients develop palpable thyroid nodules after irradiation, and of these, only one-third are malignant.45 Thyroid nodules that appear after radiation should undergo an evaluation similar to that used for nodules that occur spontaneously.
Physical Examination.
Papillary carcinoma usually is manifest as a palpably discrete thyroid nodule that moves upward when the patient swallows, but a cervical lymph node metastasis may be its only sign. A midline mass above the thyroid isthmus may be a metastatic lymph (Delphian) node or may be carcinoma within a thyroglossal duct that can be identified from its upward movement when the tongue is protruded. Papillary carcinoma may be a firm, nontender, discrete mass, but many are soft and cystic or diffusely infiltrate one lobe or the entire thyroid. Distant metastases are found less often at the time of diagnosis of papillary carcinoma than follicular carcinoma, but when they are found, the primary tumor is almost invariably large (see Fig. 40-1A and Fig. 40–5B).
Fine-Needle Aspiration.
Approximately 10% of thyroid nodules show clear evidence of malignancy, such as vocal cord paralysis, signs of invasion, or bulky lymph node metastases, whereas the others appear benign on examination. Neither the history nor the physical examination offers sufficient evidence of a nodule’s benign nature that further testing can be deferred (see Chap. 34, Chap. 35 and Chap. 39). The first test in a clinically euthyroid patient is FNA.46 Other tests are too nonspecific to be used first. Except for hyperfunctional (“hot”) nodules that are rarely malignant, most thyroid nodules—whether single in an otherwise normal gland or a dominant nodule in a multinodular goiter—require FNA for diagnosis. Although large-needle cutting biopsies can be done, FNA is considerably safer and is highly effective in obtaining sufficient cytology to identify the distinctive features of papillary carcinoma, including most of its variants46 (see Chap. 39). Nodules that yield diagnostic or suspicious cytology are excised; the others are scanned to exclude hot nodules, whereas benign nodules are simply observed or sometimes treated with thyroxine-suppressive therapy. Thyroxine suppression should not be done without prior FNA and should not be used as a diagnostic test, because thyroid carcinomas may appear to shrink.46
Prior Neck Irradiation.
FNA should be the first test performed on a palpable thyroid nodule in a euthyroid patient with a history of head-and-neck irradiation. An asymptomatic patient with a palpably normal thyroid gland who has history of head and neck irradiation should not undergo ultrasonography because nearly 90% of such patients have thyroid nodules found by this test, most of which are benign.9,47
Ultrasonography reveals thyroid lesions <1 cm in ˜50% of the healthy middle-aged population.48,49 Thyroid hormone does not prevent the appearance of thyroid nodules or cancer in previously irradiated patients with a palpably normal thyroid gland,45 but it does reduce the recurrence of nodules in irradiated patients who have undergone thyroid surgery for benign thyroid nodules.50
Ultrasonography reveals thyroid lesions <1 cm in ˜50% of the healthy middle-aged population.48,49 Thyroid hormone does not prevent the appearance of thyroid nodules or cancer in previously irradiated patients with a palpably normal thyroid gland,45 but it does reduce the recurrence of nodules in irradiated patients who have undergone thyroid surgery for benign thyroid nodules.50
FACTORS INFLUENCING PROGNOSIS
Once the diagnosis is established, the prognosis is determined by an interaction of three variables: tumor stage, the patient’s age, and therapy. The cancer-specific mortality rate is <10% over three decades, but distant metastases or serious local recurrences can occur many years after initial therapy.3,11
Delay in Diagnosis.
The author of this chapter has found that the median time from the first manifestation of thyroid cancer—nearly always a neck mass—to initial therapy was 4 months in patients who survived and 18 months in those who died of cancer (P <.001).3 The 30-year cancer mortality was nearly twice as high when therapy was delayed for longer than a year than when it was done within 12 months of a nodule’s discovery (13% vs. 6%, P <.001).3
Age at Time of Diagnosis.
Age is the most important prognostic factor. The adverse effect of age appears at ˜40 years and becomes progressively worse thereafter, increasing at a steep rate after age 60 years, when men have the worst prognosis.3,11 The response to therapy is most favorable in younger patients whose tumors concentrate 131I.51,52 and 53
Prognosis in Children.
Outcome is usually favorable in children, although their tumors are typically at a more advanced stage, with more local and distant metastases than are those of adults at the time of diagnosis.51,53,54 and 55 Recurrence rates in children are ˜40% over several decades compared with 20% in adults.3,53 The rate of pulmonary metastases in children is almost twice that in adults, reaching more than 20% in some series.3,11,54 Their prognosis for survival is, however, excellent, with or without a history of irradiation, except for those younger than age 10 years, who have high mortality rates.51,52 and 53 Nonetheless, despite the relatively good prognosis in children, one study found that children with thyroid cancer had, as a group, poorer long-term survival rates than did normal children.56
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree