General class
Mechanism
Examples
Conventional cytotoxic drugs
Platinating DNA damaging agents
Cisplatin
Cyclophosphamide
Bleomycin
Microtubule agents
Vinblastine
Paclitaxel
Antimetabolites
Gemcitabine
5-fluorouracil
Targeted drugs
Cell surface and intracytoplasmic tyrosine kinase enzymatic inhibition
Pazopanib (VEGFR)
Afatinib (EGFR)
DNA repair enzyme inhibition
Olaparib (PARP)
NCI–MATCH trial
Various (over 30)
Immunotherapy
General immune
Nivolumab, pembrolizumab, atezolizumab
Modulation: Checkpoint inhibitors (PD-1, PD-L1, CTLA-4, others)
Ipilumimab
Specific to HPV
E6 HPV vaccine
CAR-T cells
Chemotherapy: A Sparse History
Cisplatin
The sensitivity of squamous cancer to platinum drugs, that is to the say the potential for significant, major regressions, has been observed for decades. Cisplatin-based therapy, mostly in combinations with medications for which synergistic efficacy had been already demonstrated in cancers with higher incidences than for penile cancer, is the subject of several single-arm series. The mechanism of action for cisplatin and many other conventional cytotoxic drugs depends on introducing single-strand, or for others double-strand breaks and other lesions in DNA, and then thus initiating apoptosis. Other conventional type cytotoxic drugs can include those that affect nucleic acids at the point of, or before the point of incorporation, such as 5-fluoruracil or methotrexate. The taxanes and vinca alkaloids are class of medication that bind tubulin, and so affect microtubules; defects of mitotic spindle function then do also favor an apoptotic process. None of these mechanisms would be considered unique to squamous cancer.
The history of the application of cisplatin use in squamous cancer is as early as 1979, when Sklaroff and Yagoda published a penile cancer series, with one complete response (CR) and two partial responses (PR; duration 2 and 7 months) among eight patients [1]. While not all patients have a practical organ reserve for this medication, these series serve as basis for treatment planning for many patients. The factors that can be prohibitive for cisplatin use—and this would not be represented in these series—can include performance status and comorbid burden, renal function, neuropathy, and marrow reserve.
The next series using a monotherapy of cisplatin, with a schedule of 50 mg/m2/dose on days 1 and 8 of a 28-day schedule, was organized through the Southwest Oncology Group (SWOG) [2]. Only 4 of 26 patients had partial response (PR), and with duration of only up to 3 months. The practical issue for cisplatin therapy has evolved considerably since that era with innovations including understanding the importance of careful pre- and post-hydration with each dose, a depth of experience with dosing regimen and combinations, neutrophil growth factors, and anti-emetic regimens incorporating 5-HT3 antiemetics (such as ondasetron, granisetron, or palonosetron) as well as NK1 (neurokinin1) antiemetics (such as aprepitant or fosaprepitant; or rolapitant).
Earlier Cisplatin Combinations
The earliest published report is a 1990 report by Hussein and colleagues who described combination therapy , adding 5-fluorouracil (5-FU) to cisplatin, in a series of six patients (apparently earlier stage than the prior series), and finding of one complete response (CR) and five PRs, including patients then resected or treated with radiation therapy to no evidence of disease [3]. The cisplatin was dosed at 100 mg/m2 on day 1, and the 5-FU was then over 96 h starting on the second day. A report with a similar schedule, from Europe in 1992 [4] also described responders, with PR in two of eight. A more contemporary retrospective review about 25 patients was presented by Di Lorenzo and colleagues in 2012. In this series, encompassing four institutions and a decade, none had CR; eight had PR; additional ten were designated as stable [5].
A different combination approach, for which three responses were seen among 14 patients, some of whom had tumor ulceration, was described by Dexeus and colleagues from MD Anderson in 1991, with cisplatin and bleomycin intravenously (11) or intra-arterially (3) and addition of intravenous methotrexate [6]. In 1993 Kattan and colleagues also described 3 responders, among 13 patients with penile cancer in France, who had had combination treatments all of which included cisplatin; of still contemporary interest, this was 12 years’ experience, reflecting the practical difficulty for making conventional prospective, fixed-regimen format trial for studying this diagnosis [7]. A single-arm cooperative group study (cisplatin 75 mg/m2/dose, every 21 days and methotrexate and bleomycin on days 1 and 8) from SWOG was reported by Haas et al. in 1999 and described that among 45 penile squamous cancer patients, of whom 40 were evaluable had been observed five CR and eight PR, but also five treatment relate deaths, and additional six patients with at least one life-threatening toxicity event [8]. Thus, while the experience with the triplet did reproducing anticancer responses (32.5 %) seen in the prior smaller series, the key limitation of escalated intensity, a key factor in personalized care, was brought to the fore.
Taxanes
Already a decade a fter the routine use in non-small cell lung cancer, breast cancer and other settings, paclitaxel activity in the post-cisplatin setting was described with 3 of 12 partial responses in a monotherapy prospective trial, by Di Lorenzo et al. [9]. Taxane combinations have been developed further for penile cancer therapy, notably in the TIP (paclitaxel [Taxol®] ifosfamide cisplatin) and the TPF (docetaxel [Taxotere®] cisplatin 5-fluorouracil) combinations; the paclitaxel cisplatin-5-FU combination also may be termed TPF. With reference to cancers that are the subjects for more formal prospective studies, the former has resemblance to some germ cell cancer regimens; the latter to a treatments used in head and neck and esophageal cancers.
Bermejo and colleagues described that in a retrospective review of 59 patients treated at MD Anderson, among those receiving neoadjuvant intravenous chemotherapy ten had then been treated with subsequent surgical resection. Median survival of those with three or fewer positive lymph nodes was 48 months [10]. The same group addressed use neoadjuvant cisplatin (25 mg/m2/dose x3) ifosfamide (1200 mg/m2/dose x3) paclitaxel (175 mg/m2 x1) TIP chemotherapy in 30 patients. Fifteen of the 30 had major response; 22 went on to definitive-intent surgery; among these patients 11 were long-term disease-specific survivors (two died of other causes). Favorable predictive features observed included chemotherapy response, no bilateral residual cancer, no extranodal extension, and no skin involvement [11].
Nicholson and colleagues reported in 2013 on a multicenter 29 patient phase II study of TPF from the UK. Although 10 of 26 evaluable patients (38 %, 95 % CI: 20.2–59.4) had objective response, including two with locally advanced disease having CR, this did not meet the pre-specified target of >14 PR, and two-thirds of the patients had at least one toxicity of at least grade 3 [12]. Another slightly larger prospective study from China is reported by Zhang and colleagues, and described a similar response rate, 15 of 39 patients (38 %, 95 % CI 23–55 %), but still median PFS only 3 months, and median OS 7 months [13].
Irinotecan and Gemcitabine
Among combinations with relatively newer conventional cytotoxic drugs is and irinotecan. Irinotecan is a topoisomerase I inhibitor. A combination series from the EORTC was reported by Theodore and colleagues in 2008, consolidating reports for patients on neoadjuvant treatment (four cycles) or advanced disease treatment (up to eight cycles). They observed two CR and six PR among 26 evaluable patients; this ORR of 31 %, 80 % confidence interval 19–45 %, which failed to exclude the study hypothesis to exclude a response rate below 30 % [14].
Gemcitabine with cisplatin is a dominant consideration for therapy of urothelial cancers, also extends to intra-arterial treatments. Again, the largest base of experience is outside the genitourinary system, in non-small cell lung cancers. Power and colleagues described in 2009, two patients with good palliative impact for intravenous gemcitabine-cisplatin treatment [15]. An intermediate report on a phase II trial presented by Houédé and colleagues gives results for 25 patients treated between 2004 and 2010, with two PR and ten SD responses, and 5.5 months median time to progression—a response rate deemed not high enough to recommend an emphasis for further development [16].
Intravenous or Intra-arterial Therapy
This intra-arterial approach was a part of the 1991 report of Dexeus et al. [6], and is the subject of later series as well. The anatomy of the perineum, particularly being distal to the renal and most of enteric circulation, does allow for potential use of intra-arterial cisplatin or other conventional chemotherapy in a way that bypasses renal arterial first-pass exposure. Roth and colleagues describe the position as a femoral artery approach, with the tip beyond the inferior mesenteric artery, above the aortic bifurcation [17]. Roth and colleagues described the treatment of eight penile cancer patients (as well as eight anal cancer patients, by the same approach), using a five-part combination (cisplatin; 5-FU; methotrexate; mitomycin C; and bleomycin), with three each of penile and anal cancer having CR; and additional partial responses.
Clearly, for most medical oncology contexts, a reliance on intra-arterial therapy does involve coordination of the infusion process with interventional radiologist, for the placement of the intra-arterial catheter, and the administration of the chemotherapy which may be in a setting very different from a typical infusion center. Myelotoxicity or nausea risks would be expected to not be different.
An additional five patients treated in Japan with intra-arterial chemotherapy (same five drugs, one patient had carboplatin instead) were reported by Chen and colleagues. Among these neoadjuvant patients who had refused up-front surgery, were observed one CR and four PR [18]. Overall, however, the experience with this route of administration remains quite limited. Huang et al. reported one CR and two PR among three patients receiving monthly intra-arterial cisplatin methotrexate and bleomycin, reported in 1999 [19].
A Chinese series describing outcomes for 12 subjects with intra-arterial gemcitabine and cisplatin treatment of locally advanced penile cancer, over a period of 12 years, was reported by Liu and colleagues in [20]. Ten had major response, and three were long-term survivors. Overall, despite the long term of the experience, the base of intra-arterial treatments and the logistics remain limiting, although the rationale and particularly the potential for organ preservation do present a key appeal. Further formal study and standardization of protocols to accommodate prospective testing are lacking.
Timing Considerations
While there are few data to guide which chemotherapy to use, notwithstanding the above experiences of a couple hundred patients over a few decades, there are even less data to guide a uniform recommendation on treatment before, during, or after other modalities. A treatment for advanced disease that is metastatic and not potentially surgically resectable to potential cure is the most straightforward medical context; recurrent metastatic disease after surgery is a similar context, although it is logical to define a distinction between small-volume, longer-latency cancers as compared to widely metastatic, rapid early regrowth. Similarly to those patients with a demonstrable metastatic pattern, a penile cancer that at presentation technically unresectable but only apparently locally spread presents a situation for which initial medical therapy (the neoadjuvant schedule) is a relatively rational recommended consideration for initial treatment.
On the other hand, the period time immediately after potentially or apparently definitive resection of the primary cancer is one for which treatment decisions are further less well-defined. Clearly there will be some patients for whom the recurrence risk defined by the [residual] cancer in the resected specimen appears minimal—small volume, single superficial node area, for example. And at the other end of the risk spectrum will be patients (still technically resected to no evident disease) with features such as were identified as adverse by Pagliaro and colleagues [11]: Large volume of nodal disease, bilateral pattern, extranodal pattern, and skin-extension pattern. For the latter group, the balance of risk would be to favor immediate action to escalate treatment intensity, to try to tip the balance to prevent recurrence.
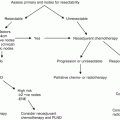
Thus is framed the question of how much disease risk should be present before a recommendation of adjuvant treatment, whether regional radiotherapy, radiotherapy with concurrent chemotherapy, or single- or multi-agent chemotherapy systemic treatment. In a small series (cisplatin/bleomycin/methotrexate) that includes some adjuvant patients, Hakenberg et al. describe the risk of treatment-related death in plain terms [21]. Adjuvant use of cytotoxic chemotherapy in most other cancer has been approached cautiously, recognizing that the incident population represents an uncertain ratio of patients who could not benefit and those who might benefit. (The former include a proportion who would have been cured anyway, or whose disease was absolutely insensitive to the treatment planned, or died first of other causes before the disease in question, or died from treatment-related toxicity; the latter are those for whom the disease is sensitive, and eventual progression is substantially delayed or actually prevented.) The situation for penile cancer therapy is that there are no generalized trial results to interpret. This risk:benefit ratio is left for the judgment of the treating physician and the patient’s own interpretive perspective. The NCCN guidelines identifies a key tipping point for greater than pathological N1 (that is pN2 or pN3; or cN3 [pelvic LN]; or pM1) for adjuvant chemotherapy, or for lymph node dimension over 4 cm for neoadjuvant chemotherapy [22].
Overall, any regimen known to show activity in advanced disease could reasonably be discussed in this adjuvant context; however this adjuvant timing for chemotherapy, as well as the choice of medications, are ones that are not amenable for description as a standardized intervention. Nicolai and colleagues present what is probably the largest specific neoadjuvant and adjuvant experience, using a taxane (paclitaxel or docetaxel), cisplatin and 5-FU for 28 neoadjuvant patients and 19 adjuvant patients with N2M0 or N3M0 pattern of regional spread. Over the 8 years in which this non-randomized series accrued (2004–2012), they describe a practice pattern shift toward neoadjuvant treatment. Among the neoadjuvant treatment patients were recorded 43 % “clinical responses” and 14 % complete pathologic remissions. Response was not associated with survival; however, associations of worse outcomes for N3 disease, and for p53 status, and for bilateral disease were observed [23].
Practically speaking the key decision for pursuing a neoadjuvant approach will be based on the clinical assessment of the extent to which the disease could be completely resected. Conversely, the key decision point on adjuvant therapy, whether as chemotherapy alone or concurrent with radiation therapy, will be on the clinical and pathologic assessment of the extent of remaining or metastatic disease.
Radiation Therapy
Radiation therapy is not per se a systemic treatment, but there are few points that can be mentioned in the interface of radiation treatment and systemic therapies. The first relates to timing in relation to other modalities of treatment. Extrapolating from the experience of other cancer diagnoses, an induction multiagent chemotherapy treatment could be used prior to a definitive-intent radiation therapy approach. While certainly rationale in favor or opposed to this for a given penile cancer, patient could be developed along lines exactly parallel to those for neoadjuvant chemotherapy before surgery; the data for this diagnosis quite limited. Extrapolating from experiences, for example, in head and neck cancer, a phase III trial adding TPF induction before definitive radiotherapy did not improve outcomes [24], although a prior meta-analysis did show better outcomes [25].
The two key interactions of radiation therapy with systemic treatments relate to concurrent use of radiosensitizing chemotherapy, and to the role of local tissue destruction as a potential priming event of an immune response [26, 27]. The former is not the subject of any series particularly devoted to penile cancer, but approaches extrapolated from experiences for therapy of other squamous cancers, such as weekly cisplatin (squamous cervical cancer, e.g. [28]; head and neck cancer, e.g. [29]) or mitomycin and 5-FU (squamous anal cancer, e.g. [30]; muscle invasive urothelial bladder cancer, e.g. [31]).
The latter concept, to leverage tumor and stromal changes from radiotherapy to cause a better anticancer immune response is among many in development in conjunction with checkpoint inhibitor treatments. Some of the latter may ultimately include penile cancer patients not as a separate cohort, but rather as part of a multi-diagnosis immunotherapy development process, particularly in early-phase clinical trials.
Immunotherapy
The potential for anticancer immunotherapy, manifested in contemporary trials for many malignancies, with checkpoint inhibitors and other new technologies, was presaged by penile cancer immunotherapy treatment with interferon and by use of imiquimod on genital warts, an HPV-associated lesion that may be precursor of some penile cancers. Work from Greece, described by Mitrolopous et al. introduced a biologic, immunotherapy partner for cisplatin. Thirteen patients with nonmetastatic cancers had cisplatin treatment (fractionated as 20 mg/m2/daily for five consecutive days) in combination with interferon α treatment. There were nine responders, four of which were CR, and patients were then able to have less extensive, but definitive-intent surgery [32]. Once again cisplatin-based therapy was understood as a part of a strategic multidisciplinary approach. The use of imiquimod is supported through conventional randomized trials leading to an on-label medical therapy of squamous penile lesions, although not actually for penile cancer [33].
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree