Myeloproliferative Neoplasms
Olga Pozdnyakova, MD, PhD
Attilio Orazi, MD
Myeloproliferative neoplasms (MPNs) are a group of clonal disorders of the hematopoietic stem cells that share similar features, such as hypercellular marrow and evolution to acute leukemia (Table 8.1). In addition, these disorders are associated with bone marrow fibrosis either at presentation or upon disease progression. Unlike myelodysplastic syndromes (MDS), MPNs demonstrate absence of bone marrow dysplasia with normal maturation and effective cell production causing elevated blood levels of one or more cell lines (i.e., erythrocytosis, leukocytosis, and thrombocytosis) (Table 8.2). Patients tend to have hepatosplenomegaly (an unusual finding in MDSs) because of sequestered blood cells, extramedullary hematopoiesis, or infiltration by neoplastic cells (Figs. 8.1 and 8.2). However, similar to MDS, MPNs may evolve into acute leukemia, albeit at lower rates. These diseases differ from acute leukemias in the slower onset and more protracted course, measured in months to years for MPNs, compared to weeks for untreated acute leukemias.
MPNs usually occur in middle-aged adults. The clinical and hematologic features of the various MPNs significantly overlap and an accurate diagnosis of a specific MPN subtype requires careful integration of morphologic, cytogenetic/molecular findings and strict adhesion to the WHO criteria for diagnosing of MPNs. Advances in gene expression analysis and next-generation sequencing have led to identification of unique somatic mutations allowing to significantly improve the diagnostic and prognostic criteria for MPNs, which have been reflected in the revised 2016 WHO Classification of Tumors of hematopoietic and Lymphoid Neoplasms1, 2, 3, 4 (Table 8.3). Most MPNs develop through acquired clonal abnormalities in genes encoding cytoplasmic or receptor protein tyrosine kinases that result in constitutively active tyrosine kinase promoting growth and replication through various signal transduction pathways in a cytokine-independent manner.5,6 BCR-ABL1 fusion protein, the result of a reciprocal translocation that gives rise to an abnormal Philadelphia chromosome, was the first tyrosine kinase to define a specific MPN subtype7 and traditionally MPN subtypes have been divided into the Ph chromosome-positive group, comprising chronic myeloid leukemia (CML) as a sole entity, and the Ph chromosome-negative group that includes the remaining MPN subtypes. The classic Ph chromosome-negative MPNs are primary myelofibrosis (PMF), polycythemia vera (PV), and essential thrombocythemia (ET)—three distinct clinical entities which share specific mutually exclusive somatic mutations (JAK2, MPL, and CALR) that result in dysregulation of the JAK/STAT signaling pathway leading to abnormal hematopoiesis, cytokine overproduction, and development of bone marrow fibrosis.3,8, 9, 10, 11 Chronic neutrophilic leukemia (CNL), another rare Ph chromosome-negative MPN, has recently been connected to an activating somatic mutation in the receptor for colony stimulating factor 3 gene (CSF3R), which has been shown to induce constitutive phosphorylation of JAK2, STAT3, AKT, and ERK.12,13
In addition to the “driver” mutations that largely define the MPN phenotype, a constantly increasing number of coexisting mutations in epigenetic modifiers, such as TET2, DMNT3A, ASXL1, EZH2, and the genes involved in hematopoietic signaling, such as NRAS, CBL, have been described in Ph chromosome-negative MPNs that may precede the “driver” mutations or occur at a later disease stage.1,14 Some of these mutations, such as TP53, TET2, SH2B3, and IDH1, are more frequently seen in leukemic transformation phase of MPNs, which, in addition to the acquisition of cytogenetic abnormalities, suggests their role in disease progression.1
Another sign of MPN progression is the development of bone marrow fibrosis, which has been associated with increased cytogenetic aberrations and higher number of specific mutations.1,15,16 Chronic inflammation has been suggested as a driver of clonal evolution in MPNs and a potential trigger of bone marrow fibrosis, which is likely a part of polyclonal stromal reaction to the proliferation of neoplastic cells.17 Aberrant production of fibrogenic factors, such as transforming growth factor-β, fibroblast growth factor, and lysyl oxidase, by the neoplastic cells (particularly megakaryocytes) leads to the stimulation of marrow cells
including fibroblasts, osteoblasts, and endothelial cells with consequent development of fibrosis and osteosclerosis18 (Figs. 8.3 and 8.4).
including fibroblasts, osteoblasts, and endothelial cells with consequent development of fibrosis and osteosclerosis18 (Figs. 8.3 and 8.4).
This chapter reviews seven MPNs and summarizes the revised diagnostic and prognostic criteria based on the new clinical, laboratory, morphologic, and molecular genetic data that have emerged in the last decade.
CHRONIC MYELOID LEUKEMIA, BCR-ABL1-POSITIVE
CML, BCR-ABL1-positive, is the most common MPN and accounts for approximately 15% to 20% of all adult leukemias with an annual incidence of 1 to 2 cases per 100,000 with a slight male predominance.19,20 It typically occurs at ages 40 to 60, with about 20% to 50% of patients asymptomatic, the diagnosis having been suggested by hepatosplenomegaly on physical examination (Fig. 8.5) or abnormal results—leukocytosis, anemia, or thrombocytosis—on routine hematologic testing. When symptoms occur (Table 8.4), they usually relate to splenomegaly (left upper quadrant discomfort or early satiety), problems from an increased white cell production (bone pain, mild fever, night sweats, weight loss), or anemia (dyspnea, fatigue, pallor).21,22
CML is defined by the presence of the Philadelphia chromosome, a derivative chromosome 22 that harbors the BCR-ABL1 fusion gene, the result of reciprocal translocation t(9;22)(q34;q11) (Fig. 8.6). The BCR-ABL1 fusion gene results in the formation of the BCR-ABL1 fusion protein with constitutive tyrosine kinase activity of ABL1.23 Although most cases of CML can be diagnosed based on peripheral blood findings combined with detection of BCR-ABL1 by molecular genetic techniques (karyotype, fluorescence in situ hybridization [FISH] or reverse transcription polymerase chain reaction [RT-PCR]), a bone marrow biopsy and aspirate are essential for morphologic evaluation to confirm disease phase.
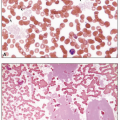
The disease goes through three stages: chronic phase (CML-CP), accelerated phase (CML-AP), and blast phase (CML-BP). In the chronic phase, which is present at the time of the diagnosis in approximately 85% of patients, the blood studies typically show mild anemia and leukocytosis that usually exceeds 25 × 109/L (median white count of about 170 × 109/L), primarily comprising neutrophils in various stages of maturation, especially myelocytes and mature neutrophils (Table 8.5). Basophils are universally increased, and eosinophilia is common24 (Fig. 8.7). The platelet count is normal or elevated, and may exceed 1,000 × 109/L, but resulting thrombosis is unusual.21,22 Serum lactate dehydrogenase and uric acid are commonly increased, reflecting the underlying excessive cell proliferation. The bone marrow shows hypercellularity due to marked granulocytic hyperplasia, blasts constitute less than 5% of the cells, and megakaryocytes are small and hypolobular (Figs. 8.8 and 8.9). Eosinophils may be prominent (Fig. 8.10). In about 50% of patients, the megakaryocytes are increased in number, and, especially in this group, but also in others, reticulin fibrosis of the bone marrow may be apparent. Because of the excessive hematopoiesis, the number of cells that eventually die increases, and macrophages containing the lipids from the dead cells may be visible as sea-blue histiocytes or pseudo-Gaucher cells (Fig. 8.11).
In the era of therapy with tyrosine kinase inhibitors (TKI) the accelerated phase is becoming less common and currently there are no universally accepted criteria for its definition. The existing diagnostic WHO criteria for CML that include hematologic, morphologic, and cytogenetic and molecular parameters25 have been revised to include the “provisional response-to-TKI” criteria (Table 8.6). The accelerated phase should be diagnosed if any one or more of the criteria is present. Morphologic signs of the accelerated phase include peripheral blood basophilia of at least 20% and 10% to 19% of blasts in peripheral blood or bone marrow (Fig. 8.12). The finding of even less than 10% of lymphoblasts in peripheral blood or bone marrow warrants a complete clinical and molecular genetic workup as leukemic transformation may be imminent. Bone marrow findings suggestive of, but not definitive for, CML-AP are the presence of large clusters or sheets of abnormal small megakaryocytes associated with marked reticulin or collage fibrosis; these morphologic findings are usually present in conjunction with other criteria of the accelerated phase.
The blast phase is defined by one or more of these features: (1) blasts accounting for at least 20% of peripheral white cells or nucleated bone marrow cells; (2) blasts proliferating in extramedullary sites, such as the skin, lymph nodes, and spleen; and (3) large aggregates of blasts occurring in the bone marrow. The blasts are usually myeloid but in about 20% to 30% of cases, they are lymphoid, usually B lymphoblasts (Figs. 8.13, 8.14, 8.15 and 8.16).
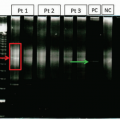
Development of small molecule TKI targeting the constitutively active tyrosine kinase has been one of the most successful stories in modern medicine improving the 10-year survival rate from 20% to 90%.6 Current guidelines recommend the three commercially available TKI (imatinib, dasatinib, and nilotinib) as the first-line treatment for chronic phase CML, with most patients demonstrating an excellent response within the first year of treatment.26 The TKI therapy is continued indefinitely as long as it is tolerated and the treatment milestones are met. Treatment response in CML is defined as hematologic, cytogenetic, and molecular27,28 (Table 8.7). Patients should continuously undergo molecular and/or cytogenetic monitoring. Chromosome banding analysis of at least 20 marrow cell metaphases is necessary to determine the degree of cytogenetic response or to identify secondary abnormalities associated with disease progression (Fig. 8.17). Quantitative reverse transcriptase polymerase chain reaction (QRT-PCR) is generally performed on peripheral blood (Fig. 8.18). Whenever possible, both cytogenetic and molecular tests are recommended until complete cytogenetic response (CCyR) or major molecular
response (MMR) are achieved28 (Fig. 8.19; Table 8.8). Rising levels of BCR-ABL1 transcripts may be associated with disease progression or development of drug resistance. Conventional Sanger sequencing is recommended to detect mutations affecting TKI binding either by directly altering an amino acid on the drug-binding site (e.g., T315I, F317J, F359C/V) or indirectly by altering protein conformation (e.g., G250E, Q252H, E255K/V).29 An alternative TKI or allogeneic stem cell transplant therapies are used in patients who become intolerant to a first-line TKI, show excessive toxicity, treatment failure, or suboptimal response. The prognosis of CML in accelerated or blast phases is dismal, particularly for patients with prior TKI therapy, as many of them develop resistance (Fig. 8.20). There is a significant relapse rate even after successful treatment with TKI and these patients should be considered for transplantation.
response (MMR) are achieved28 (Fig. 8.19; Table 8.8). Rising levels of BCR-ABL1 transcripts may be associated with disease progression or development of drug resistance. Conventional Sanger sequencing is recommended to detect mutations affecting TKI binding either by directly altering an amino acid on the drug-binding site (e.g., T315I, F317J, F359C/V) or indirectly by altering protein conformation (e.g., G250E, Q252H, E255K/V).29 An alternative TKI or allogeneic stem cell transplant therapies are used in patients who become intolerant to a first-line TKI, show excessive toxicity, treatment failure, or suboptimal response. The prognosis of CML in accelerated or blast phases is dismal, particularly for patients with prior TKI therapy, as many of them develop resistance (Fig. 8.20). There is a significant relapse rate even after successful treatment with TKI and these patients should be considered for transplantation.
CHRONIC NEUTROPHILIC LEUKEMIA
CNL is a very rare MPN in which marrow granulocytic hypercellularity results in persistent blood neutrophilia. Mature neutrophils and bands constitute most of the neutrophils, with few being less immature forms. The neutrophils may appear normal, but sometimes contain coarse granules. The bone marrow shows normal numbers of blasts or promyelocytes, but myelocytes and mature neutrophils are increased (Fig. 8.21). Splenomegaly and hepatomegaly usually are present. Until recently, CNL was a diagnosis of exclusion requiring an extensive workup for other reactive or neoplastic conditions presenting with neutrophilia. However, the discovery of mutations in the CSF3R gene affecting the receptor for colony stimulating factor in a large subset of CNL patients has changed the approach to CNL30 and the WHO diagnostic algorithm has been revised to include the presence of CSF3R T618I or other activating CSF3R mutation as a diagnostic criterion of CNL (Table 8.9).4 Mutational frequency of CSF3R in CNL has been reported to be between 89% and 100%.30,31 CSF3R mutations, driver mutations in CNL, are often accompanied by recurrent mutations, such as SETBP1 and ASXL1, present in other myeloid neoplasms. The presence of these mutations in nearly all CNL cases greatly simplifies its distinction from reactive neutrophilia. Two types of CSFR3 mutations have been described: membrane proximal, the main mutations that include T618I (exon 14 C-to-T substitution at nucleotide 1853) and T615A, and truncating mutations often seen in association with T618I/T615A. Although both mutational types demonstrate growth factor-independent proliferation, JAK-STAT activation and resulting sensitivity for a JAK1/2 inhibitor ruxolitinib is only seen in the presence of the membrane proximal type. The truncating mutations are sensitive to the multikinase inhibitor dasatinib.30,32 The presence of CSF3R mutations is uncommon in other myeloid neoplasms and has been described in rare cases of de novo acute myeloid leukemia (AML; <1%) and atypical CML (3%), the main differential diagnosis for CNL.31,33 Both, CNL and atypical CML present with persistent peripheral blood leukocytosis; however, in contrast to CNL where immature myeloid cells are rare (<10% of the blood WBC), in atypical CML, promyelocytes, myelocytes, and metamyelocytes account for at least 10% of blood WBC. Unlike CNL, atypical CML displays dysgranulopoiesis (e.g., nuclear hypolobularity, abnormal chromatin clumping) with or without dyserythropoiesis or dysmegakaryopoiesis (Fig. 8.22; Table 8.10).4
Allogeneic stem cell transplant is the preferred therapy option for CNL patients with hydroxyurea as the first-line drug therapy. Investigational therapy includes the use of ruxolitinib, a JAK1/2 inhibitor, in patients who failed hydroxyurea; this treatment may provide symptom palliation without necessarily affecting disease outcome.34,35
POLYCYTHEMIA VERA
PV is one of the classic Philadelphia-negative MPNs characterized by increased red cell production independent of the mechanisms that normally regulate erythropoiesis associated with the somatic gain-of-function mutations in the cytoplasmic tyrosine kinase JAK2 that lead to hypersensitivity to cytokines and trilineage proliferation. The incidence of PV is estimated at 1.9/100,000 per year with a slight male predominance and the average age of 60 at diagnosis.36, 37, 38 Many patients are asymptomatic, and the diagnosis may be suspected by the findings of plethora and splenomegaly on examination or abnormalities on a routine blood count. The symptoms typically arise from the erythrocytosis, which causes hyperviscosity and a tendency for venous and arterial thromboses, such as myocardial infarctions, strokes, and venous thromboses of the legs (Table 8.11).37,39 In some patients, concomitant platelet abnormalities abet this propensity to clot. Venous occlusion of the intraabdominal vessels—such as the portal, hepatic (Budd-Chiari syndrome), and mesenteric veins—is especially suggestive of PV.40 Other features presumably related to the erythrocytosis include dizziness, tinnitus, headache, paresthesias, visual disturbances, and painful feet, sometimes associated with digital ischemia and ulcerations despite palpable pulses. A finding probably originating from platelet abnormalities is erythromelalgia—a combination of a burning sensation, erythema, and warmth in the hands and feet that is worsened by exercise, dependency of the extremity, and heat and that is reduced by elevation, cooling, and aspirin.36 Hemostatic problems, such as epistaxis, gingival hemorrhage, and gastrointestinal bleeding, are common and probably relate to abnormal platelet function and, in about one-third of patients, primarily those with marked thrombocytosis, to the development of acquired von Willebrand disease. The reduction in von Willebrand factor apparently occurs from absorption of large von Willebrand multimers onto platelets, resulting in their removal from the circulation and subsequent destruction. Another symptom, present in
about one-half of patients, is itching on exposure to hot water (aquagenic pruritus), probably caused in part by histamine release from basophils. Excessive cell production can lead to weight loss and sweating, presumably from hypermetabolism, and to hyperuricemia, which often leads to attacks of gout.41 Findings on examination include hypertension and ruddy cyanosis, apparent on the nose, cheeks, lips, ears, neck, and digits. The eyes can appear bloodshot because of conjunctival plethora, and fundus examination may reveal distended, tortuous, and unusually violaceous vessels. Approximately 70% of patients have palpable splenomegaly and almost half have hepatomegaly.42
about one-half of patients, is itching on exposure to hot water (aquagenic pruritus), probably caused in part by histamine release from basophils. Excessive cell production can lead to weight loss and sweating, presumably from hypermetabolism, and to hyperuricemia, which often leads to attacks of gout.41 Findings on examination include hypertension and ruddy cyanosis, apparent on the nose, cheeks, lips, ears, neck, and digits. The eyes can appear bloodshot because of conjunctival plethora, and fundus examination may reveal distended, tortuous, and unusually violaceous vessels. Approximately 70% of patients have palpable splenomegaly and almost half have hepatomegaly.42
The diagnosis of PV requires fulfillment of the WHO criteria that are based on hemoglobin and/or hematocrit levels, bone marrow biopsy morphologic findings, presence of JAK2 V617F or JAK2 exon 12 mutation and subnormal serum erythropoietin levels, the only minor criterion that is not required if the first three criteria are met (Table 8.12). One of the major criteria, the JAK2 V617F mutation, is present in greater than 95% of PV patients,43 which although establishes disease clonality, is not specific, as the same mutation is present in a large proportion of ET and PMF cases, although mutant allele burden and evidence of JAK2 V617F homozygosity is significantly higher in PV.44,45 On the other hand, JAK2 exon 12 mutation, a rare mutation seen in only 3% of patients, is specific to PV46,47 (Table 8.14).
PV progresses through three stages that define clinical presentation and morphologic findings: pre-polycythemic/early phase, polycythemic phase, and post-polycythemic myelofibrosis (post-PV MF) phase. Most PV patients present in the polycythemic phase that is associated with a significantly increased red cell mass leading to markedly elevated hematocrit values, frequently above 60%, leukocytosis of greater than 10,000/µL, and thrombocytosis of greater than 450,000/µL. Because excessive red cell production depletes iron stores, erythrocytes may be microcytic and hypochromic (Fig. 8.23). Other findings sometimes seen on a peripheral blood smear include polychromatophilia, basophilic stippling, nucleated red cells, and markedly enlarged and misshapen platelets.
Patients presenting in early-phase PV, also known as masked PV, may have hemoglobin and hematocrit values that are just slightly elevated; if a red cell mass evaluation is not performed, such patients may be mistakenly diagnosed as ET, especially, those presenting with very high platelet counts.48,49 However, discrimination between masked PV and ET is straightforward on morphologic evaluation of a bone marrow biopsy that shows findings similar to those seen in the polycythemic stage of PV.50 To better capture the early-phase PV patients, the WHO has lowered the hemoglobin/hematocrit levels and included bone marrow biopsy findings as one of the required major criteria to reflect the importance of morphologic evaluation.
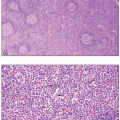
Bone marrow biopsy findings in PV (both early and overt polycythemic stages) include age-adjusted hypercellularity and trilineage proliferation, which includes pleomorphic megakaryocytes often occurring in loose clusters18 (Fig. 8.24). Some patients, especially those with more pronounced megakaryocytic proliferation, may display MF-1 bone marrow fibrosis. Iron stores are nearly always absent.
After a variable time, which is often of many years, approximately 12% to 21% of PV patients progress to a “spent” phase of the disease that is associated with the development of post-PV MF.51,52 This phase is characterized by anemia and other cytopenias caused by ineffective erythropoiesis, marrow fibrosis, hypersplenism, and extramedullary production of cells with shortened life spans. The spent phase occurs in up to 50% of patients and, on average, appears about 10 years after the diagnosis. The blood smear shows leukoerythroblastosis (the presence in the peripheral blood of nucleated red cells and cells in the granulocyte series that are more immature than bands [e.g., metamyelocytes, myelocytes]), poikilocytosis, and teardrop cells (Fig. 8.25). The bone marrow demonstrates reticulin and collagen fibrosis (at least MF-2), areas of trilineage hypocellularity, intrasinusoidal hematopoiesis, and osteosclerosis. Atypical megakaryocytes may be present (Fig. 8.26). Less than 10% of PV will develop accelerated or blast phases which may resemble AML or MDS53 (Fig. 8.27).
To date, no drug has been shown to improve survival, delay in development of marrow fibrosis, or lower risk of leukemic transformation in PV. However, current available treatment modalities extend patient survival to over 13 years.51 The overall treatment goals in PV patients are to alleviate clinical symptoms and prevent complications associated with thrombotic events and progression to myelofibrosis and/or AML/MDS.54 Patients without active thrombosis or at low risk of thrombotic complications are treated with therapeutic phlebotomy. In high-risk patients, phlebotomy is supplemented by cytoreductive agents, such as hydroxyurea and interferon-α. Therapeutic considerations for patients with progression to post-PV MF include allogeneic stem cell transplant, JAK1/JAK2 inhibitors, or other investigational disease-modifying agents (these therapies are discussed in more detail in the next section).
PRIMARY MYELOFIBROSIS
PMF is the least common Ph-negative MPN that is characterized by a clonal proliferation of megakaryocytes and granulocytic precursors in the bone marrow through dysregulation of the JAK/STAT signaling pathway that is accompanied by various degrees of marrow fibrosis and extramedullary hematopoiesis. In contrast with PV and ET, two other classic subtypes of Ph-negative MPNs, PMF demonstrates a significantly reduced median overall survival estimated at 6.5 years and an increased risk of leukemic transformation.55 A number of prognostic scoring systems have been developed to stratify patents into four risk categories (low, intermediate-1, intermediate-2, and high) associated with significantly different overall survival and leukemic transformation rate.56,57 Such patient stratification is based on clinical (age >65 years, presence
of constitutional symptoms and transfusion requirement), hematologic (leukocyte count >25,000/µL, hemoglobin <10 g/dL, platelets <100,000/µL, and circulating blasts ≥1%), and cytogenetic (complex karyotype or sole or two abnormalities that include inv(3), -5/5q-, -7/7q-, +8, 11q23 rearrangements, 12p- or i(17q)) parameters. Although accurate, this scoring system does not reflect the complex genetic landscape of PMF. In contrast to the unfavorable karyotype that is present only in 7% of PMF patients, mutations in JAK2 V617F, CALR, and MPL are found in approximately 60%, 20%, and 5%, respectively (Table 8.13). Approximately 10% to 15% of PMF patients lack any of these mutations and are designated as “triple-negative.”3 These mutational groups are associated with different median survival, and the mutational analysis of JAK2, CALR, and MPL should be performed for diagnostic and prognostic purposes. The most recently described CALR mutation is associated with the longest median survival (15.9 years), followed by mutated MPL (9.9 years) and mutated JAK2 V617F (5.9 years); the “triple-negative” group is associated with the shortest median survival of 2.3 years.51 In addition, the presence of ASXL1, SRSF2, and EZH2 mutations has been shown to be independently associated with inferior outcome, such as premature death and leukemic transformation identifying “high molecular risk” patients.58
of constitutional symptoms and transfusion requirement), hematologic (leukocyte count >25,000/µL, hemoglobin <10 g/dL, platelets <100,000/µL, and circulating blasts ≥1%), and cytogenetic (complex karyotype or sole or two abnormalities that include inv(3), -5/5q-, -7/7q-, +8, 11q23 rearrangements, 12p- or i(17q)) parameters. Although accurate, this scoring system does not reflect the complex genetic landscape of PMF. In contrast to the unfavorable karyotype that is present only in 7% of PMF patients, mutations in JAK2 V617F, CALR, and MPL are found in approximately 60%, 20%, and 5%, respectively (Table 8.13). Approximately 10% to 15% of PMF patients lack any of these mutations and are designated as “triple-negative.”3 These mutational groups are associated with different median survival, and the mutational analysis of JAK2, CALR, and MPL should be performed for diagnostic and prognostic purposes. The most recently described CALR mutation is associated with the longest median survival (15.9 years), followed by mutated MPL (9.9 years) and mutated JAK2 V617F (5.9 years); the “triple-negative” group is associated with the shortest median survival of 2.3 years.51 In addition, the presence of ASXL1, SRSF2, and EZH2 mutations has been shown to be independently associated with inferior outcome, such as premature death and leukemic transformation identifying “high molecular risk” patients.58
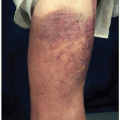
PMF occurs mainly in middle-aged and elderly patients, and the median age at presentation is 67 years.59 About 30% to 40% of patients are asymptomatic at diagnosis, which may be suggested by splenomegaly, the hallmark of PMF, or abnormal blood tests. Symptoms typically occur from anemia, hypermetabolism from high cell turnover, splenomegaly, or thrombocytopenia (Table 8.14).60 Hypermetabolism may cause weight loss, fever, sweats, or problems associated with hyperuricemia, including attacks of gout or renal stones. Anemia can lead to complaints of fatigue, dyspnea, weakness, and palpitations. Splenomegaly can produce left upper quadrant discomfort, a sensation of early satiety from compression of the stomach, or diarrhea from pressure on the bowels. On examination, splenomegaly is nearly universal and often enormous, and hepatomegaly is present in about one-half of the patients.61 Thrombocytopenia or altered platelet function may cause cutaneous hemorrhage, and, occasionally, extramedullary hematopoiesis in the skin produces erythematous or purplish nodules, papules, and plaques. Extramedullary hematopoiesis is most common in the spleen and liver, but can also affect many other sites, such as the dura mater, lymph nodes, lung, and pleura (Fig. 8.28).62
PMF represents a stepwise evolution from an initial early (prefibrotic or minimally fibrotic) phase to an overt fibrotic phase. Clinical, laboratory, and morphologic findings depend on the disease phase. Approximately one-third of the patients are diagnosed during the prefibrotic phase, in which the characteristic findings are borderline anemia, thrombocytosis, and mild leukocytosis clinically mimicking ET.63 The blood smear may be unremarkable or only show occasional teardrop-shaped erythrocytes (dacrocytes) and large platelets. Discrimination between early/prefibrotic PMF and ET is clinically relevant, as patients presenting in the prefibrotic PMF phase demonstrate worse overall and event-free survival, higher rate of progression to fibrotic stage, and transformation to acute leukemia.64 Because of a significant overlap in clinical, laboratory, and molecular findings, the distinction between prefibrotic PMF and ET remains based predominantly on bone marrow biopsy morphologic assessment that is reflected in the new WHO criteria for early (prefibrotic or minimally fibrotic) PMF (Table 8.15).4 Unlike ET, the marrow cellularity in prefibrotic PMF is increased because of granulocytic and megakaryocytic proliferation. Megakaryocytes are seen in loose and tight clusters, and demonstrate an atypical appearance because of the presence of an increased nuclear:cytoplasmic ratio, abnormal chromatin clumping and hyperchromatic changes producing the so-called “bulbous” or “cloud-like” nuclei; presence of bare megakaryocytic nuclei is another commonly observed finding (Table 8.16; Figs. 8.29 and 8.30).65 Fibrosis at this stage is absent or minimal (MF-0 or MF-1). The prominent hematologic findings of the overtly fibrotic phase (MF-2 or MF-3) are anemia and a leukoerythroblastic blood smear (nucleated erythrocytes, immature granulocytes) with numerous dacrocytes. White cell and platelet counts vary widely, but a few myeloblasts are common, and platelet abnormalities include large size, bizarre shape, circulating megakaryocyte nuclei, and micromegakaryocytes (Fig. 8.31). Bone marrow aspiration is usually unsuccessful, but the bone marrow biopsy demonstrates variable cellularity, with increased deposition of reticulin and/or collagen fibrosis and numerous atypical megakaryocytes in large aggregates. Osteosclerosis (new bone formation) may be visible (Figs. 8.32 and 8.33). Overt PMF, especially the advanced stages, may be difficult to distinguish from fibrotic stages of ET and PV.66 The diagnosis of overt PMF requires meeting three major and at least one minor WHO criteria that are based on clinical, morphologic, laboratory, and molecular findings (Table 8.17).4
One of the important diagnostic and prognostic morphologic findings in PMF is the presence of myelofibrosis. Absent or minimal myelofibrosis (MF-0 and MF-1) define early/prefibrotic PMF, whereas advanced myelofibrosis grades (MF-2 and MF-3) define overt PMF, as well as progression of ET and PV to the post-ET myelofibrosis (post-ET MF) and post-PV MF stages.4,67 Progressive bone marrow fibrosis is associated with increasing density of reticulin fibers and collagen deposition, and is graded on a semiquantitative scale that utilizes reticulin and trichrome stains (Figs. 8.34 and 8.35; Table 8.18).4,68 Strict adherence to the WHO myelofibrosis grading criteria significantly improves accuracy and interobserver reproducibility.69 In addition, in the era of the novel disease-modifying agents (see below) that have been shown to diminish or resolve bone marrow fibrosis, dynamic myelofibrosis grade is utilized for treatment response assessment and prognosis.18 It is important to mention that myelofibrosis is not specific to Ph-negative MPNs and may accompany a large number of other neoplastic and nonneoplastic conditions, which should be excluded, especially in the absence of any clonal mutations (Table 8.19).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree