Medical Care of Athletes and Infection Risk
Deverick J. Anderson
Samuel Hume
Introduction
Team medical personnel play a critical role in infection prevention among athletes. Primarily, medical personnel are responsible for practicing rigorous infection control strategies while providing medical care, preventative strategies, and appropriate education to athletes. Secondarily, team medical personnel must help promote best practices for infection prevention as part of a culture of safety.
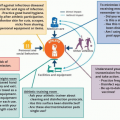
This chapter provides best practices, recommendations, and information for infection prevention while performing direct medical care. Many of the recommendations included in this chapter are based on the premise that the athletic training facility and treatment rooms are medical facilities. In addition to strategies for direct medical care, we also provide several recommendations on indirect strategies for prevention, including standardization and building a culture of safety in the facility.
The athletic training facility is a medical facility. The same infection prevention practices used in hospitals and clinics must be used in the training facility.
General Information
Medical personnel interact with athletes on multiple levels. In addition to providing direct care, medical personnel are responsible for “setting the tone” in the facility. Adherence to key infection prevention principles improves care in real time and provides an example to others in the facility. Basic principles like standardization and building a culture of safety are proven strategies to improve care and adherence to best practices. More practical principles include single-use items (when possible) and simply put, making it easy to do the right thing. Examples of all of these principles are provided throughout this chapter.
Basic Infection Prevention Principles Can Be Summarized in a Few Simple Statements
Use a systematic approach to implementation
Develop a culture of safety
Use single-use disposable medical equipment
Make it easy to do the right thing
Standardization
Rationale: Standard practice guidelines improve patient care in the hospital setting1,2 and are a cornerstone in the fight to prevent healthcare-associated infections.3 Successful infection prevention methods often combine a number of evidence-based interventions into an easy-to-follow “bundle” or checklist.4,5 Despite the success of these standardized interventions, each healthcare facility must critically analyze and tailor standard practice guidelines to their local environment.
Sample policies provided in the appendices can be directly used or further modified to fulfill this recommendation. Ideally, athletic trainers, team physicians, coaches, and administration participate in the creation of their local infection prevention policies.
In addition to standardizing practices through policies, materials should be standardized throughout the facility when possible. For example, we recommend using the same types of surface disinfectants in all areas of the training facility (eg, training room, equipment room, weight room, and even offices). This approach ensures users are familiar with the products and ensures that materials are more easily ordered and stocked. Other materials that can be standardized include hand hygiene products (gel, foam, or liquid) and soap.
Rationale: Standard practice guidelines are most helpful when they are easily accessible.6 Many professional medical societies post best practice guidelines online to improve accessibility and adherence. All teams should disseminate infection prevention guidelines to their staff. Readily accessible documents not only help to improve standardization but also can be easily referenced if/when specific questions arise. Electronically available guidelines improve adherence to policies in hospitals.
However, easy and convenient access to electronic guidelines is not a practical solution in most training facilities. Thus, we recommend creating and placing a policy folder in treatment rooms where it can be readily accessed when needed.
Training in Infection Prevention
 Best Practice Ensure team medical personnel know basic and advanced infection prevention strategies and recommendations.
Best Practice Ensure team medical personnel know basic and advanced infection prevention strategies and recommendations. Recommendation 1 Athletic trainers must review educational materials regarding infection prevention annually.
Recommendation 1 Athletic trainers must review educational materials regarding infection prevention annually.Athletic trainers are the most important members of the infection prevention team for athletes.
Rationale: Team athletic trainers have the most interaction with and provide the most care to players. Athletic trainers are important and key members of the infection prevention team. As a result, athletic trainers must stay up-to-date with standards of infection prevention and team infection prevention policies.
Annual review of educational materials concerning common infections among players, such as skin and soft-tissue infections due to methicillin-resistant Staphylococcus aureus (MRSA), will improve the care that athletic trainers provide for players. A study of more than 150 athletic trainers revealed conflicting viewpoints about MRSA, hand hygiene, and the use of disinfectants.7 These discrepancies highlight the need for ongoing education and standardization among athletic trainers. Furthermore, infection prevention is a dynamic field. The epidemiology of infectious agents changes over time, and advances in infection prevention principles and technology lead to continually evolving standards of care.
 Recommendation 4 Ensure team physicians know of changes to infection prevention policies when changes are made.
Recommendation 4 Ensure team physicians know of changes to infection prevention policies when changes are made.Rationale: Annual infection prevention training is required of all physician staff at most medical institutions. Thus, we assume that most, if not all, team physicians receive formal training and education in basic infection prevention strategies each year. However, specific components of infection prevention policies at one institution may vary significantly from components at a different institution (eg, the types of antiseptic agent used preoperatively). Thus, we recommend that team physicians thoroughly understand and periodically review infection prevention policies written specifically for your team (and outlined in this manual). First, this approach will ensure that the physicians are aware of potential differences in policy components. Second, this approach will simultaneously promote discussion when differences in infection prevention approaches are identified. These discussions may help identify the best approaches to use at the training facility.
Culture of Safety
Rationale: When a culture of safety is in place, all members of the athletic training staff and team are actively engaged in monitoring and improving safe practices. Pressure to improve practices comes from peers and, more importantly, leaders. According to the Institute for Healthcare Improvement, an organization can develop a culture of safety only when leaders are openly and visibly committed to change, improvement, and open sharing of information. The sum of our proposed best practices and recommendations will augment this culture of safety, but this culture must ultimately be generated and fostered within each facility and training staff.
Pressure to improve practices comes from peers and, more importantly, leaders.
Rationale: Knowledge of best practices is easier to achieve than rigorous implementation of best practices. Thus, additional effort is required to ensure adherence to best practices and recommendations. One basic approach to assist implementation is to ensure easy access to materials necessary to implement best practices. For example, all athletic trainers know hand hygiene is important, but washing hands is a more difficult task if the only available alcohol dispenser or sink is located across the training room. Similarly, using a systematic approach improves implementation. For example, we recommend that facilities identify a single agent for disinfection and use it throughout the facility instead of using a unique disinfectant agent in every setting. Following a systematic approach improves understanding of the why and how practices must be completed. Finally, while athletes inevitably will be exposed to potentially infectious organisms, exposure can be limited by the use of single-use items. Use of single-use items eliminates the risk of cross-transmission and simplifies processes (making it easier to do the right thing).
 Best Practice Provide visual reminders about the importance of infection prevention strategies and policies.
Best Practice Provide visual reminders about the importance of infection prevention strategies and policies. Recommendation Add signs or posters throughout the facility to improve infection prevention practices.
Recommendation Add signs or posters throughout the facility to improve infection prevention practices.Rationale: Signs increase awareness of important infection prevention policies. Such signs are routinely placed in most medical facilities. Educational signage improves the culture of safety by providing constant reminders of core principles to players and athletic trainers. Their presence demonstrates the team’s acknowledgment of the importance of these policies.
Signs Improve the Culture of Safety
Avoid “sign fatigue” with simple strategies.
Move signs on a rotating basis
Change the physical appearance of the signs (though not necessarily changing the content)
Changing the content or wording of signs while retaining the same message
Signs are recommended to improve hand hygiene, discourage cosmetic body shaving, improve infection prevention in the hydrotherapy room, and promote safe injection policies. For examples of these type of signs, visit the e-book version of this book.
Basic Medical Care
Best practices and recommendations provided throughout this book are founded on one major tenet: athletic training facilities are medical facilities. Many of the same infection prevention practices and recommendations related to medical care provided in hospitals and outpatient clinics are applicable guides for infection prevention in athletic training facilities. Overall, these infection prevention approaches can be categorized as (1) strategies that decrease the inherent risk of infection from medical care or (2) strategies that decrease the inherent risk of infection from athletic activities.
Athletic training facilities are medical facilities.
Basic Strategies to Decrease Risk of Infection in Athletes
Hygiene—emphasize skin care with regular hand and body hygiene
Use protocols to guide treatment of common infections
Safe injection practices
Vaccination
Vaccination is a critical component of infection prevention. See Chapter 6 for detailed information about which vaccines are most important for athletes.
Decrease Risk of Infection From Medical Care
Simply put, medical care involves risk of infection. Any time skin is disrupted during a procedure—even simply drawing blood or administering an IV—risk of infection occurs. In fact, the biggest culprit for transmission of infection while providing medical care is contaminated, unwashed hands of medical personnel. Simple but effective strategies can reduce these risks.
Medical care involves risk of infection.
HAND HYGIENE
 Best Practice Actively promote and increase the use of hand hygiene as the primary method of infection prevention in the facility.
Best Practice Actively promote and increase the use of hand hygiene as the primary method of infection prevention in the facility. Recommendation 1 Increase the number and locations of alcohol-based hand hygiene dispensers throughout the facility.
Recommendation 1 Increase the number and locations of alcohol-based hand hygiene dispensers throughout the facility. Recommendation 2 Ensure hand hygiene stations (ie, sink and soap dispensers and/or alcohol-based hand cleaning agents) are located at convenient locations in all medical treatment areas and at entry and exit doors in other areas of the facility.
Recommendation 2 Ensure hand hygiene stations (ie, sink and soap dispensers and/or alcohol-based hand cleaning agents) are located at convenient locations in all medical treatment areas and at entry and exit doors in other areas of the facility.Rationale: Direct (hand-to-skin) transmission is the primary method by which most bacterial pathogens (such as MRSA) are transmitted in healthcare facilities. Similar direct transmission also plays an important role in the spread of viruses such as those causing influenza or the common cold.8,9 Common pathogens that are temporarily present on the hands of athletic trainers or players can be transmitted to others through direct skin contact if hand washing is omitted, inadequate, or performed with an inappropriate agent.10 Put another way, your hands are contaminated with potential pathogens every time you provide player treatment.
Hand hygiene is the #1 method for preventing transmission.
Alcohol-based hand hygiene products are highly effective in reducing direct, person-to-person transmission of almost all of these pathogens, including MRSA.11,12 In fact, alcohol-based products are as effective as traditional hand washing with soap and water in most situations (see Recommendation 4 for important exceptions). Use of these products takes less time and results in less skin irritation than soap or other antiseptic agents that require water.13
Increasing the number and use of hand hygiene product dispensers has two positive effects. First and most importantly, placing hand hygiene products close to the point of care makes it both convenient and easy for athletic trainers and players to perform hand hygiene. Second, increasing the number of hand hygiene dispensers underscores, highlights, and makes overt the team’s commitment to hand hygiene as the cornerstone of all infection prevention activities.
We recognize that there is a continuum of risk of transmitting pathogens via routine player treatments. This risk is lowest and minimal during activities such as taping of intact skin and highest when there is direct hand-to-skin contact with abraded, cut, or nonintact skin.
Tips and Advice: Make Hand Hygiene Easy to Access
We recommend adding alcohol-based hand hygiene dispensers in the following specific strategic locations:
Entrances and “pinch points” through which people routinely pass
Entry to the general facility
Entry to the training room
Entry to the examination procedure room
Entry to the locker room
Entry to the hydrotherapy room
Entry to the cafeteria and other refreshment areas
Throughout the training room, including between treatment beds
Throughout the players’ locker room
Particularly emphasize player hand hygiene when entering and leaving the weight room. Some athletes have a higher prevalence of MRSA colonization of the hands.
Alcohol-based hand hygiene products can be delivered as foams, gel, or liquids. There is no difference in effectiveness between these forms.
Dispensers should be located in easily accessible and visible locations.
 Recommendation 3 Clean hands with soap and water instead of alcohol-based products when they are visibly soiled or heavily contaminated with dirt, debris, body fluids, or blood.
Recommendation 3 Clean hands with soap and water instead of alcohol-based products when they are visibly soiled or heavily contaminated with dirt, debris, body fluids, or blood. Recommendation 4 Clean hands with soap and water instead of alcohol-based products after treating a player with a diarrheal illness.
Recommendation 4 Clean hands with soap and water instead of alcohol-based products after treating a player with a diarrheal illness.Rationale: Alcohol-based hand hygiene products are not effective when hands are heavily soiled with dirt or other material such as oils or blood. Alcohol does not kill two important pathogens that cause highly transmissible diarrheal illness—norovirus and Clostridioides difficile (also known as “C. diff”).
 Recommendation 5 Ensure moisturizing hand lotions or emollients are available to staff and players to prevent dry or cracked skin.
Recommendation 5 Ensure moisturizing hand lotions or emollients are available to staff and players to prevent dry or cracked skin.Rationale: Frequent use of alcohol-containing hand hygiene products can occasionally lead to dry and cracked skin. While most hand hygiene products also contain emollients to prevent drying of skin, frequent use may still result in some degree of drying and cracking of skin. Dry, cracked, or irritated skin is more likely to harbor pathogens such as MRSA. Thus, ensuring that lotion is available for use can help overcome this potential side effect of increased hand hygiene.
Dry, cracked, or irritated skin is more likely to harbor pathogens such as MRSA.
 Recommendation 6 Use hospital-grade soap from prefilled/packaged dispensers in the training, treatment, and examination/procedure rooms.
Recommendation 6 Use hospital-grade soap from prefilled/packaged dispensers in the training, treatment, and examination/procedure rooms.Rationale: As detailed above, a few situations require hand washing with soap and water rather than alcohol-based products. Medical treatment facilities typically use antibacterial soap dispensed from prefilled, replaceable dispensers.
We acknowledge that the use of antibacterial soap is controversial. There is no compelling evidence that antibacterial soaps prevent infection in the community better than plain soap and water.14 Furthermore, triclosan, the active ingredient of most commercially available antibacterial liquid soaps, may disrupt reproductive or thyroid function in aquatic life and laboratory animals.15,16 In fact, the FDA recently concluded that the majority of chemicals used in over-the-counter antiseptic soap, including triclosan, were “not generally recognized as safe and effective.”17 Thus, we currently do not recommend the use of soaps containing triclosan in training facilities. Instead, we recommend liquid soaps containing chloroxylenol, benzalkonium, or chlorhexidine in prefilled dispensers. Finally, refillable soap dispensers have a known risk of becoming contaminated with potential pathogens such as Pseudomonas and Serratia. Thus, we recommend using prefilled/packaged dispensers that can be replaced when the soap content becomes empty or low instead of “topping off” refillable dispensers.
Decrease Risk of Infection From Athletic Activities
PLAYER HYGIENE
As described above, medical personnel must diligently perform hand hygiene to decrease the indirect transmission of pathogens from player to player through transiently contaminated medical personnel hands. Player hygiene is equally important. Improved player hygiene leads to decreased risk of direct transmission from player to player and indirect transmission through decreased environmental contamination.
Athletes have unique risks compared to the general population. For example, rates of MRSA colonization among athletes are as high as rates of MRSA colonization among patients admitted to intensive care units. Investigators from Brown University published a meta-analysis of the worldwide literature on the prevalence of asymptomatic MRSA nasal and/or skin colonization in athletes.18 They reviewed 382 prior studies of MRSA infections in athletes and selected 15 studies in which prospective screening cultures for MRSA colonization were performed in team athletes. The mean pooled prevalence of MRSA colonization was 8% among American athletes.
MRSA colonization among athletes was six times higher than the rate of colonization in the community.
Colonization rates were higher in collegiate athletes (13%) than in professional athletes and significantly higher in wrestlers (22%) as compared to football players (4%-8%) and basketball players (8%).
The risk of subsequent MRSA infection was seven times higher for colonized athletes than noncolonized athletes.
These findings reinforce numerous recommendations made throughout this manual related to regular hand hygiene by players and staff, prompt care of cuts and other skin injuries, and attention to disinfection of training tables and medical equipment.
 Best Practice Shower after practice to decrease the risk of bacterial transmission from player to player and from player to environment.
Best Practice Shower after practice to decrease the risk of bacterial transmission from player to player and from player to environment.Rationale: The highest risk of transmission of important pathogens such as MRSA from one player to another likely occurs during and immediately after practice. Approximately 1% to 3% of the general American population is colonized with MRSA,19,20 but up to 10% to 20% of athletes are colonized.18,21,22 Put another way, if a team roster includes 50 players, approximately 5 to 10 players may be silently colonized with MRSA. Thus, contact with MRSA is inevitable in team settings. MRSA exposure in the setting of skin abrasions and cuts acquired through routine athletic activities substantially increases the risk of subsequent infection.
Athletes are more likely to have MRSA on their skin than nonathletes.
Rationale: Bar soap has been previously implicated as a potential source of MRSA transmission; sharing bar soap was associated with MRSA infections during an outbreak among football players and in an outbreak in a prison.23 Thus, the easiest way to reduce the risk of sharing or using previously used bars of soap in the shower is to remove them. Nonrefillable soap dispensers (ie, dispensers that utilize prefilled/packaged liquid soap products) should be used instead of refillable dispensers.
 Recommendation 1 Instruct players to bathe with chlorhexidine gluconate (CHG) three times each week.
Recommendation 1 Instruct players to bathe with chlorhexidine gluconate (CHG) three times each week. Recommendation 2 Add 2% or 4% CHG soap dispensers to team showers to promote routine source control.
Recommendation 2 Add 2% or 4% CHG soap dispensers to team showers to promote routine source control.Rationale: “Source control” is the routine application of a disinfectant to skin to reduce the number of pathogenic bacteria that live on the skin. Source control has not been studied in the athletic team environment. Our recommendations are based on the use of source control strategies in ICU patients and military recruits. The use of CHG for routine bathing of ICU patients has emerged as a key practice for preventing infections in the hospital. Daily bathing with CHG in the ICU reduces the infection burden by 37% compared to routine bathing.24 Two studies in military recruits using three times weekly CHG cloths demonstrated a statistically significant reduction in acquisition of Staphylococcus aureus and MRSA (3.3% vs 6.5%) colonization.25,26 Thus, while the use of CHG is typically targeted at reducing the risk of MRSA, it also reduces the risk from methicillin-susceptible Staphylococcus aureus (MSSA).
FDA Announcement About Chlorhexidine Gluconate
The FDA released a safety communication about chlorhexidine gluconate (CHG) in 2017 warning about potential but rare allergic reactions, likely related to increased use of CHG throughout health care. Thus, as with any chemical, it is important to monitor for and routinely inquire about a history of CHG allergy. A recent study analyzed over 25,000 patients admitted to ICUs who received daily bathing with CHG; seven patients had mild adverse events related to CHG, and no patients experienced serious adverse events. We believe the substantial benefit from CHG outweighs potential risks and continue to strongly advocate for CHG use in myriad infection prevention interventions.
The delivery of CHG includes logistical challenges. Ideally, source control would be performed using CHG-impregnated wipes. However, the process of using wipes requires distribution of wipes and reliance on players to use them properly. Thus, for routine source control, we recommend use of the liquid, detergent-based preparations containing 2% to 4% CHG. For additional information about using CHG for source control and strategies for implementation, please see Appendix 1J. In the event that a cluster of infections occurs, we recommend changing from liquid to wipes, as outlined in Chapter 5.
Other products can potentially be used to reduce the number of pathogens on the skin. These products have limitations related to logistics and/or lack of clinical data. For example, daily dilute bleach baths (15-minute soak in ¼ cup bleach per tub) are highly effective at reducing colonization with MRSA; some centers recommend bleach baths as part of a decolonization strategy.27 This strategy is not practical for widespread use in the team environment. However, this strategy has been made more practical by the development of bleach-containing body washes. Small trials involving 28 to 50 children with moderate to severe atopic dermatitis demonstrated that use of a bleach-containing body wash three times weekly led to decreases in the amount and severity of atopic dermatitis symptoms and S. aureus colonization.28 Finally, some companies have begun using silver antiseptics for body washes. One formulation coupled with aloe, vitamin E, and allantoin was deemed “noninferior” to 4% CHG in a study involving effectiveness of inguinal fold disinfection in 81 health volunteers.29
 Best Practice Develop and enforce a policy to guide hygienic use of common balms, lotions, and creams.
Best Practice Develop and enforce a policy to guide hygienic use of common balms, lotions, and creams.Rationale: A central theme throughout this manual is promoting the use of individual-use equipment and materials. The use of pumps to dispense balms, lotions, and creams, when available, achieves the spirit of this theme and greatly reduces the risk of contaminating these commonly used materials with unclean hands. In the event that a specific balm or cream cannot be dispensed via a pump (ie, due to viscosity), the following two recommendations should be followed.
 Recommendation 2 Ensure athletic trainers and players perform hand hygiene prior to accessing balms, lotions, and creams in communal tubs, even if gloves are being used.
Recommendation 2 Ensure athletic trainers and players perform hand hygiene prior to accessing balms, lotions, and creams in communal tubs, even if gloves are being used. Recommendation 3 Ensure athletic trainers and players do not reenter communal tubs without performing hand hygiene prior to reentry.
Recommendation 3 Ensure athletic trainers and players do not reenter communal tubs without performing hand hygiene prior to reentry.Rationale: For materials that can only be provided in communal tubs, contamination of the materials can be greatly reduced by performing hand hygiene and wearing gloves prior to entry. Other strategies for obtaining materials, such as the use of wooden tongue depressors, may serve as an alternative to using hands. Importantly, some healthcare workers believe that wearing gloves is equivalent to performing hand hygiene. This assumption is incorrect. In fact, hands frequently become contaminated with important pathogens (eg, MRSA) present on gloves as gloves are removed.30 Thus, hand hygiene must be performed after removing gloves.
Wearing gloves? Sorry, you still need to wash your hands.
Overall, the risk of transmission of important pathogens from one player or athletic trainer to another via contaminated balms, lotions, or creams is low, particularly when these materials are applied to intact skin. Thus, we acknowledge that the effort to closely follow
and police the above recommendations and policy may be difficult to achieve. If or when one or more cases of MRSA is detected in the training facility, however, we believe the effort to enforce these policies must be expended.
and police the above recommendations and policy may be difficult to achieve. If or when one or more cases of MRSA is detected in the training facility, however, we believe the effort to enforce these policies must be expended.
CUT CARE
Healthy, intact skin is the body’s primary defense against infection.
Rationale: Although simple abrasions and other breaks in the integrity of the normal cutaneous barrier are usually uncomplicated, such injuries are also a common antecedent in players who develop skin and soft-tissue infections. Thus, we advise the use of a standard protocol that includes specific details about wound cleaning, the use of antibacterial ointments, and wound dressings.
A sample “Cut Care Protocol” is provided in Appendix 1C.
Rationale: Although soap and water are adequate cleaning agents, we recommend the general use of an antiseptic agent for standard wound care. Specifically, we advise using 4% CHG (Hibiclens or Betasept wash) for standard wound care.
Why Use Chlorhexidine Gluconate for Wound Care?
Chlorhexidine gluconate (CHG) products have in vitro bactericidal activity against methicillin-resistant and methicillin-susceptible staphylococci, streptococci, and most bacteria that colonize normal skin.31
They are well tolerated and safe.
Unlike Betadine/iodophors, CHG’s activity is not diminished by the presence of organic debris or material such as blood.
CHG produces a prolonged antibacterial effect after application.
CHG “sticks” are now available for use and should replace Betadine sticks in all settings.
While bacterial resistance has been demonstrated to chlorhexidine,32 this occurs only rarely and its clinical significance in everyday practice is currently insignificant.
Iodophor-containing topical agents such as povidone-iodine (Betadine) are also effective topical agents. In head-to-head comparison studies, however, CHG has been shown to have superior antibacterial activity and less inactivation by organic debris or body fluids such as blood.33 For this reason, we advise CHG-containing agents prior to most surgical procedures and for all wound care.
We are not aware of any data demonstrating superiority of topical agents containing quaternary ammonium such as benzathonium chloride (eg, dermal skin cleanser). Some quaternary ammonium compounds also contain surfactants that facilitate cleansing dirt and debris from the skin. However, in laboratory tests, topical quaternary ammonium components are
bacteriostatic. We prefer agents that are bactericidal and known to rapidly kill pathogenic bacteria on contact. In addition, contamination of quaternary ammonium compounds by organisms such as Pseudomonas aeruginosa is a rare but well-recognized phenomenon and risk. This contamination is especially likely to occur when large-volume containers are used to store stock solutions that are dispensed into smaller containers.
bacteriostatic. We prefer agents that are bactericidal and known to rapidly kill pathogenic bacteria on contact. In addition, contamination of quaternary ammonium compounds by organisms such as Pseudomonas aeruginosa is a rare but well-recognized phenomenon and risk. This contamination is especially likely to occur when large-volume containers are used to store stock solutions that are dispensed into smaller containers.
Topical solutions (including povidone-iodine and chlorhexidine) that contain alcohol have the best and fastest antibacterial effects. However, we advise against using alcohol-containing solutions for routine care of cuts and abrasions for the following reasons:
These solutions cause burning when applied to nonintact skin.
These solutions predictably cause drying of the skin.
We advise against the use of iodophors such as povidone-iodine (Betadine) for routine care of cuts and abrasions for the following reasons:
Although iodophors are generally safe and well tolerated, these compounds are more likely to cause contact dermatitis than other commonly used skin antiseptics.
Iodophor solutions are prone to becoming contaminated (especially with P. aeruginosa) when stock solutions are stored in large bottles. Contaminated Betadine solutions have been associated with numerous outbreaks of healthcare-associated infections, particularly when used prior to injections or other invasive procedures. Note, this risk is eliminated when single-unit doses of iodophors are used (eg, individually wrapped, premoistened swabs).
 Recommendation 3 Apply mupirocin or silver-containing ointments to cuts and scrapes as part of standard care.
Recommendation 3 Apply mupirocin or silver-containing ointments to cuts and scrapes as part of standard care.Rationale: Many athletic trainers and medical professionals routinely apply antibacterial ointments prior to the placement of a clean, dry dressing. These agents are safe and effective. Although topical antibacterial ointments are probably not required for every cut or abrasion, we recommend their use for most skin injuries, especially if extensive, deep, or otherwise a concern. Choices for topical therapy include the following:
Mupirocin
Silver sulfadiazine creams and silver nanoparticle creams
Bacitracin
Bacitracin/polymyxin B or bacitracin, neomycin, and polymyxin B
Of the four therapeutic options listed above, we prefer mupirocin for the following reasons:
It has established activity against MRSA, other strains of S. aureus, and common skin pathogens such as streptococci.
It is safe and has few, if any, significant side effects.
MRSA is the predominant pathogen causing skin and soft-tissue infection in the United States34; thus, using an agent with proven activity against MRSA for primary prevention is logical.
Although emergence of antimicrobial resistance is a concern when mupirocin is used, the likelihood that regular use of mupirocin as topical therapy will result in the emergence of resistance in a small closed population such as an athletic team is unlikely.
Silver-containing creams and ointments can be used in place of mupirocin but are typically more costly. While older silver sulfadiazine creams frequently became dry, crusty, and difficult to use, these issues have been obviated by new nanoparticle formulations of silver ointment.
Bacitracin topical activity is inferior to mupirocin against MRSA and other common bacterial pathogens.35
Bacitracin, bacitracin/polymyxin B, neomycin, double-antibiotic, and triple-antibiotic creams are often used for topical therapy. However, we prefer mupirocin over these other agents for the following reasons:
Bacitracin topical activity is inferior to mupirocin against MRSA and other common bacterial pathogens.35
Bacitracin, neomycin, and polymyxin B and bacitracin/polymyxin B ointments are not effective against MRSA and can lead to allergic reactions that may be confused with an infection.
Rationale: We recognize that there are no reliable published data to guide the choice of dressings for wounds or large areas of skin damage. Thus, the choice and type of dressing to apply to skin injuries is primarily a matter of clinical judgment and local preference. Although Band-Aid and other “breathable” bandages are fine for small and minor skin injuries, hydrocolloid dressings such as DuoDERM are preferable to gauze for deeper and more extensive skin injuries. Hydrocolloid dressings protect injured skin from drying and can be left in place for longer periods of time. Finally, these products are easy and painless to remove. Hydrocolloidal dressings containing CHG are also available, but few and limited data are currently available to assess their superiority compared to standard hydrocolloid dressings.
We are aware of increasingly advanced wound dressings, such as “electroceutical” wound dressings that generate a low-level electrical field in the bandage when wound exudate is present. For example, silver-zinc redox-coupled electroceutical wound dressings can disrupt Pseudomonas biofilm formation in laboratory conditions36 and can reduce costs in chronic wound patients when coupled with negative-pressure wound therapy.37 To date, however, we are unable to determine if these dressings offer advantages over standard dressings and disinfection strategies described above.
 Recommendation 5 Ensure all materials required for effective wound therapy are readily available and easily accessible via a “wound care cart.”
Recommendation 5 Ensure all materials required for effective wound therapy are readily available and easily accessible via a “wound care cart.”Rationale: A central theme throughout our manual is to “make it easy to do the right thing” through strategic placement of materials and organization of strategies (eg, hand hygiene or influenza vaccines). Wound care can be made easier through the use of a well-organized “wound care cart” or designated wound care area that provides easy access to all necessary materials and equipment.
HYDROTHERAPY
 Recommendation 1 Prevent players with open wounds from entering pools unless the wounds are covered with an impermeable dressing.
Recommendation 1 Prevent players with open wounds from entering pools unless the wounds are covered with an impermeable dressing. Recommendation 3 Prevent players with known MRSA infections from entering the pools until the infection is completely resolved.
Recommendation 3 Prevent players with known MRSA infections from entering the pools until the infection is completely resolved.Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree