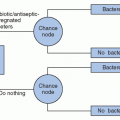
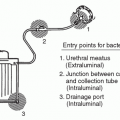
Description (Reference) |
Cause of the Events |
Corrective Measures/Recommendations |
CHEMICAL CONTAMINATION OF DIALYSATE |
Aluminum intoxication and seizures in seven patients (281) |
Exhausted deionization tanks unable to remove aluminum in incoming tap water |
Monitor deionization tanks daily; install reverse osmosis unit |
Aluminum intoxication, neurologic symptoms, dementia and elevated serum aluminum levels in 64 patients; three deaths (174) |
Aluminum pump was used to transfer acid concentrate to the treatment area |
Utilize components in the fluid distribution systems that are compatible and do not leach into the dialysate |
Elevated serum aluminum levels detected in 10 patients during routine screening (176) |
Replacement transfer pump used to pump acid concentrate from 55 gallon drum into jugs used at the machine contained aluminum components |
Contact manufacturer of acid concentrate for a compatible pump; utilize components in the fluid distribution systems that are compatible and do not leach into the dialysate |
16 patients developed nausea, vomiting, chills, some develop fever; two deaths (282) |
Water used to prepare dialysate contained volatile organic compounds (CS2, CH3S, etc.); multiple causes: citric acid used to adjust incoming water pH to aid in removal of chlorine by carbon tanks; water treatment system not functioning properly; microbial quality of water above AAMI limits |
Suspend injection of citric acid (use another pH additive); redesign and replace water treatment system |
Hemolytic anemia in 41 patients (177) |
Facility increased capacity of water treatment system without adjusting size of pretreatment carbon beds; potable water supplier uses monochloramine as residual disinfectant, which was not removed completely by the carbon tank |
Use larger carbon beds to give adequate empty bed contact time to remove chloramines; monitor for total chlorine breakthrough after first carbon tank |
Fluoride intoxication in eight patients; one death (46) |
Accidental spill of hydrofluosilic acid at drinking water plant lead to excessive fluoride levels in water entering a dialysis unit; water treatment at the dialysis facility only included softening |
Install reverse osmosis unit |
Fluoride intoxication in nine patients; three deaths (45,283) |
Exhausted deionization tanks discharged a bolus of fluoride |
Deionization tanks should be monitored with temperature compensated resistivity alarms that include both visual and audible alarms |
Formaldehyde intoxication in five patients; one death (178) |
Disinfectant not properly rinsed from the distribution system |
Eliminate stagnant flow areas; test for residual disinfectant |
Decreased hemoglobin in three pediatric dialysis patients (284) |
Hydrogen peroxide used to disinfect the system not adequately rinsed from the system; facility used flat bottom storage tank that could not be drained |
Thoroughly rinse disinfectant from the system; use an appropriate residual test kit; installation of a storage tank with conical bottom and outflow at lowest point would allow for storage tank to empty completely |
116 of 130 patients (89%) at a dialysis center in Brazil, had visual disturbances, nausea, and vomiting associated with hemodialysis and 50 died; following the initial investigation it was reported an additional 26 patients had died of liver failure (285,286) |
Water contaminated with cyanobacterial toxins not removed by water treatment system; patients exposed to dialysate containing microcystin-LR |
Have finished drinking water supplied to the facility; important to appropriately design (based on system feed water), install, monitor, and maintain water treatment systems |
Severe hypotension in nine patients (287) |
Dialysate contaminated with sodium azide used as a preservative from new ultrafilters (filters were labeled “not for medical use”) |
Rinse system after modification or installation of new components |
BACTEREMIA, FUNGEMIA, OR PYROGENIC REACTIONS NOT RELATED TO DIALYZER REUSE |
Pyrogenic reactions in 49 patients (31) |
Untreated tap water contained high levels of endotoxin |
Install a reverse osmosis water treatment system |
Pyrogenic reactions in 45 patients (30) |
Inadequate disinfection of the fluid distribution system |
Increase disinfection frequency and disinfectant contact time |
Pyrogenic reactions and bacteremia in five patients; two patients had bacteremia (one with Klebsiella pneumonia, the other had both K. pneumonia and Pseudomonas aeruginosa) (288) |
Two weeks before the outbreak a pump that pumped sodium hypochlorite through the distribution and machines failed; the distribution system and machines were inadequately disinfected; P. aeruginosa, K. pneumonia, and Pantoea (Enterobacter) agglomerans were recovered from water, dialysate, and other environmental sources |
Routine disinfection of the fluid distribution system and dialysis machines following repair of the pump ended the outbreak |
Pyrogenic reactions in 14 patients; two with bacteremia; one death (29) |
Reverse osmosis water storage tank contaminated with bacteria |
Remove or properly maintain and disinfect storage tank |
Bacteremia in 35 (51.5%) patients with central venous catheters (CVCs) (142) |
CVCs used as primary access; median duration of catheter use among affected patients was 311 days; improper aseptic techniques |
Use CVCs only when necessary (i.e., bridge to maturing fistula or graft or as an access of last resort); use appropriate aseptic techniques when inserting CVC and performing catheter care |
Three pyrogenic reactions and seven Enterobacter cloacae bloodstream infections…(111) |
Incompetent check valves in the waste handling option (WHO-port) of one type of dialysis machine allowed backflow of spent dialysate into patient blood lines during circuit priming and dialysis initiation; Machines and ports were contaminated with E. cloacae |
Routine maintenance, disinfection, and valve competency testing of the WHO should be carried out |
Gram-negative bacteremia in 1 patient (6 E. cloacae, 4 P. aeruginosa, 2 Escherichia coli; 2 had polymicrobial bacteremia) (112) |
Incompetent check valves in the waste handling option (WHO-port) of one type of dialysis machine allowed backflow of spent dialysate into patient blood lines during circuit priming and dialysis initiation; machines and ports were contaminated with E. cloacae and P. aeruginosa |
Routine maintenance, disinfection, and valve competency testing of the WHO should be carried out |
Outbreak of pyrogenic reactions and gram-negative bacteremia in 11 patients (4 had bacteremia) (28) |
Water distribution system was not routinely disinfected and machines were not disinfected in accordance with manufacturer’s instructions; water and dialysate cultures were performed using a 10-3 calibrated loop on blood agar plates—results were often reported as no growth |
Disinfect machines according to manufacturer’s instructions; include water distribution system in the weekly disinfection of the RO system. Do not use calibrated loops. These are not sensitive to detect when AAMI limits have been reached; use spread plate or membrane filtration techniques and Trypticase Soy Agar (TSA) for testing (94) |
Phialemonium curvatum access infections in four hemodialysis patients, two patients died of systemic disease (289,290). |
Phialemonium species were only recovered from the condensation drip pans under the blowers of the HVAC system that supplied air to the dialysis center. Observations at the facility noted some irregularities with site prep for needle insertion; all infected patients had synthetic grafts. |
Review infection control practices, clean and disinfect the HVAC system where water accumulates; perform surveillance on facility patients. Observe proper aseptic technique during access cannulation. |
Fungemia with Phialemonium curvatum in two maintenance hemodialysis patients (291). |
Both patients were dialyzed on the same machine. Facility used machines with a waste handling port; Phialemonium was isolated from reverse osmosis unit (WHO-port and valves were not available for culture) |
WHO ports have been previously been associated with cases of bacteremia; remediation of water distribution system and maintenance and discontinuing use of WHO ports ended the outbreak. |
ADVERSE EVENTS ASSOCIATED WITH HEMODIALYZER REUSE |
Mycobacterial infections in 27 patients (292) |
Inadequate concentration of dialyzer disinfectant |
Increase concentration of formaldehyde used to disinfect dialyzers to 4% |
Mycobacterium abscessus infection in five patients treated with high-flux hemodialyzers (38) |
Inadequate dialyzer disinfection due to over diluted disinfectant; inadequate disinfection of water distribution system |
Use higher concentration of dialyzer disinfectant; follow manufacturer’s label; more frequent disinfection of the water treatment system |
Bacteremia and pyrogenic reactions in six patients (293) |
Dialyzer disinfectant diluted to improper concentration |
Use disinfectant at recommended dilution and verify concentration |
Bacteremia in six patients (CDC, unpublished data) |
Inadequate concentration of dialyzer disinfectant; water for reuse did not meet AAMI standards |
Use AAMI quality water for reprocessing hemodialyzers; ensure proper disinfectant concentration in reprocessed dialyzers |
Nine pyrogenic reactions and five gram-negative bacteremias in 11 patients undergoing dialysis (67) |
Inadequate mixing of dialyzer disinfectant |
Thoroughly mix dialyzer disinfectant and verify proper concentration |
Bacteremia in 33 dialysis patients at two dialysis clinics (104,294) |
New dialyzer disinfectant created holes in dialyzer membranes |
Change disinfectant (manufacturer withdrew product from the market place) |
Six chronic hemodialysis patients acquired bloodstream infections (BSIs) with Klebsiella pneumoniae of the same serotype and similar plasmid profile (72) |
Dialyzers contaminated during removal and cleaning of headers with a common gauze pad; staff not routinely changing gloves; dialyzers not reprocessed for several hours after disassembly and cleaning |
Do not use gauze or similar material to remove clots from header; rinse headers with treated water and disinfect header components before reassembly; change gloves frequently; reprocess dialyzers immediately after rinsing and cleaning |
Pyrogenic reactions in three high-flux dialysis patients (295) |
Dialyzers reprocessed with two different disinfectants; water used for reprocessing hemodialyzers did not meet AAMI standards |
Do not disinfect dialyzers with multiple disinfectants; disinfect water treatment system more frequently |
Pyrogenic reactions in 14 high-flux dialysis patients; one death (296) |
Dialyzers rinsed with tap water; water for reuse did not meet AAMI standards |
Do not rinse or clean dialyzers with city tap water; use treated water that has been maintained to meet AAMI standards |
Pyrogenic reactions in 18 patients (69) |
Dialyzers reprocessed with city tap water containing high levels of endotoxin; water did not meet AAMI standards |
Do not rinse or clean dialyzers with city tap water; use treated water that has been maintained to meet AAMI standards |
Pyrogenic reactions in 22 patients (71) |
Water for reuse did not meet AAMI standards; improper microbial assay method was employed to monitor dialysis fluids |
Use correct microbial assay procedure; disinfect water treatment system to maintain microbial quality of treated water and meet AAMI standards |
Bacteremia with gram-negative water bacteria in 13 patients (297) |
Water distribution system had flow and pressure problems; B. cepacia and Ralstonia spp. were isolated from water and dialysate. Water was sporadically above AAMI limits; no bacteremia in patients when reuse was suspended |
Evidence that system had biofilm present; suggestions were made to consider replacing the distribution loop which would also help with the flow and pressure problems. Scrapings of section of pipe that was removed had evidence of biofilm. |
Acute hypersensitivity/allergic reactions |
Acute allergic reactions in hundreds of patients using reprocessed hemodialyzers in at least 31 dialysis centers (70,170). |
Related to the use of angiotensin-converting-enzyme (ACE) inhibitors, and possibly to chemicals used in cleaning dialyzer or inadequate disinfectant rinse out. |
No specific recommendations were made. |
Nationwide outbreak of severe allergic-type reactions that were first detected in a single hemodialysis facility. 152 cases identified (21). |
Use of heparin manufactured by Baxter Healthcare was the factor most strongly associated with reactions; heparin was found to be contaminated with over-sulfated chondroitin sulfate (OSCS) |
Manufacturer recalled all lots implicated. |
Hemolysis and other miscellaneous investigations |
Severe hemolytic events among dialysis patients in dialysis facilities in five states; at least three deaths (24) |
Three lots of bloodlines had a manufacturing defect that caused approximately 10% of the bloodlines to have occlusions; these inclusions blocked between 20% and 80% of the internal diameter of the lines |
Manufacture recalled several lots of blood tubing sets |
State medical examiner’s office notified CDC of a cluster of deaths due to vascular access hemorrhage (18) |
Reviewed CMS and medical examiner records; Identified 88 confirmed cases; risk factors included: presence of an arteriovenous graft, access-related complications within 6 months of death, hypertension |
Nephrologists should review primary and secondary prevention measures with their patients; particularly those who have had vascular access complications |
CDC notified of 28 patients with nephrogenic systemic fibrosis (NSF) at one medical center (19). |
14 of 19 confirmed cases had receipt of gadolinium-containing contrast solution the year before onset; gadolinium contrast exposure was associated with NSF in a dose dependent manner |
Receipt of gadolinium-containing contrast solution should be avoided when possible in patients with end stage renal disease, particularly those receiving peritoneal dialysis. |
TRANSMISSION OF VIRAL AGENTS |
26 patients seroconvert to HBsAg-positive (185) |
Blood leaks in coil dialyzers and use of recirculating bath dialysis machine |
Separation of HBsAg-positive patients and equipment from susceptible patients; pressure-leak testing of reused dialyzers was inadequate |
19 patients and one staff member seroconvert to HBsAg-positive in a 14-month time frame (182) |
No specific cause identified; false-positive HBsAg test results caused some susceptible patients to be dialyzed with infected patients |
Laboratory confirmation of HBsAg-positive results; strict adherence to glove use and use of separate (dedicated) equipment |
40 patients (24 in-center, 12 home dialysis, and 4 home training) and 10 staff members convert to HBsAg-positive during a 10 month period (186) |
Home patients were excluded from the investigation due to paucity of data; staff were not wearing gloves; environmental surfaces not routinely cleaned/disinfected; improper handling of sharps |
Separation of HBsAg-positive patients and their dedicated equipment from susceptible patients; proper precautions by staff (e.g., gloving, handling of needles and sharps, and disinfection of environmental surfaces) |
13 patients and one staff member convert to HBsAg-positive in a single month (180) |
Extrinsic contamination of intravenous hypertonic glucose being prepared adjacent to an area where blood work was handled |
Separate medication preparation area and blood processing for diagnostic tests |
10 patients seroconvert to HBsAg-positive in a 1-month period (179) |
Extrinsic contamination of bupivacaine that was shared between HBsAg-positive and susceptible patients |
No sharing of equipment, supplies, and medications between patients |
Eight patients seroconvert to HBsAg-positive over a five month period (CDC, unpublished data) |
Sporadic screening for HBsAg; HBsAg-positive patient not isolated; major bleeding incident with environmental contamination |
Perform monthly screening of patients for HBsAg; isolate HBsAg-positive patients with dedicated equipment and staff; vaccinate all susceptible patients |
Seven patients seroconvert to HBsAg-positive during a 3-month period (184) |
Same staff caring for HBsAg-positive and HBsAg-negative patients |
Separate HBsAg-positive patients from HBsAg-negative patients; same staff should not care for both HBsAg-positive and negative patients on the same shift |
Eight patients convert to HBsAg-positive during 1 month (187) |
Not consistently using pressure transducer protectors; same staff members cared for both HBsAg-positive and negative patients on the same shift |
User pressure transducer protectors and replace after each patient; same staff members should not care for both HBsAg-positive and -negative patients on the same shift |
14 patients seroconverted to HBsAg-positive during a 6-week period (188) |
Failure to review laboratory results of admission and monthly HBsAg testing; inconsistent hand hygiene and use of gloves; adjacent clean and contaminated areas; <20% of patients vaccinated |
Proper infection control precautions for dialysis units; routine review of laboratory testing; hepatitis B vaccination of all dialysis patients |
Seven patients seroconverted to HBsAg-positive during a 2-month period (188) |
Staff members cared for both HBsAg-positive and negative patients on the same shift; common medication and supply carts were moved between stations, and multidose vials were shared; no patients were vaccinated |
Dedicated staff for HBsAg-positive patients; no sharing of medications or supplies between patients; centralized medication and supply areas; vaccinate all patients against hepatitis B. |
Four patients seroconverted to HBsAg-positive during a 3-month period (188) |
Transmission appeared to occur during hospitalization at an acute care facility |
Vaccinate all patients to protect against hepatitis B |
11 patients seroconverted to HBsAg-positive during a 3-month period (188) |
Staff, equipment, and supplies were shared between HBsAg-positive and -negative patients |
Dedicate staff for HBsAg-positive patients; no sharing of supplies or medications between patients; vaccinate all patients to protect against hepatitis B |
Two patients seroconvert to HBsAg-positive during a 4-month period (181) |
Same staff members cared for HBsAg-positive and -negative patients; no patients were vaccinated |
Dedication of staff for HBsAg-positive patients; hepatitis B vaccination for all patients |
36 patients with elevated liver enzymes consistent with non-A non-B hepatitis (298) |
Environmental contamination with blood |
Monthly liver enzyme screening; use proper infection control precautions (e.g., gloving, environmental cleaning) |
35 patients with elevated liver enzymes over a 22-month period; 82% of cases were anti-HCV positive (189) |
Inconsistent use of infection control precautions, especially hand hygiene and glove use |
Strict compliance with aseptic techniques and infection control precautions for all dialysis patients |
Seven of 51 patients seroconvert to anti-HCV positive (239,242) |
Dialysis on same machine immediately after patient with chronic HCV infection; preparation of multidose intravenous medication at the dialysis station; failure to routinely clean dialysis machine or dialysis station surfaces between patients |
Strict compliance with aseptic techniques and infection control precautions for all dialysis patients; routine anti-HCV testing |
HCV infection developed in 5/95 patients (239,242) |
Dialysis on same machine immediately after patient with chronic HCV infection; preparation of multidose intravenous medication at the dialysis station; failure to routinely clean dialysis machine or dialysis station surfaces between patients; use of a mobile medication or supply cart that was moved between dialysis stations |
Strict compliance with aseptic techniques and infection control precautions for all dialysis patients including environmental cleaning and disinfection of the dialysis machine and station, medication preparation in a separate clean area; and discontinue use of mobile carts to deliver supplies and medications between dialysis stations |
Incident HCV infection in 3/24 patients (239,242) |
Patients dialyzed at the station adjacent to that of patients with chronic HCV infection; use of a mobile medication or supply cart that was moved between dialysis stations |
Do not use mobile medication or supply carts that move from station to station; deliver clean supplies and medications to each station individually; follow recommended infection control precautions for all dialysis patients |
Incident HCV infection in 7/64 patients (239,242) |
Patients dialyzed at the station adjacent to that of patients with chronic HCV infection; receipt of intravenous medication from vial (including single-dose vials) used for <1 patient; dialysis during the shift following that of patient with chronic HCV infection but not with the same machine; routine cleaning and disinfection of environmental surfaces not performed; mobile carts used for delivery of medications and supplies |
Do not use mobile medication or supply carts that move from station to station; single-dose vials should not be shared; clean and disinfect dialysis machine surfaces (including prime buckets) between patients, clean and disinfect station area before setting up and priming next patient’s dialyzer and extracorporeal circuit; follow infection control precautions for all dialysis patients |
11/75 patients develop HCV infection (239) |
Preparation of injections in contaminated environment; failure to separate clean and contaminated areas; failure to change gloves and perform hand hygiene after handling contaminated dialysis equipment |
Follow recommendations for infection control precautions for all dialysis patients, hand hygiene, and environmental infection control |
7/183 patients develop HCV infection (239) |
Use of mobile cart to deliver injectable medications to multiple patients; reuse of single-dose vials of epoetin alfa on multiple patients; failure to clean dialysis equipment between patients |
Do not use mobile carts to deliver injectable medications or supplies; clean and disinfect external surfaces of the dialysis machine and patient station between each patient; follow recommendations for infection control precautions for all dialysis patients |
HCV infections develop in nine patients (191) |
Multiple breaches in infection control practices including inadequate cleaning and disinfection of the machine and station (visible blood was present on dialysis chairs, machines, and floor); inappropriate glove use, inconsistent hand hygiene; and deficiencies in training |
Patients transferred to other facilities and dialysis center closed by the State regulatory agency, after several attempts made to correct infection control deficiencies |
HCV infections in eight patients (240) |
New infections were seen in patients dialyzed after or on the same machine a patient with chronic HCV infection. Infection control lapses in medication handling; lack of cleaning of access ports on CVCs; heparin and saline were prepared at dialysis stations; medications prepared and delivered using mobile carts. Patients were “bled on” to WHO-port and WHO-port was not disinfected between patients. Environmental cleaning and disinfection were suboptimal; routine disinfection of machines conducted with patient still in dialysis chair. |
Follow recommended infection control precautions for dialysis setting; proper use of gloves; perform hand hygiene and use new pair of gloves before performing vascular access care; use antiseptic to disinfect CVC hubs and injection ports prior to accessing; prepare medications in an “clean” area dedicated for that purpose; parenteral medications should never be prepared at the dialysis station; if facility chooses to use medication and supply carts, these should remain stationary in a designated clean area; use an EPA-registered disinfectant for cleaning blood spills; |
21 patients in a hospital-based outpatient dialysis unit develop HCV infection (192) |
Breaches in infection control practices identified by the local health department included: medication preparation and delivery; suboptimal environmental cleaning and disinfection |
Strict compliance with aseptic techniques and infection control precautions for all dialysis patients including environmental cleaning and disinfection of the dialysis machine and station, medication preparation in a separate clean area; and discontinue use of mobile carts to deliver supplies and medications between dialysis stations |
Two HCV infections in an outpatient hemodialysis facility (193) |
No breaches in infection control identified but epidemiologic and laboratory evidence suggested in-center transmission. |
Follow recommendations for preventing infections |
HCV infection in six dialysis patients (194) |
Limited separation between “clean” and “contaminated” areas; lab specimens processed in the same location as medication preparation; overcrowding; significant lapses in cleaning and disinfection were observed; blood glucose meters and clamps were not routinely disinfected between patients; clean supplies obtained by staff wearing contaminated gloves; blood glucose meters returned after use to “clean” supply table; ports on blood tubing and catheters not routinely disinfected before accessing |
Facility should identify a staff member responsible for infection control; conduct regular meetings to discuss infection control issues; insure that monthly serology results are reviewed promptly; segregate clean and contaminated areas; consider facility design and work flow; consider redesign of treatment area to provide sufficient space between stations, and clean and contaminated areas; insure proper use of gloves and hand hygiene; items used on more than one patient should be cleaned and disinfected between each patient; patient’s station (chair and machine) should be cleaned and disinfected with a EPA-registered disinfectant |
13 patients at one dialysis center in Colombia tested positive for HIV in 12 months; 9/13 seroconverted from negative to positive during this period and 2/9 had other risk factors (195) |
Facility reprocessed vascular access needles by soaking in a common container containing a low-level benzalkonium chloride disinfectant; needles could have been shared among patients |
Access needles are single-use only; if they are to be reused then they should be cleaned and sterilized between uses. |
HIV infection in 39 patients at two hemodialysis centers in Egypt (196) |
Practices that resulted in sharing of syringes among patients were observed at both centers |
Do not share syringes. Follow infection control recommendations for preventing infections among dialysis patients |