Abstract
Survival in children with cancer has increased dramatically over the past five decades. This progress is due not only to advances in oncologic therapies, but also to advances in supportive care and an improved ability to manage life-threatening complications. Oncologic emergencies can occur as an initial manifestation of cancer, as a side effect of cancer therapy, or at the time of recurrence or progression. The following emergencies are addressed in this chapter:
- •
Metabolic emergencies including hyperleukocytosis, tumor lysis syndrome, and associated electrolyte derangements.
- •
Cardiothoracic emergencies including superior vena cava syndrome and mediastinal masses.
- •
Acute abdominal processes.
- •
Renal dysfunction and hypertension.
- •
Neurologic emergencies.
- •
Endocrinologic emergencies.
- •
Treatment-related emergencies.
Keywords
Tumor lysis syndrome, hyperleukocytosis, hypercalcemia, hyponatremia (SIADH), superior vena cava syndrome, gastritis, pancreatitis, bowel obstruction, constipation, ileus, typhlitis, spinal cord compression, seizure, alteration in mental status, cerebrovascular accident, increased intracranial pressure
Survival in children with cancer has increased dramatically during the past five decades. This progress is due to advances in specific oncologic therapies as well as supportive care, and an improved ability to manage life-threatening complications. Oncologic emergencies can occur as an initial manifestation of cancer, as a side effect of therapy, or at the time of progression or recurrence of the disease.
Excellent cancer management requires mastery of the following:
- •
Metabolic emergencies including hyperleukocytosis, tumor lysis syndrome (TLS), and associated electrolyte derangements.
- •
Cardiothoracic emergencies including superior vena cava syndrome and mediastinal masses.
- •
Acute abdominal processes.
- •
Renal dysfunction and hypertension.
- •
Neurologic emergencies.
- •
Endocrine emergencies.
- •
Treatment-related emergencies.
Metabolic Emergencies
Hyperleukocytosis
Hyperleukocytosis is defined as a total white cell count greater than 100,000/mm 3 . Hyperleukocytosis is seen at presentation in 9–13% of children with acute lymphocytic leukemia (ALL) and 5–22% of children with acute myeloid leukemia (AML). It occurs in an even higher percentage of patients with chronic myeloid leukemia (CML).
The presence of a high number of blasts in the microcirculation leads to sludging, which interferes with oxygenation of local tissue, ultimately leading to tissue ischemia. This in turn leads to an adhesive reaction between abnormal vascular endothelium and the circulating blasts worsening leukostasis, thrombosis, and leading to secondary hemorrhage. The higher metabolic rate of the blasts and the local production of cytokines also contribute to tissue hypoxia. Thrombi in the circulation lead to vascular damage and parenchymal ischemia manifested as pulmonary or cerebrovascular hemorrhage and edema.
Myeloblasts are larger, less deformable, and more adherent to vasculature than lymphoblasts. Due to these intrinsic properties, leukostasis and thrombosis are far more prevalent in AML than in ALL. At presentation, patients with AML are more likely to have intracranial hemorrhage or thrombosis or pulmonary hemorrhage and leukostasis, whereas ALL is more likely to lead to metabolic disturbance from tumor lysis syndrome.
As leukostasis is associated with early morbidity and mortality, any patient presenting with a white blood cell count greater than 50,000/mm 3 should be evaluated closely for clinical signs and symptoms of leukostasis.
Clinical Features
- •
Central nervous system (CNS): blurred vision, confusion, somnolence, delirium, stupor, coma and papilledema.
- •
Computed tomography (CT) may reveal hemorrhage or leukemic infiltrate.
- •
- •
Pulmonary: tachypnea, dyspnea, hypoxia.
- •
Chest radiograph may reveal varying degree of diffuse interstitial or alveolar infiltrates.
- •
- •
Genitourinary: oliguria, anuria, rarely priapism.
- •
Vascular symptoms which include disseminated intravascular coagulation (DIC), retinal hemorrhage, myocardial infarction, and renal vein thrombosis.
Risk Factors
- •
WBC counts >200,000/mm 3 in AML, and >300,000/mm 3 in ALL and CML.
- •
Age less than 1 year.
- •
M4, M5 AML (higher lysozyme activity).
- •
Cytogenetic abnormalities including MLL 11q23, t(4:11), inv16, Philadelphia positive, and Philadelphia-like ALL and FLT3-ITD.
Tumor Lysis Syndrome
TLS arises due to the rapid release of intracellular metabolites (such as phosphorous, potassium, and uric acid) from dying tumor cells in quantities that exceed the excretory capacity of the kidneys.
In patients with a high tumor burden or rapid cell proliferation, such as Burkitt or Burkitt-like lymphoma, B-cell ALL, and T-cell leukemia or lymphoma, significant cell death and release of intracellular ions, even prior to chemotherapy initiation, may result in the following metabolic complications:
- •
Hyperuricemia.
- •
Hyperkalemia.
- •
Hyperphosphatemia.
- •
Hypocalcemia.
- •
Renal insufficiency/failure.
If not successfully treated, TLS can result in cardiac arrhythmias, renal failure, seizures, coma, DIC, and death.
Diagnostic Criteria for Laboratory TLS and Clinical TLS
Laboratory TLS (LTLS): The presence of two or more abnormal serum values at presentation (i.e., uric acid ≥8 mg/dl, potassium ≥6 mg/dl, phosphate ≥2.1 mmol/l, calcium ≤1.75 mmol/l, creatinine >normal).
Clinical TLS (CTLS): Presence of LTLS and one or more of the following clinical complications: renal insufficiency, cardiac arrhythmias, seizures or sudden death.
Recognition of risk factors, close monitoring, and appropriate preventative intervention are vital in managing TLS. Table 32.1 lists patient stratification by risk for TLS for various types of cancer.
| Type of cancer | Risk | ||
|---|---|---|---|
| High | Intermediate | Low | |
| NHL | Burkitt, lymphoblastic | DLBCL | Indolent NHL |
| ALL | WBC≥100,000/mm 3 | WBC 50,000–100,000/mm 3 | WBC≤50,000/mm 3 |
| AML | WBC≥50,000/mm 3 | WBC 10,000–50,000/mm 3 | WBC≤10,000/mm 3 |
| Other hematologic malignancies (including CML) and solid tumors | Rapid proliferation with expected rapid response to therapy | Remainder of patients | |
Prevention and Management of TLS
Prevention
- 1.
Fluids and hydration
- a.
Promote excretion of uric acid and phosphorus; increase glomerular filtration rate (GFR) and renal blood flow.
- b.
Hydration at a rate of >2 l/m 2 /day should start 24 h before chemotherapy.
- c.
Urine output goal of 3 ml/kg/h should be maintained. Diuretics may be needed to maintain this urine output.
- a.
- 2.
Allopurinol
- a.
A xanthine analog which blocks conversion of xanthine and hypoxanthine to uric acid; works as a competitive inhibitor of xanthine oxidase.
- b.
Does not remove preformed uric acid.
- c.
Slow reduction in uric acid levels.
- d.
Decreased risk of uric acid crystallization in kidney tubules.
- e.
Adverse effects include hypersensitivity reaction and a build up of xanthine and hypoxanthine, which may lead to renal insufficiency as well.
- a.
- 3.
Rasburicase
- a.
Indicated for patients at high risk of TLS.
- b.
Assess patient’s G6PD status or risk of having G6PD, as may cause methemoglobinemia or severe hemolytic anemia.
- c.
May require reinitiation of allopurinol several days after rasburicase.
- a.
Active management
Table 32.2 outlines the management of hyperleukocytosis and TLS.
| Management/objective | Guidelines |
|---|---|
| Aggressive hydration | Usually 2–4 times maintenance (alkalinization is avoided so as not to precipitate xanthine calculi and cause acute kidney injury) |
| Diuresis | Furosemide 0.5–1 mg/kg |
| Mannitol 0.5 g/kg can be used if patient has oliguria unresponsive to increased hydration and furosemide | |
| Uric acid reduction | (1) Allopurinol 300 mg/m 2 /day or 10 mg/kg/day PO (maximum dose 800 mg/day) or 200 mg/m 2 /day IV (maximum dose 600 mg/day) may be used if available; (2) Rasburicase (recombinant urate oxidase) 0.15–0.2 mg/kg/day IV, the dose can be repeated |
| Leukocyte reduction | Leukapheresis or exchange transfusion (for infants) can be used at any WBC count if the patient is symptomatic. In asymptomatic patients, leukapheresis should be considered if the initial white cell count is greater than 200,000/mm 3 in AML or 300,000/mm 3 in ALL or CML |
| Transfusion | Platelet transfusion to keep platelet count over 20,000/mm 3 to decrease the risk of intracranial hemorrhage. Avoid packed RBC transfusions if cardiovascularly stable, because it increases blood viscosity. Fresh frozen plasma transfusion and administration of vitamin K can be considered if coagulopathy is present prior to any invasive procedures |
| Chemotherapy | Chemotherapy should be started when patient is stabilized and has adequate urine output |
| Dialysis | Dialysis is indicated for progressive renal failure with potassium >6 mEq/l, phosphate >10 mg/dl, oliguria, anuria, or volume overload unresponsive to the above measures |
| Monitor | Electrolytes; calcium, phosphorus, potassium, uric acid, BUN, and creatinine every 4–12 h depending upon risk of TLS. |
| Complete blood counts 1–2 times per day. | |
| Respiratory, CNS, and cardiac monitoring if hyperkalemia or hypocalcemia are present | |
| Imaging | Brain CT with contrast if no renal insufficiency in the presence of neurologic symptoms or signs. MRI, MRA, or MRV should be carried out if thrombosis is suspected |
Management of the Various Metabolic Derangements in TLS
Hyperuricemia
- 1.
Pharmacologic treatment should be based on risk stratification as outlined in Figure 32.1 , which shows a treatment algorithm for the prevention and management of hyperuricemia.
- a.
For low-risk patients: Observation and frequent laboratory monitoring are required. Allopurinol can be initiated as first-line therapy at diagnosis.
- b.
For intermediate-risk patients: Allopurinol should be initiated as first-line therapy at diagnosis. Rasburicase should be considered if laboratory or clinical evidence of TLS is present despite allopurinol.
- c.
For high-risk patients: Consider the use of rasburicase at diagnosis for tumors with high proliferative index, large tumor burden, or tumors that are highly chemo-sensitive. Rasburicase should also be considered as first-line therapy for patients with clinical evidence of TLS despite risk stratification. Rasburicase must be used with caution in patients with unknown G6PD status and/or at high risk for having G6PD deficiency.

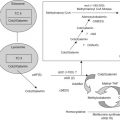
Figure 32.1
Treatment algorithm for the prevention and management of hyperuricemia. The treatment approach for low-risk patients is close observation with or without hydration. For those with intermediate risk, rasburicase is recommended if hyperuricemia develops despite prophylactic treatment with allopurinol. Vigorous hydration is recommended for all patients in the intermediate-to-high-risk groups, or those with diagnosed TLS. The use of rasburicase is recommended for the treatment of patients with hyperuricemia associated with diagnosed TLS, or in the initial management of patients considered to be at high risk of developing TLS. TLS, tumor lysis syndrome; NHL, non-Hodgkin lymphoma; LDH, lactate dehydrogenase; DLBCL, diffuse large B-cell lymphoma.
Adapted from Hochberg and Cairo (2008) . Reprinted with permission.
- a.
- 2.
Hydration >2 l/m 2 /day should start before chemotherapy and urine output goal of >100 ml/m 2 /h should be maintained during chemotherapy. Strict measurement of intake and output should be carried out every 2–4 h. Diuretics may be needed to maintain this urine output (i.e., furosemide 0.5–1 mg/kg/dose or mannitol 0.5 g/kg of the 25% solution over 5–10 min repeated every 6 h as needed).
Hyperkalemia
- 1.
Potassium should be avoided unless dangerously low until tumor lysis is controlled.
- 2.
Mild (<6 mEq/l) and asymptomatic:
- a.
Hydration and diuretics as described in hyperuricemia.
- b.
Sodium polystyrene sulfonate (kayexelate) at dose of 1 g/kg every 6 h with sorbitol 50–150 ml will remove 1 mEq of potassium per liter per gram of resin over 24 h.
- a.
- 3.
Moderate to severe hyperkalemia:
- a.
Obtain an EKG.
- b.
Rapid insulin (0.1 U/kg/h) plus glucose (dextrose 0.5 g/kg/h); in emergency cases, 50% dextrose can be used at 1 ml/kg through a central line. Monitor serum glucose closely.
- c.
For life-threatening arrhythmias IV calcium gluconate (100–200 mg/kg) or calcium chloride (10 mg/kg) slow IV infusion rate. The onset of action is within minutes and duration of activity lasts about 30 min (cannot be administered in the same line as NaHCO 3 ).
- d.
NaHCO 3 to stabilize myocardial cell membrane and to reverse acidosis; 1–2 mEq/kg IV. For every increase of 0.1 pH unit, potassium is decreased about 1 mEq/l. Onset of action is within 30 min and duration of action lasts several hours.
- e.
Dialysis should be considered while these rescuing approaches are underway.
- a.
Hyperphosphatemia
- 1.
Lymphoblasts contain four times the amount of phosphate present in normal lymphocytes; consider a low-phosphate diet.
- 2.
Aluminum hydroxide 150 mg/kg/day divided into doses every 4–6 h should be administered. This will prevent absorption of oral phosphate intake, but has little direct effect on lowering serum phosphate. Sevelamer hydrochloride (RenaGel), a noncalcium phosphate binder (400 mg twice daily for older children) can also be used.
- 3.
Hydration necessary to maintain a urine output of ≥3 ml/kg/h.
- 4.
Dialysis may be necessary to lower serum phosphate and prevent metastatic calcium deposition.
Hypocalcemia
- 1.
As the product of serum calcium and phosphate increases over 60 due to hyperphosphatemia, a compensatory hypocalcemia may occur to maintain the calcium phosphate product at 60. At a calcium×phosphate product of >60 renal calcification can occur and exacerbate renal damage.
- 2.
For symptomatic hypocalcemia (e.g., tetany), 10 mg/kg of elemental calcium (i.e., 0.5–1.0 ml/kg of 10% calcium gluconate) should be given. Calcium administration should be discontinued when symptoms resolve. Dialysis should be carried out if hyperphosphatemia persists. (Caution: Do not administer calcium in the same line as NaHCO 3 .)
Renal Dysfunction from Tumor Lysis
Mechanisms of Renal Dysfunction
- 1.
Precipitation of urates in the acid environment of the renal tubules.
- 2.
Precipitation of hypoxanthine when the urine pH exceeds 7.5.
- 3.
Increase in the hypoxanthine levels after starting treatment with allopurinol.
- 4.
Precipitation of calcium phosphate in renal microvasculature and renal tubules when the product of serum calcium and phosphate values exceeds 60.
Indications for Dialysis Include the Following
- 1.
Presence of hyperphosphatemia (>6 mg/dl) and hypercalcemia which promotes deposition in renal interstitium and tubular system, exacerbating kidney damage.
- 2.
An estimated GFR less than 50%.
- 3.
Persistent hyperkalemia with QRS interval widening and/or level exceeding 6 mEq/l.
- 4.
Severe metabolic acidosis.
- 5.
Volume overload unresponsive to diuretic therapy.
- 6.
Anuria and overt uremic symptoms (i.e., encephalopathy).
- 7.
Severe symptomatic hypocalcemia.
- 8.
Hypertension (BP>150/90) and inadequate urine output at 10 h from start of treatment.
- 9.
Congestive heart failure.
Hemodialysis or hemofiltration should be used when renal failure occurs. Continuous renal replacement therapy (CRRT) may be used for hemodynamically unstable patients because it is less likely to exacerbate hypotension. CRRT is not as effective for the treatment of hyperphosphatemia. Peritoneal dialysis should not be used. Dialyzable chemotherapy, such as cyclophosphamide, is given immediately after dialysis and not before. Renal dialysis usually needs to be repeated every 12 h while there is continuous rapid tumor lysis.
Cardiothoracic Emergencies
Superior Vena Cava Syndrome and Superior Mediastinal Syndrome
Superior vena cava syndrome (SVCS) consists of the signs and symptoms of superior vena cava (SVC) obstruction due to compression or thrombosis. This condition is frequently due to a large anterior mediastinal mass compressing the SVC. Rapid growth of the mediastinal mass does not permit the development of effective collateral circulation to compensate and results in the signs and symptoms of compression of the SVC.
Superior mediastinal syndrome (SMS) consists of SVCS with tracheal compression.
The trachea and main stem bronchus are more compressible in children, making them more susceptible to SMS. In pediatrics, the terms SVCS and SMS are often used synonymously.
Etiology
- 1.
Intrinsic causes: vascular thrombosis following the introduction of a catheter, intravascular tumor thrombosis (Wilms, lymphoma).
- 2.
Extrinsic causes: malignant anterior mediastinal tumors
- a.
Hodgkin lymphoma.
- b.
Non-Hodgkin lymphoma.
- c.
Teratoma or other germ cell tumor.
- d.
Thyroid cancer.
- e.
Thymoma.
- a.
Clinical Features
- 1.
Superior vena cava syndrome:
- a.
Swelling, plethora, and cyanosis of the face, neck, and upper extremities.
- b.
Suffusion of the conjunctiva.
- c.
Engorgement of collateral veins.
- d.
Altered mental status.
- a.
- 2.
Superior mediastinal syndrome:
- a.
Respiratory symptoms: cough, hoarseness, dyspnea, orthopnea, wheezing, and stridor. Supine position worsens symptoms.
- b.
Dysphagia.
- c.
Chest pain.
- d.
Altered mental status and syncope.
- a.
Management
- 1.
Extreme care is required in handling the patient. The following may precipitate respiratory arrest:
- a.
Supine position (as for CT or operative procedures)—Do not place patient in recumbent position.
- b.
Medications that cause intercostal muscle relaxation.
- c.
Stress.
- d.
Sedation (conscious sedation, anxiolytics, or general anesthesia). The patient may have to be intubated. Extubation may not be possible until the anterior mediastinal mass has significantly decreased in size. Extracorporeal membrane oxygenation may be required if intubation is not possible.
- a.
- 2.
Diagnosis should be made quickly in the least invasive manner.
- a.
Radiograph of the chest and CT (if tolerated).
- b.
Screening blood work such as CBC, LDH, uric acid, α-fetoprotein, and β-hCG (to screen for germ cell tumors and lymphoma).
- c.
Echocardiogram, to assess anesthesia risk, cardiac function and for possible intravascular thrombus if no evidence of mass on chest radiograph.
- d.
Determine anesthesia risk. If high risk, perform the least invasive technique with local anesthesia (bone marrow, pleurocentesis, pericardiocentesis, lymph node biopsy, or fine-needle aspirate). If low risk use sedation or anesthesia and monitor closely.
- a.
- 3.
Therapy:
- a.
Establishing a tissue diagnosis may not be possible and patients may need empiric treatment as a life-saving measure. First-line treatment in emergent situations is high-dose steroids, although they may confound the diagnosis. Prednisolone 60 mg/m 2 /day (2 mg/kg/day) or methylprednisolone 48 mg/m 2 /day (1.6 mg/kg/day) divided into two daily doses should be employed. This will treat hematologic malignancies and decrease airway edema. The patient should undergo biopsy as soon as the mass shrinks and the patient is stable.
- b.
If poor response to steroids, chemotherapy such as vincristine, cyclophosphamide with or without an anthracycline can be added. Tumor-specific chemotherapy should be instituted after a biopsy has been obtained.
- c.
If a solid tumor not responsive to steroids or chemotherapy, emergent radiation can be performed.
- d.
For symptomatic venous thrombosis with no evidence of hemorrhage, anticoagulation can be initiated using systemic or low-molecular-weight heparin (LMWH):
- i.
Unfractionated heparin can be started with a 75 U/kg bolus followed by 18 U/kg/h (for children) to 28 U/kg/h (infants) continuous infusion. Titrate to a goal-activated partial thromboplastin time of 60–85 s or anti-Xa level of 0.3–0.7 U/ml.
- ii.
LMWH 1 mg/kg every 12 h. Titrate to a goal anti-Xa level of 0.5–1 U/ml.
- i.
- a.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree