Management of High-Risk Breast Disease | 2 |
Arielle Cimeno, Stephanie Richards, Susan B. Kesmodel, Olga Ioffe, Katherine H. R. Tkaczuk, and Emily Bellavance
INTRODUCTION
General Characteristics
High-risk breast lesions represent a category of disease that, while “benign,” confers an increased risk of future malignancy. These lesions may require surgical excision to exclude the presence of an invasive breast cancer or ductal carcinoma in situ (DCIS). Included in this category are atypical ductal hyperplasia (ADH), atypical lobular hyperplasia (ALH), lobular carcinoma in situ (LCIS), flat epithelial atypia (FEA), and intraductal papillomas (IPs). These lesions are typically asymptomatic. The actual incidence of high-risk breast lesions is unknown, primarily because they are often clinically silent. Up to 4% of core needle biopsies (CNBs) and 23% of excisional biopsies are reported to contain a high-risk lesion (1,2). Atypical hyperplasia is found in 10% to 17% of biopsies (3–5). LCIS is an incidental finding in up to 3.8% of benign breast biopsies (3), FEA is a rare lesion found in 1.2% of CNB (6), and 5% of lesions on CNB are identified as IPs (7).
Workup
A. Improvements in breast imaging have resulted in more abnormalities detected on screening mammography, leading to an increased number of breast biopsies.
B. Image-guided CNB is the first modality utilized to obtain tissue for diagnosis.
C. Studies of CNB techniques show that larger gauge needles, higher number of cores obtained, and the use of vacuum-assisted devices result in a lower rate of upgrading of the pathology upon excision, need for rebiopsy, and radiologic–pathologic discordance (8,9,10).
Management
A. A high-risk lesion found on CNB is considered for surgical excision based on the likelihood of malignancy (risk of upgrading) on final pathology. Discordance or concern that the intended lesion was not sampled is also an indication for surgical excision (11).
B. Depending on the results of surgical excision and lifetime risk of breast cancer, patients may be eligible for chemoprevention.
C. Select patients may be candidates for prophylactic mastectomy.
ATYPICAL DUCTAL HYPERPLASIA
Histology
ADH is a proliferative intraductal lesion that fulfills some, but not all, of the criteria for DCIS (Figure 2.1). Like DCIS, it carries an increased risk for the development of invasive breast cancer (12,13). Clinically, ADH is a lesion that does not present as a palpable mass; it is usually identified on screening mammography by the presence of microcalcifications. ADH identified on core biopsy may signify the presence of a higher grade adjacent lesion; cases of ADH on biopsy are not infrequently upgraded to DCIS or invasive carcinoma on excision (1). ADH confers a mildly increased risk of breast cancer—4–5×relative risk (RR).
There are no universally accepted criteria to reliably distinguish between ADH and DCIS (14). Generally, though, a diagnosis of DCIS requires the presence of at least two duct cross-sections that are fully involved by the atypical proliferative lesion (14). Anything less than two fully involved ductal cross-sections is more appropriately characterized as ADH.
ADH may also have significant microscopic overlap with usual ductal hyperplasia (UDH) or benign intraductal hyperplasia. Unlike DCIS, UDH is characterized by cells of nonuniform shape with nuclei oriented parallel to their long axes (streaming cells) and indistinct cell borders. However, atypical features need to be present in order to diagnose a lesion as ADH. ADH is a proliferation of atypical epithelial cells that involve individual ductal spaces. The distinction between ADH and DCIS is the extent of proliferation, with ductal involvement of <2mm and less than two ductal spaces in ADH (3).
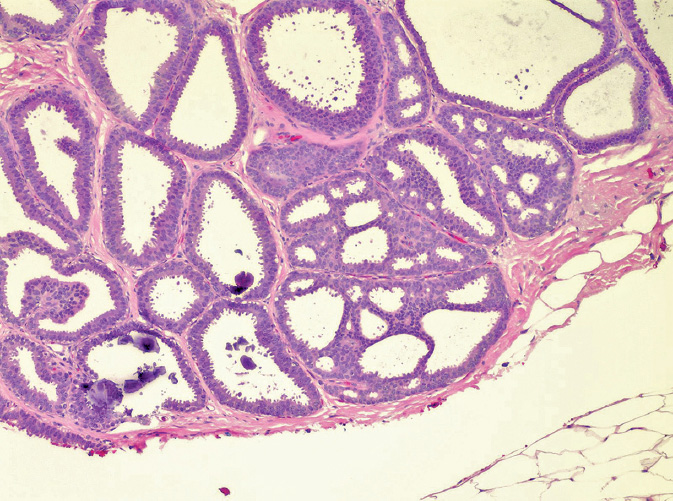
Figure 2.1 Atypical ductal hyperplasia (ADH). The ductal profiles in this field exhibit focal cribriforming and monotonous cellular proliferation; however, this process does not fulfill criteria for ductal carcinoma in situ (does not involve two profiles completely) and is therefore classified as ADH.
Upgrade Rate
A. Surgical excision of ADH found on biopsy has variable published upgrade rates in the range of 18% to 31% (1,8). In a study that assessed upgrade rates in 685 women with ADH, 123 (18%) were found to have more significant pathology in the breast at surgical excision (1). The majority of these cases showed DCIS (82%), with invasive cancer identified in the remaining 18%. In a smaller study that examined pathology in 70 patients with ADH undergoing surgical excision, malignancy was identified in 31% of the patients (8). Again, the majority of patients had DCIS (74%) on final pathology.
Treatment
A. Based on the reported upgrade rates, ADH found on biopsy is considered an indication for surgical excision.
 We currently recommend excision in women with ADH on image-guided biopsy who are surgical candidates.
We currently recommend excision in women with ADH on image-guided biopsy who are surgical candidates.
B. There is interest in elucidating a set of criteria that would determine which patients can forgo surgical excision for imaging surveillance; however, currently there is no consensus and individualized treatment approaches are recommended.
C. Based on cumulative risk factors for breast cancer, patients with a diagnosis of ADH may be eligible for chemoprevention, which is covered later in this chapter.
Risk of Future Malignancy
The cumulative risk of in situ or invasive cancer in patients with ADH is approximately 20% at 20 years (11), an RR of 4 (15). In a study conducted by the Mayo Clinic that examined the risk of subsequent breast cancer in women with atypical hyperplasia, 698 women were identified from pathology review (16). This study included patients with ADH and ALH. With a mean follow-up of 12.5 years, breast cancer was detected in 143 patients (20.4%), with similar rates in patients with ADH and ALH. The majority of cancers developed in the ipsilateral breast and most patients with ADH were diagnosed with invasive breast cancer.
Factors associated with an increased risk of developing breast cancer after a diagnosis of atypical hyperplasia include multifocal disease, less age-related lobular involution in the breast, and younger age at diagnosis.
ATYPICAL LOBULAR HYPERPLASIA
Histology
Like ADH, ALH is an atypical proliferative lesion that is not sufficiently developed to meet the criteria for LCIS. Histologically, ALH—like LCIS—is composed of bland cells that lack E-cadherin expression and fill preexisting lobular structures. However, unlike in LCIS, these cells do not expand the lobules (17). ALH confers a mildly increased bilateral breast cancer risk of the same magnitude as ADH (4–5× RR). ALH and LCIS are distinguished by the degree of lobular involvement, with distortion of <50% of involved lobular acinar spaces categorized as ALH and >50% as LCIS (3).
Upgrade Rate
Published pathology upgrade rates for excision of ALH vary widely, from 0% to 43% (8,10,11), because ALH is commonly found with other lesions on biopsy. In a pooled analysis of studies that examined the results of surgical excision in patients with high-risk breast lesions and included 280 patients with ALH, breast cancer was identified in 53 cases (19%) on surgical excision with 74% of these showing DCIS.
Treatment
A. Because of variable upgrade rates and the high frequency of incidental ALH and concomitant lesions, surgical excision of ALH is controversial. Surgical biopsy should be offered in patients with ALH diagnosed on CNB for definitive diagnosis. Patients should also be offered the option of clinical and imaging follow-up in 6 to 12 months after careful consideration of the risks and benefits of surgical excision.
B. Based on cumulative risk factors for breast cancer, patients with a diagnosis of ALH may be eligible for chemoprevention, which is covered later in this chapter.
Risk of Future Malignancy
A. The RR of developing cancer with ALH is similar to ADH (RR of 4) (15). Cumulative incidence of developing a malignancy in either breast approaches 20% at 20 years, 30% at 25 years, and 35% at 30 years (4,11,18). Breast cancers that develop in women with ALH are more commonly invasive ductal carcinomas than other histologies and, similar to ADH, occur more frequently in the ipsilateral breast (16).
B. The same factors that increase the risk of breast cancer in patients with ADH, including multifocal disease and younger age at diagnosis, apply for ALH (16,18).
LOBULAR CARCINOMA IN SITU
Histology
LCIS is a proliferative intralobular lesion that is often incidentally detected on biopsy of an unrelated lesion (Figure 2.2). It is often multifocal and bilateral, and as such, it is considered to be a marker of increased bilateral risk (8–10 × RR) (19) for invasive breast cancer rather than a true precursor lesion (20,21). LCIS rarely forms a discrete mass; occasionally, a palpable region of firmness will be present due to surrounding tissue reaction (17). LCIS is characterized by lobules that are filled and expanded by discohesive neoplastic bland round cells with eccentric nuclei and occasional signet ring forms. On immunohistochemistry, LCIS cells exhibit loss of E-cadherin, a cellular adhesion protein. Pagetoid spread of LCIS cells into the ducts underneath the normal ductal epithelium is relatively common.
Upgrade Rate
A. The rate of upgrade at surgical excision ranges widely from 0% to 60% in published studies, as many have small sample sizes and include LCIS found with other lesions (8,10,22). In a pooled analysis that reported results of surgical excision in 241 patients with LCIS, upgrade to DCIS or invasive cancer was observed in 32% of cases. The majority of these cases were invasive cancer (64%). Upgrade rates are higher when other high-risk lesions (ADH) are identified in combination with LCIS (22).
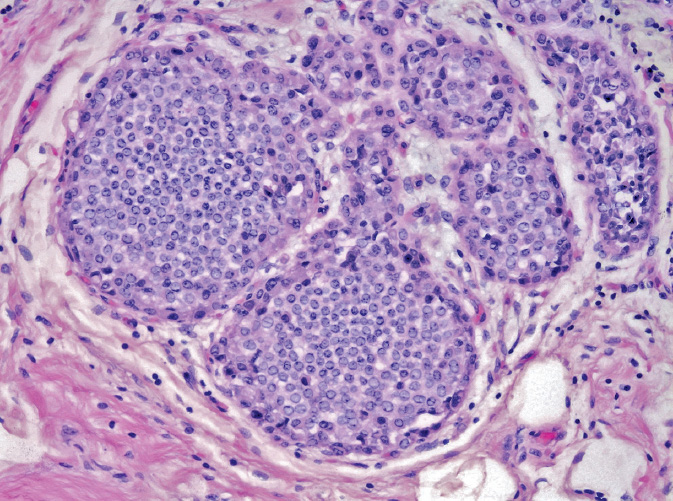
Figure 2.2 Lobular carcinoma in situ (LCIS). Small monotonous discohesive cells fill acini in this lobule.
B. One subtype, pleomorphic LCIS, is a more aggressive lesion with a higher risk of concurrent malignancy.
Treatment
A. Surgical excision for LCIS is controversial and should depend on institutional upgrade rates. Similar to ALH, surgical biopsy should be offered in patients for definitive diagnosis. Patients should also be offered the option of clinical and imaging follow-up in 6 to 12 months after careful consideration of the risks and benefits of surgical excision. Since surgical excision is performed for sampling, negative margins are not necessary.
 Patients should be offered the option of surgical excision as well as the alternative of close follow-up with imaging.
Patients should be offered the option of surgical excision as well as the alternative of close follow-up with imaging.
B. Pleomorphic LCIS is a more aggressive subtype and may represent a precursor lesion to invasive cancer.
 We currently recommend treating this entity with surgical excision. The importance of obtaining microscopically negative margins in the excision of pleomorphic LCIS is controversial. Invasive cancer is identified more frequently on excision of pleomorphic LCIS than standard LCIS (10).
We currently recommend treating this entity with surgical excision. The importance of obtaining microscopically negative margins in the excision of pleomorphic LCIS is controversial. Invasive cancer is identified more frequently on excision of pleomorphic LCIS than standard LCIS (10).
C. Based on cumulative risk factors for breast cancer, patients with a diagnosis of LCIS may be eligible for chemoprevention (see the following).
D. Patients with a diagnosis of LCIS may be considered for more aggressive screening strategies including more frequent clinical breast exams and breast MRI. This should be considered in the context of the patients’ other risk factors for breast cancer.
Risk of Future Malignancy
A. LCIS confers an RR of lifetime malignancy of 10 (19). The risk of developing an invasive cancer is about 0.7% to 1% per year (11).
B. LCIS confers a greater risk for both ductal and lobular cancers, which can occur in either breast (10,11).
ATYPICAL COLUMNAR CELL CHANGE (FLAT EPITHELIAL ATYPIA)
Histology
Columnar cell change is a ubiquitous alteration of benign breast epithelium characterized by tall cuboidal to columnar epithelial cells replacing the normal low cuboidal ductal cells. Features that signify atypical columnar cell change (FEA) are similar to those of ADH: a uniformly sized cell population with basally oriented, hyperchromatic monotonous nuclei. Similar to ADH, though, FEA does not meet the criteria to diagnose DCIS (14). FEA is currently considered to represent a nonobligate precursor to estrogen receptor (ER)/progesterone receptor (PR) positive, low-grade DCIS and invasive carcinoma; as such, it imparts a slightly increased risk of future breast cancer—between that of ADH and florid proliferative breast disease.
Upgrade Rate
A. Published upgrade rates vary from 0% to 25% in small series (2,23).
B. Rates are higher when found with ADH or DCIS. A study that examined upgrade rates in patients with FEA and included 95 patients with pure FEA who underwent surgical excision showed an upgrade rate of only 3.2%, with 1 patient having DCIS and 2 patients having invasive cancer and DCIS. In those patients with concomitant FEA and ADH who underwent surgical excision (43 patients), the upgrade rate was 18.6%, with 4 patients having DCIS alone and 4 patients having an invasive component (2).
C. Upgrade rates with pure FEA may be lower. In a study that assessed upgrade rates to DCIS or invasive cancer in 104 patients with pure FEA who underwent surgical excision, an upgrade rate of 9.6% was observed, with 5 patients having invasive cancer and 5 patients having DCIS. Other histopathology that was identified on surgical excision included LCIS, ADH, ALH, and FEA (6).
Treatment
A. FEA found with other lesions warranting excision should be excised given the higher upgrade rates when found in combination with ADH, ALH, or LCIS (24).
B. The need for excision of pure FEA is unclear. The decision to excise FEA should take into account imaging studies, the findings on pathology, and the overall health of the patient. If there is concern that the finding on imaging has not been adequately sampled or that the imaging and pathology are discordant, then surgical excision is warranted. In general, we consider excision for all patients with FEA given that even in pure FEA, the upgrade rates can be as high as 10%.
Risk of Future Malignancy
A. FEA is a relatively new diagnosis, and long-term studies on the risk of malignancy associated with FEA have not been reported (11).
INTRADUCTAL PAPILLOMAS
Histology
IP is a lesion consisting of a fibrovascular stalk and surrounding epithelial proliferation (7).
Upgrade Rate
A. Upgrade rates for IP vary from 2% to 35% and depend on whether atypia is present in the biopsy (Table 2.1) (1,11).
B. In a study that looked at upgrade rates at surgical excision for 99 papillary lesions without atypia, the upgrade rate was only 2% and both patients had DCIS.
C. Upgrade rates are higher when atypia is present and are closer to rates seen with ADH, ALH, and LCIS. A comparison of upgrade rates in patients with IP without atypia to those with atypia showed an upgrade rate of 6% in patients with IP without atypia and a rate of 21% in patients with IP with atypia (27).
Treatment
IP with or without atypia should be excised if the presenting symptom is a mass or pathologic nipple discharge, there is radiologic–pathologic discordance, or if the CNB was not obtained with a vacuum-assisted device (7). Patients with IP with atypia should always be considered for surgical excision given the similar upgrade rates to other proliferative breast lesions with atypia.
Risk of Future Malignancy
A. The long-term risk of malignancy is linked to the presence of atypia with an RR of 4 with atypia and 2 without atypia (Table 2.2) (11).
CHEMOPREVENTION
Risk Reduction
The use of hormonal (endocrine) therapy has been shown to reduce the incidence of breast cancer in women with increased risk. Only a very small proportion of women who might benefit from chemoprevention are prescribed and/or take these medications, largely due to fear of adverse side effects (4). In a meta-analysis of endocrine prevention of breast cancer with selective estrogen receptor modulators (SERMs), incidence of invasive ER-positive breast cancer was reduced both during treatment and for at least 5 years after completion. Careful consideration of risks and benefits is needed to identify women who are most likely to benefit from prevention (28).
Selective Estrogen Receptor Modulators
A. Agents
1. Tamoxifen—in 1999 the U.S. Food and Drug Administration (FDA) approved tamoxifen for primary prevention of breast cancer.
2. Raloxifene—in 2007 the FDA approved raloxifene for reducing the risk of invasive breast cancer in postmenopausal women with osteoporosis and in postmenopausal women at high risk for invasive breast cancer.
B. Mechanism of action: competitive inhibitor of ERs on breast tissue
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


