Table 19-1
Inflammatory Conditions and Infectious Agents That Are Associated with Specific Types of Cancers
| Condition/Infection | Associated Neoplasm(s) |
| Asbestos | Lung cancer |
| Bronchitis | Lung cancer |
| Gingivitis | Oral cancer |
| Inflammatory bowel disease | Colorectal cancer |
| Skin inflammation (UV) | Skin cancer |
| Hepatitis | Liver cancer |
| AIDS | Non-Hodgkin’s lymphoma |
| Chronic pancreatitis | Pancreatic cancer |
AIDS, Acquired immunodeficiency syndrome; UV, ultraviolet.
Table 19-2
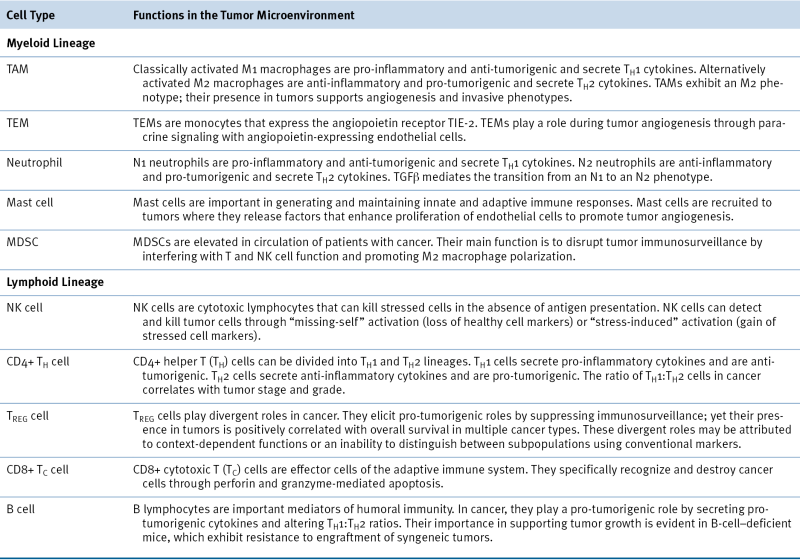
Stromal Cell Populations in the Tumor Microenvironment Are Defined by Various Cell Surface Markers and Have Distinct Functions During Tumorigenesis
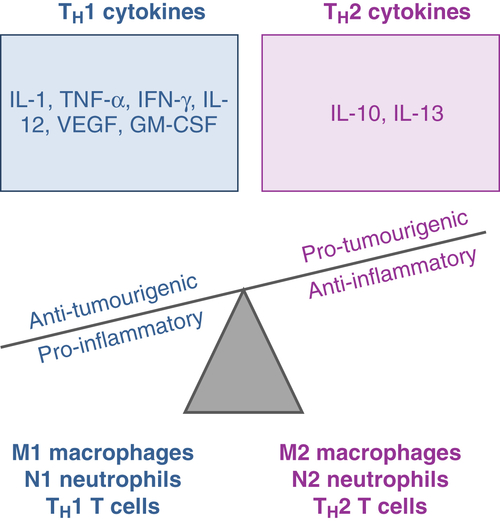
Inflammation and the Metastatic Cascade
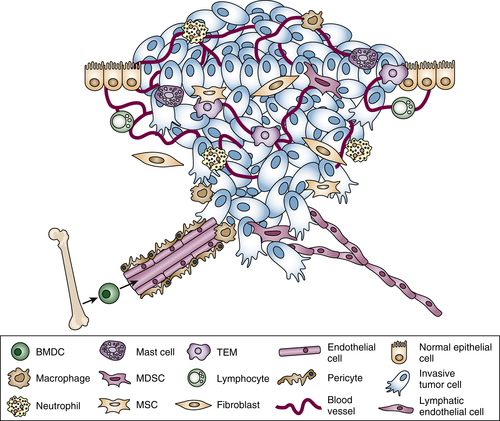
Immune Cells in Cancer
Myeloid Lineage
Macrophages
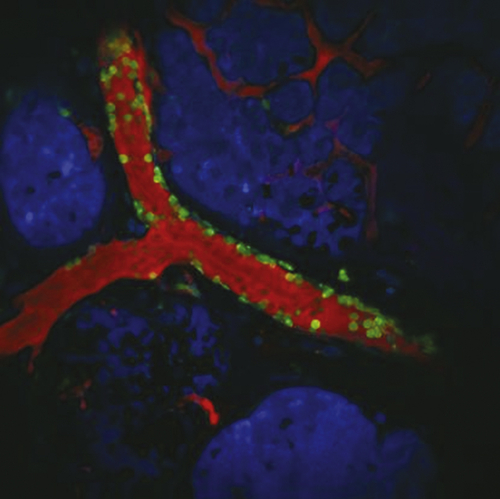
TIE-2–Expressing Monocytes
Neutrophils
Mast Cells
Myeloid-Derived Suppressor Cells
Lymphoid Lineage
NK Cells
CD4+ Helper T Cells
Regulatory T Cells (TREGS)
CD8+ Cytotoxic T Cells
B Cells
Nonhematopoietic Stromal Cell Types in the Tumor Microenvironment
Cancer-Associated Fibroblasts
Mesenchymal Stem Cells
Summary
1. Inflammation and cancer . Nature . 2002 ; 420 : 860 – 867 .
2. Microenvironmental regulation of metastasis . Nat Rev Cancer . 2009 ; 9 : 239 – 252 .
3. Diversification of haematopoietic stem cells to specific lineages . Nat Rev Genet . 2000 ; 1 : 57 – 64 .
4. Inflammation and cancer: back to Virchow? Lancet . 2001 ; 357 : 539 – 545 .
5. Origin and physiological roles of inflammation . Nature . 2008 ; 454 : 428 – 435 .
6. The behavior of chicken sarcoma implanted in the developing embryo . J Exp Med . 1912 ; 15 : 119 – 132 .
7. A transmissible avian neoplasm. (Sarcoma of the common fowl.) . J Exp Med . 1910 ; 12 : 696 – 705 .
8. Inflammation is responsible for the development of wound-induced tumors in chickens infected with Rous sarcoma virus . Cancer Res . 1994 ; 54 : 4334 – 4341 .
9. Wounding and its role in RSV-mediated tumor formation . Science . 1985 ; 230 : 676 – 678 .
10. Hepatitis B virus and hepatocellular carcinoma . J Hepatol . 2003 ; 39 ( suppl 1 ) : S59 – S63 .
11. Global burden of cancers attributable to infections in 2008: a review and synthetic analysis . Lancet Oncol . 2012 ; 13 : 607 – 6215 .
12. The Human Microbiome Project in 2011 and beyond . Cell Host Microbe . 2011 ; 10 : 287 – 291 .
13. The human microbiome: at the interface of health and disease . Nat Rev Genet . 2012 ; 13 : 260 – 270 .
14. A complex microworld in the gut: gut microbiota and cardiovascular disease connectivity . Nat Med . 2012 ; 18 : 1188 – 1189 .
15. Cancer. Bacteria deliver a genotoxic hit . Science . 2012 ; 338 : 52 – 53 .
16. The gut microbiota in IBD . Nat Rev Gastroenterol Hepatol . 2012 ; 9 : 599 – 608 .
17. The Human Microbiome Project: getting to the guts of the matter in cancer epidemiology . Cancer Epidemiol Biomarkers Prev . 2008 ; 17 : 2523 – 2524 .
18. Leukocyte complexity predicts breast cancer survival and functionally regulates response to chemotherapy . Cancer Discov . 2011 ; 1 : 54 – 67 .
19. Inflammation and breast cancer. Balancing immune response: crosstalk between adaptive and innate immune cells during breast cancer progression . Breast Cancer Res . 2007 ; 9 : 212 .
20. Contribution of platelets to tumour metastasis . Nat Rev Cancer . 2011 ; 11 : 123 – 134 .
21. Myeloid-derived suppressor cells: linking inflammation and cancer . J Immunol . 2009 ; 182 : 4499 – 4506 .
22. Impaired recruitment of bone-marrow-derived endothelial and hematopoietic precursor cells blocks tumor angiogenesis and growth . Nat Med . 2001 ; 7 : 1194 – 1201 .
23. Tumor-infiltrating dendritic cell precursors recruited by a beta-defensin contribute to vasculogenesis under the influence of Vegf-A . Nat Med . 2004 ; 10 : 950 – 958 .
24. The role of tumour-associated macrophages in tumour progression: implications for new anticancer therapies . J Pathol . 2002 ; 196 : 254 – 265 .
25. Tumour-educated macrophages promote tumour progression and metastasis . Nat Rev Cancer . 2004 ; 4 : 71 – 78 .
26. Cysteine cathepsins and the cutting edge of cancer invasion . Cell Cycle . 2007 ; 6 : 60 – 64 .
27. Direct visualization of macrophage-assisted tumor cell intravasation in mammary tumors . Cancer Res . 2007 ; 67 : 2649 – 2656 .
28. Setup and use of a two-laser multiphoton microscope for multichannel intravital fluorescence imaging . Nat Protoc . 2011 ; 6 : 1500 – 1520 .
29. Macrophages: obligate partners for tumor cell migration, invasion, and metastasis . Cell . 2006 ; 124 : 263 – 266 .
30. Tumor microenvironment of metastasis in human breast carcinoma: a potential prognostic marker linked to hematogenous dissemination . Clin Cancer Res . 2009 ; 15 : 2433 – 2441 .
31. Exploring the full spectrum of macrophage activation . Nat Rev Immunol . 2008 ; 8 : 958 – 969 .
32. The chemokine system in diverse forms of macrophage activation and polarization . Trends Immunol . 2004 ; 25 : 677 – 686 .
33. IL-4 induces cathepsin protease activity in tumor-associated macrophages to promote cancer growth and invasion . Genes Dev . 2010 ; 24 : 241 – 255 .
34. Macrophages and cathepsin proteases blunt chemotherapeutic response in breast cancer . Genes Dev . 2011 ; 25 : 2465 – 2479 .
35. Inhibition of cysteine cathepsin protease activity enhances chemotherapy regimens by decreasing tumor growth and invasiveness in a mouse model of multistage cancer . Cancer Res . 2007 ; 67 : 7378 – 7385 .
36. Cathepsin cysteine proteases are effectors of invasive growth and angiogenesis during multistage tumorigenesis . Cancer Cell . 2004 ; 5 : 443 – 453 .
37. Alternative activation of tumor-associated macrophages by IL-4: priming for protumoral functions . Cell Cycle . 2010 ; 9 : 4824 – 4835 .
38. Identification of proangiogenic TIE2-expressing monocytes (TEMs) in human peripheral blood and cancer . Blood . 2007 ; 109 : 5276 – 5285 .
39. Tie2 identifies a hematopoietic lineage of proangiogenic monocytes required for tumor vessel formation and a mesenchymal population of pericyte progenitors . Cancer Cell . 2005 ; 8 : 211 – 226 .
40. Targeting the ANG2/TIE2 axis inhibits tumor growth and metastasis by impairing angiogenesis and disabling rebounds of proangiogenic myeloid cells . Cancer Cell . 2011 ; 19 : 512 – 526 .
41. Neutrophil-lymphocyte ratio as a prognostic factor in colorectal cancer . J Surg Oncol . 2005 ; 91 : 181 – 184 .
42. Pre-treatment neutrophil to lymphocyte ratio is elevated in epithelial ovarian cancer and predicts survival after treatment . Cancer Immunol Immunother . 2009 ; 58 : 15 – 23 .
43. Neutrophil/lymphocyte ratio and its association with survival after complete resection in non-small cell lung cancer . J Thorac Cardiovasc Surg . 2009 ; 137 : 425 – 428 .
44. High preoperative neutrophil-lymphocyte ratio predicts poor survival in patients with gastric cancer . Gastric Cancer . 2010 ; 13 : 170 – 176 .
45. Neutrophil/lymphocyte ratio predicts chemotherapy outcomes in patients with advanced colorectal cancer . Br J Cancer . 2011 ; 104 : 1288 – 1295 .
46. Granulocyte-colony stimulating factor promotes lung metastasis through mobilization of Ly6G+Ly6C+ granulocytes . Proc Natl Acad Sci U S A . 2010 ; 107 : 21248 – 21255 .
47. Infiltrating neutrophils mediate the initial angiogenic switch in a mouse model of multistage carcinogenesis . Proc Natl Acad Sci U S A . 2006 ; 103 : 12493 – 12498 .
48. Tumor entrained neutrophils inhibit seeding in the premetastatic lung . Cancer Cell . 2011 ; 20 : 300 – 314 .
49. Neutrophil chemokines secreted by tumor cells mount a lung antimetastatic response during renal cell carcinoma progression . Oncogene . 2013 ; 32 : 1752 – 1760 .
50. Tumor-associated neutrophils: friend or foe? Carcinogenesis . 2012 ; 33 : 949 – 955 .
51. Polarization of tumor-associated neutrophil phenotype by TGF-beta: “N1” versus “N2” TAN . Cancer Cell . 2009 ; 16 : 183 – 194 .
52. The CXCL12-CXCR4 chemotactic pathway as a target of adjuvant breast cancer therapies . Nat Rev Cancer . 2004 ; 4 : 901 – 909 .
53. Analysis of the prognostic effects of inclusion in a clinical trial and of myelosuppression on survival after adjuvant chemotherapy for breast carcinoma . Cancer . 2001 ; 91 : 2246 – 2257 .
54. Role of infiltrated leucocytes in tumour growth and spread . Br J Cancer . 2004 ; 90 : 2053 – 2058 .
55. The role of mast cells in tumour angiogenesis . Br J Haematol . 2001 ; 115 : 514 – 521 .
56. Mast cells are required for angiogenesis and macroscopic expansion of Myc-induced pancreatic islet tumors . Nat Med . 2007 ; 13 : 1211 – 1218 .
57. Inflammatory mast cells up-regulate angiogenesis during squamous epithelial carcinogenesis . Genes Dev . 1999 ; 13 : 1382 – 1397 .
58. Myeloid-derived suppressor cells in inflammatory bowel disease: a new immunoregulatory pathway . Gastroenterology . 2008 ; 135 : 871 – 881 881 e1–e5 .
59. MyD88-dependent expansion of an immature GR-1(+)CD11b(+) population induces T cell suppression and Th2 polarization in sepsis . J Exp Med . 2007 ; 204 : 1463 – 1474 .
60. Myeloid-derived suppressor cells infiltrate the heart in acute Trypanosoma cruzi infection . J Immunol . 2011 ; 187 : 2656 – 2665 .
61. Abrogation of TGF beta signaling in mammary carcinomas recruits Gr-1+CD11b+ myeloid cells that promote metastasis . Cancer Cell . 2008 ; 13 : 23 – 35 .
62. Increased circulating myeloid-derived suppressor cells correlate with clinical cancer stage, metastatic tumor burden, and doxorubicin-cyclophosphamide chemotherapy . Cancer Immunol Immunother . 2009 ; 58 : 49 – 59 .
63. Mechanism of immune dysfunction in cancer mediated by immature Gr-1+ myeloid cells . J Immunol . 2001 ; 166 : 5398 – 5406 .
64. Myeloid suppressor lines inhibit T cell responses by an NO-dependent mechanism . J Immunol . 2002 ; 168 : 689 – 695 .
65. Suppression of T cell proliferation by tumor-induced granulocyte-macrophage progenitor cells producing transforming growth factor-beta and nitric oxide . J Immunol . 1996 ; 156 : 1916 – 1922 .
66. Myeloid-derived suppressor cell inhibition of the IFN response in tumor-bearing mice . Cancer Res . 2011 ; 71 : 5101 – 5110 .
67. Reduction of myeloid-derived suppressor cells and induction of M1 macrophages facilitate the rejection of established metastatic disease . J Immunol . 2005 ; 174 : 636 – 645 .
68. Cross-talk between myeloid-derived suppressor cells and macrophages subverts tumor immunity toward a type 2 response . J Immunol . 2007 ; 179 : 977 – 983 .
69. Expansion of spleen myeloid suppressor cells represses NK cell cytotoxicity in tumor-bearing host . Blood . 2007 ; 109 : 4336 – 4342 .
70. Cancer-expanded myeloid-derived suppressor cells induce anergy of NK cells through membrane-bound TGF-beta 1 . J Immunol . 2009 ; 182 : 240 – 249 .
71. Targeting natural killer cells and natural killer T cells in cancer . Nat Rev Immunol . 2012 ; 12 : 239 – 252 .
72. The prognostic significance of peripheral T-lymphocyte subsets and natural killer cells in patients with colorectal cancer . Hepatogastroenterology . 2009 ; 56 : 1310 – 1315 .
73. Prognostic significance of natural killer cell activity in patients with gastric carcinoma: a multivariate analysis . Am J Gastroenterol . 2001 ; 96 : 574 – 578 .
74. Prognostic significance of tumor infiltrating natural killer cells subset CD57 in patients with squamous cell lung cancer . Lung Cancer . 2002 ; 35 : 23 – 28 .
75. Preventing post-operative metastatic disease by inhibiting surgery-induced dysfunction in natural killer cells . Cancer Res . 2013 ; 73 : 97 – 107 .
76. The immune contexture in human tumours: impact on clinical outcome . Nat Rev Cancer . 2012 ; 12 : 298 – 306 .
77. Interactions between lymphocytes and myeloid cells regulate pro- versus anti-tumor immunity . Cancer Metastasis Rev . 2010 ; 29 : 309 – 316 .
78. Prognostic impact of tumour-infiltrating Th2 and regulatory T cells in classical Hodgkin lymphoma . Hematol Oncol . 2009 ; 27 : 31 – 39 .
79. Phenotypic analysis of tumor-infiltrating lymphocytes from human breast cancer . Anticancer Res . 1992 ; 12 : 1463 – 1466 .
80. Quantification of regulatory T cells enables the identification of high-risk breast cancer patients and those at risk of late relapse . J Clin Oncol . 2006 ; 24 : 5373 – 5380 .
81. Increased regulatory T cells correlate with CD8 T-cell impairment and poor survival in hepatocellular carcinoma patients . Gastroenterology . 2007 ; 132 : 2328 – 2339 .
82. Foxp3+ regulatory T cells: differentiation, specification, subphenotypes . Nat Immunol . 2009 ; 10 : 689 – 695 .
83. Attenuation of CD8(+) T-cell function by CD4(+)CD25(+) regulatory T cells in B-cell non-Hodgkin’s lymphoma . Cancer Res . 2006 ; 66 : 10145 – 10152 .
84. CD4+CD25+Foxp3+ regulatory T cells induce alternative activation of human monocytes/macrophages . Proc Natl Acad Sci U S A . 2007 ; 104 : 19446 – 19451 .
85. Prognostic value of tumor-infiltrating CD4+ T-cell subpopulations in head and neck cancers . Clin Cancer Res . 2006 ; 12 : 465 – 472 .
86. FOXP3 and survival in urinary bladder cancer . BJU Int . 2011 ; 108 : 1672 – 1678 .
87. High frequency of tumor-infiltrating FOXP3(+) regulatory T cells predicts improved survival in mismatch repair-proficient colorectal cancer patients . Int J Cancer . 2010 ; 126 : 2635 – 2643 .
88. Localization and density of immune cells in the invasive margin of human colorectal cancer liver metastases are prognostic for response to chemotherapy . Cancer Res . 2011 ; 71 : 5670 – 5677 .
89. Tumor-infiltrating CD8+ lymphocytes predict clinical outcome in breast cancer . J Clin Oncol . 2011 ; 29 : 1949 – 1955 .
90. Immunotype and immunohistologic characteristics of tumor-infiltrating immune cells are associated with clinical outcome in metastatic melanoma . Cancer Res . 2012 ; 72 : 1070 – 1080 .
91. B cells inhibit induction of T cell-dependent tumor immunity . Nat Med . 1998 ; 4 : 627 – 630 .
92. Tumor-evoked regulatory B cells promote breast cancer metastasis by converting resting CD4(+) T cells to T-regulatory cells . Cancer Res . 2011 ; 71 : 3505 – 3515 .
93. Multiple mechanisms of immune suppression by B lymphocytes . Mol Med . 2012 ; 18 : 123 – 137 .
94. Myofibroblasts and mechano-regulation of connective tissue remodelling . Nat Rev Mol Cell Biol . 2002 ; 3 : 349 – 363 .
95. Fibroblasts in cancer . Nat Rev Cancer . 2006 ; 6 : 392 – 401 .
96. Breast carcinoma-associated fibroblasts rarely contain p53 mutations or chromosomal aberrations . Cancer Res . 2010 ; 70 : 5770 – 5777 .
97. Carcinoma-associated fibroblasts direct tumor progression of initiated human prostatic epithelium . Cancer Res . 1999 ; 59 : 5002 – 5011 .
98. Heterotypic signaling between epithelial tumor cells and fibroblasts in carcinoma formation . Exp Cell Res . 2001 ; 264 : 169 – 184 .
99. Molecular mechanisms and clinical applications of angiogenesis . Nature . 2011 ; 473 : 298 – 307 .
100. Tumor induction of VEGF promoter activity in stromal cells . Cell . 1998 ; 94 : 715 – 725 .
101. Human mesenchymal stem cells modulate allogeneic immune cell responses . Blood . 2005 ; 105 : 1815 – 1822 .
102. Hepatocyte growth factor is a regulator of monocyte-macrophage function . J Immunol . 2001 ; 166 : 1241 – 1247 .
103. The hepatocyte growth factor/Met pathway in development, tumorigenesis, and B-cell differentiation . Adv Cancer Res . 2000 ; 79 : 39 – 90 .
104. Breast cancer cells induce cancer-associated fibroblasts to secrete hepatocyte growth factor to enhance breast tumorigenesis . PLoS One . 2011 ; 6 : e15313 .
105. Mesenchymal stem cells in health and disease . Nat Rev Immunol . 2008 ; 8 : 726 – 736 .
106. Critical role for lysyl oxidase in mesenchymal stem cell-driven breast cancer malignancy . Proc Natl Acad Sci U S A . 2012 ; 109 : 17460 – 17465 .
107. Mesenchymal stem cells induce resistance to chemotherapy through the release of platinum-induced fatty acids . Cancer Cell . 2011 ; 20 : 370 – 383 .
108. Mesenchymal stem cells within tumour stroma promote breast cancer metastasis . Nature . 2007 ; 449 : 557 – 563 .
109. Human bone marrow-derived mesenchymal stem cells suppress human glioma growth through inhibition of angiogenesis . Stem Cells . 2013 ; 31 : 146 – 155 .
110. Mesenchymal stem cells derived from bone marrow favor tumor cell growth in vivo . Exp Mol Pathol . 2006 ; 80 : 267 – 274 .
111. Mesenchymal stem cell-natural killer cell interactions: evidence that activated NK cells are capable of killing MSCs, whereas MSCs can inhibit IL-2-induced NK-cell proliferation . Blood . 2006 ; 107 : 1484 – 1490 .
112. Mesenchymal stem cells inhibit the formation of cytotoxic T lymphocytes, but not activated cytotoxic T lymphocytes or natural killer cells . Transplantation . 2003 ; 76 : 1208 – 1213 .
113. Plasticity in tumor-promoting inflammation: impairment of macrophage recruitment evokes a compensatory neutrophil response . Neoplasia . 2008 ; 10 : 329 – 340 .
114. Deficiency of the macrophage growth factor CSF-1 disrupts pancreatic neuroendocrine tumor development . Oncogene . 2012 ; 31 : 1459 – 1467 .
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree