Summary of Key Points
- •
Despite two decades of progress, the prognosis for patients with lung cancer remains poor. Therefore forming new hypotheses and conducting clinical trials to investigate this patient population are still important.
- •
Accordingly, in the past 25 years, the number of clinical trials involving patients with lung cancer has increased dramatically ( Fig. 60.1 ); however, only a minority of these trials have been conducted on an academic basis. In addition, only a few of these trials have led to a real change in practice. For example, fewer than 10 new drugs were approved for the management of cancer during 2013, with only one drug having a new indication and two drugs having an expanded indication for patients with lung cancer.
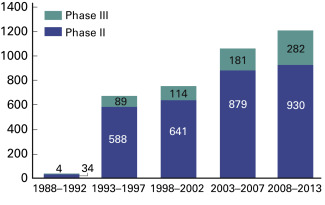
Fig. 60.1
Number of published phase II and phase III clinical trials involving lung cancer from 1988–2013.
- •
While international societies, including the International Association for the Study of Lung Cancer, have encouraged patient participation in clinical trials, the number of patients actually included in such trials worldwide is very low. The actual proportion of patients with cancer who participate in oncology clinical trials is difficult to determine, usually ranging from 2% to 7% in Western countries. However, if we compare the 7.5 million people who die of lung cancer worldwide per year with the reported accrual in clinical trials, the actual proportion of patients with lung cancer who are included in clinical trials worldwide is probably closer to 0.1% to 1%.
- •
Therefore the improvement and promotion of clinical research related to lung cancer are of crucial importance. This chapter provides some proposals that are intended to enable researchers to better organize and promote clinical trials that are designed to advance research and science toward reducing the burden of thoracic malignancies worldwide.
How to Organize Clinical Trials in Lung Cancer
Issues
Definition of Lung Cancer Today
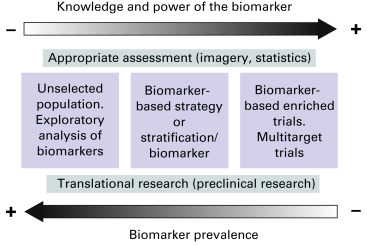
Does lung cancer still exist as an entity? The answer is that it probably does not, considering the progress that had been made in developing a better clinical, pathologic, and biologic definition of this disease. Consequently, including all of these patients in a single trial is irrelevant. However, most clinical studies focus on obtaining a reliable estimate of the average treatment effect in a broad population of patients. In practice, clinical trials in oncology involve a delicate balance between the need for reliable evidence for a large population and the need to integrate biomarkers and thus to focus on the population carrying these biomarkers against which the targeted therapies are supposed to be efficient. For example, many drug developments have been stopped because of the absence of focus on predefined biomarkers. The first clinical trials analyzing the effect of epidermal growth factor receptor (EGFR)-tyrosine kinase inhibitors such as gefitinib independently of EGFR status or cetuximab with an inappropriate biomarker threshold led to disappointing results, despite the high number of patients enrolled. Conversely, the selection of patients on the basis of the accurate biomarker (EGFR-activating mutation, anaplastic lymphoma kinase [ALK] translocation) recently led to impressive results with fewer patients. Therefore lung cancer is now considered to comprise a mosaic of rare diseases, and clinical trials should be designed adequately.
Expectations for Quick Progress and Impact on Financial Issues
Patients are obviously waiting for rapid improvement in the treatment of the disease, and the delay between the discovery of a new biologic target, preclinical proof of principle, and the approval of a new drug (or a new strategy) is generally viewed as unacceptably long. From this perspective, at least, the expectations of drug companies are consistent with those of patients. Is this situation changing? Perhaps, indeed, the delays between the results of the first phase I study and the European Medicines Agency (EMA) and/or the US Food and Drug Administration (US FDA) approval of new biomarker-guided drugs appear shorter when compared with those for previous standard chemotherapy ( Fig. 60.2 ). In addition, the results presented during the 2013 American Society of Clinical Oncology (ASCO) meeting by Canadian colleagues suggest that biomarker-guided treatments clearly have the greatest chance of demonstrating an activity after starting clinical development than do standard, nonbiomarker-guided, treatments. Indeed, after the assessment of more than 2400 trials, the authors found that the likelihood of a new drug passing all phases of clinical testing and being approved (i.e., the cumulative clinical trial success rate) was 11%, which was less than the expected industry aggregate rate (16.5%). The success of phase III trials was found to be the biggest obstacle for drug approval, with a success rate of only 28%. Biomarker-guided targeted therapies (with a success rate of 62%) and receptor-targeted therapies (with a success rate of 31%) were found to have the highest likelihood of success in clinical trials. Accordingly, Subramanian et al. conducted a survey to review ongoing clinical trials involving patients with nonsmall cell lung cancer (NSCLC) as listed in the ClinicalTrials.gov registry in 2012 and found that the number of clinical trials with biomarker-based treatment selection for lung cancer had significantly increased since 2009 (from 7.9% to 25.8%; p < 0.001).
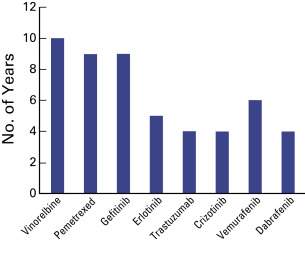
The costs of a clinical trial depend on many factors. An audit of industry-sponsored clinical trials in oncology recently indicated that the average cost is $165 million for basic research and discovery, $87 million for preclinical development, $130 million for a phase I trial, $190 million for a phase II trial, and $268 million for a phase III trial. It seems that the use of a validated biomarker, such as human EGFR-2 for breast cancer, reduces clinical trial risk by as much as 50%, resulting in cost savings of 27% in advanced and metastatic breast cancer. This percentage is comparable with the results of a large study in which the use of biomarkers decreased the cost of drug development by 26%. It is difficult to have a worldwide picture of the funding of clinical trials in oncology. However, one recent example in the United States showed how academic-sponsored clinical trials in oncology are fragile. At the time of the federal budget cuts in 2013, a survey showed that a large proportion of US oncologists consequently reduced the end points of trials (36.9%), closed or participated in fewer cooperative group trials (28.3%), postponed the launch of clinical trials (26.7%), and limited enrollment in clinical trials (23.1%). Therefore adapting the design of oncology clinical trials and durably ensuring their funding are important.
Selection of Hypotheses for Clinical Trials and Impact of Biomarkers on Design
Instead of testing therapeutic strategies or new chemotherapeutic drugs, clinical researchers now face a huge amount of basic-science data and preclinical concepts. The challenge is to build the most appropriate trials to validate some of these hypotheses. A dispensable time in the building of a clinical trial is to deeply analyze the preclinical data and to anticipate all issues that may impair the development of the drugs (efficacy, toxicity, pharmacokinetics, etc.).
Advances in biotechnology and genomics gradually have uncovered the biology of lung cancer. Deeper understanding of the biology of the disease can facilitate the development of new treatments, whereas deeper understanding of the heterogeneity of the disease can facilitate the development of effective biomarkers or diagnostic tests that can be used to select appropriate treatments for individual patients. In particular, the recent establishment of high-throughput molecular assay technologies, such as high-throughput sequencing, single-nucleotide polymorphism arrays, gene-expression microarrays, and protein arrays, has allowed for the discovery of potential new biomarkers and the development of composite genomic signatures for personalized medicine.
Consequently, different trial designs have been proposed on the basis of the incidence of a given biomarker. For example, early phase trials initially tested the efficacy of a drug in a population harboring a rare molecular abnormality such as v-raf murine sarcoma viral oncogene homolog B (BRAF) mutation or ALK translocation. Then, randomized trials were conducted in populations with a more frequent or better characterized molecular alteration. Nevertheless, those studies can be criticized, as the prognostic value of the biomarker by itself (independent of its predictive value) is often underestimated. Therefore we again need new designs of clinical trials in oncology.
National, Multinational, and International Cooperation or Competition
Thousands of studies are carried out every year around the world, involving thousands of patients and billions of dollars of investment. In addition, conducting clinical trials, especially when the patient population is small, as is the case for the current development of the vast majority of biomarker-guided inhibitors, is necessarily competitive. First, at the time of targeted clinical trials in oncology, only centers with high-level facilities can still test and enroll patients, and, when two or more studies are in competition, substantial time and resources that otherwise could have been applied to other valuable projects inevitably will be wasted. Second, the winner is not necessarily the best treatment, as the trials are conducted sequentially. Third, there is also a competition between industrial and academic trials in that they often involve the same patients but have different trial designs. Fourth, there is competition between countries as each country has its own way of funding trials. Thus it is sometimes harder to conduct international trials because of the complexity of administrative procedures. Therefore collaborations, networks, and simplified rules are needed.
Possible Solutions
New Designs for Modern Clinical Trials
Freidlin et al. recently proposed three designs for biomarker-based clinical trials ( Fig. 60.3 ). If there are two or more existing treatment options with no definitive evidence for one being preferred, the most efficient trial design for evaluating biomarker utility is the biomarker-stratified design. Biomarker status is assigned, and two groups are established: one group is biomarker positive, and the other is biomarker negative. In each group, randomization is performed for treatment assignment. The analysis plan will be centered on determining treatment effect dependence according to the biomarker status. The biomarker-stratified design maximizes the advantage of randomization by providing unbiased estimates of benefit-to-risk ratios across different biomarker-defined subgroups and for the entire randomly assigned population. For predictive biomarkers, the biomarker-stratified design can assess whether the marker is useful for selecting the best among two or more treatments for a given patient.
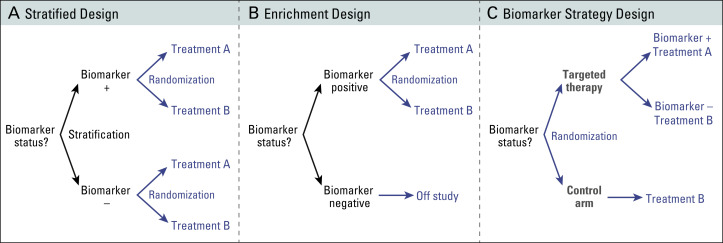
In some situations, sufficiently convincing evidence based on preclinical or clinical data is available and suggests that the potential treatment benefit is limited to a certain biomarker-defined patient subgroup. In these situations, the clinical utility of the biomarker can be partially assessed in a trial with an enrichment design: the biomarker is evaluated in all patients, but random assignment is restricted to patients who have biomarker-positive tumors.
In the third type of trial, with a biomarker-strategy design, patients are randomly assigned to an experimental treatment arm in which the biomarker is used to determine therapy or to a control arm in which a biomarker is not used.
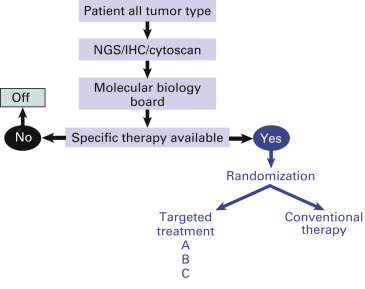
Because of the multiplicity of biomarkers and limited resources, some recent trials have been designed to analyze several targets and subsequently to test several dedicated drugs ( Fig. 60.4 ). The main objectives of this type of trial are to determine the feasibility of such a wide molecular screening, the superiority of tailored treatment, and, in some cases, the effect of new drugs on selected targets. The first example of such a design is the Biomarker-integrated Approaches of Targeted Therapy for Lung Cancer Elimination (BATTLE) trial. Following an initial equal randomization period, patients with NSCLC that was refractory to chemotherapy were adaptively randomly assigned to treatment with erlotinib, vandetanib, erlotinib plus bexarotene, or sorafenib on the basis of relevant molecular biomarkers. Following this trial, BATTLE-2 randomly assigned patients to erlotinib, erlotinib plus MK-2206, MK-2206 plus AZD6244, or sorafenib, stratified by Kirsten rat sarcoma (KRAS) status. Other trials of the same type are currently being conducted in France and other countries. In the Molecular Screening for Cancer Treatment Optimization trial (initiated at Institut Gustave Roussy in France), the molecular profile of patients with refractory cancer is determined using comparative genomic hybridization array and a panel of hot-spot mutations in 96 amplicons from a biopsy sample taken from a metastatic site. Patients are enrolled into specific phase I trials according to the presence of a molecular abnormality. Each patient who is enrolled in the trial with a matched molecular-targeted agent is used as his or her own control for the evaluation of the efficacy of this approach. As another example, the SHIVA trial was a randomized proof-of-concept phase II trial in which therapy based on tumor molecular profiling was compared with conventional therapy in patients with refractory cancer. One hundred and ninety-five patients were randomly assigned to each arm, with a crossover at disease progression whenever possible. In the experimental arm, patients were treated with an approved molecularly targeted agent based on an actionable molecular abnormality. However, an actionable molecular abnormality might not be identified in every patient. In contrast to traditional randomized trials in oncology that are performed in a homogeneous population of patients with a specific type of tumor and in a specific setting, the goal of this trial was to look for heterogeneity in tumor types to establish the proof of concept of whether targeted agents should be developed according to their tumor molecular profile rather than according to tumor type.
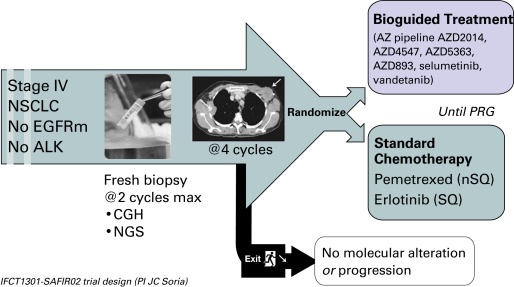
Similarly, the design just described now has been adapted to lung cancer with the SAFIR02 lung trial ( Fig. 60.5 ). This open-label phase II randomized trial involves the use of high-throughput genome analysis as a therapeutic decision tool for patients with stage IV NSCLC that has not progressed after induction chemotherapy. The trial will compare a maintenance treatment administered according to the identified molecular anomaly of the lung tumor with a maintenance treatment administered without consideration of the tumor genome analysis; pemetrexed will be given to patients with nonsquamous cell carcinoma, and erlotinib will be given to patients with squamous cell carcinoma.
The Lung Master Protocol (Lung-MAP) trial is another study that was designed to test several targeted therapies for patients with advanced squamous cell lung cancer. The trial is a biomarker-driven phase II/III with multiple substudies that open and close independently. A common platform of next-generation DNA sequencing is used to identify a patient’s actionable molecular abnormalities, and the patient is assigned to the investigational drug that targets the mutation or to a “nonmatch” substudy that consists of an immunotherapy or combination therapy.
Lastly, another way to test several hypotheses in a clinical trial is to test a targeted drug in patients with several cancer types harboring the target. For example, the VE-BASKET trial tests the efficacy of vemurafenib (which is known to be efficient for the treatment of melanoma that harbors the BRAF V600E mutation) for the treatment of other solid tumors. This trial enrolls patients with histologically confirmed cancers or myelomas (excluding melanoma and papillary thyroid cancer) that harbor a BRAF V600 mutation and are refractory to standard therapy or for which standard or curative therapy does not exist.
New Designs: Mixing Avatar Models and Clinical Trials
Targeted therapies have demonstrated efficacy against specific subsets of molecularly defined cancers. Although most patients with lung cancer are stratified according to a single oncogenic driver, cancers harboring identical activating genetic mutations show large variations in their responses to the same targeted therapy. The biology underlying this heterogeneity is not well understood, and the impact of coexisting genetic mutations, especially the loss of tumor suppressors, has not been fully explored. Chen et al. used genetically engineered mouse models to conduct a so-called co-clinical trial that mirrors an ongoing human clinical trial in patients with KRAS-mutant lung cancers. The aim of this trial is to determine if the mitogen-activated protein kinase kinase (MEK) inhibitor selumetinib (AZD6244) increases the efficacy of docetaxel, a standard-of-care chemotherapy. The authors demonstrated that concomitant loss of either p53 (also known as TP53) or Lkb1 (also known as Stk11), two clinically relevant tumor suppressors, markedly impaired the response of KRAS-mutant cancers to docetaxel monotherapy. They observed that the addition of selumetinib provided substantial benefit for mice with lung cancer caused by KRAS and p53 mutations, but mice with KRAS and Lkb1 mutations had primary resistance to this combination therapy. These co-clinical results identified predictive genetic biomarkers that should be validated by interrogating samples from patients enrolled in the concurrent clinical trial. These studies also highlight the rationale for synchronous co-clinical trials, which is not only to anticipate the results of ongoing human clinical trials but also to generate clinically relevant hypotheses that can inform the analysis and design of human studies.
Cooperation and Networks
The issues described can be partially solved by organizing clinical research in a national and/or transnational way. Our ability to strengthen international collaborations will result in maximization of resources and the access of patients to clinical trials in oncology.
On the one hand, drug companies and clinical research organizations, which are mainly international companies, usually know how to use the various resources that are nationally or internationally available. Pending budget availability, these entities built the so-called ideal clinical and translational network needed to conduct a defined trial. The influence of clinicians, cooperative groups, and even national health authorities (except for the EMEA and FDA) on the study design is often limited.
On the other hand, academic clinical trials in oncology initially were mainly organized by local or regional centers. This organization responds first to the wishes of patients who want to be treated closer to home and second to the opportunity to easily have access to a clinical trial, as the distance from a physician’s practice to the nearest clinical trial site is inversely associated with referral and recruitment. Cooperation between centers is essential for the successful conception, funding, and performance of clinical trials in oncology. Consequently, national groups have been created to promote clinical and translational research. For example, in the United States, the National Cancer Institute (NCI) Clinical Trials Cooperative Group Program has served as the leading network for federally funded clinical research on cancer. In Asia and Europe, many countries have organized themselves according to the same model, with one or more cooperative groups involved in clinical trials focusing on lung cancer. In Europe, the European Organisation for Research and Treatment of Cancer Lung Cancer Study Group and the European Thoracic Oncology Platform are performing multinational clinical trials.
Regulatory, logistic, and financial hurdles, however, often hamper the conduct of joined trials. Working together should help researchers to overcome these barriers, with the International Association for the Study of Lung Cancer (IASLC) as a central link.
Worldwide Standardization of Oncology Clinical Trials Allowing Cross-Trial Comparisons
Besides rules regarding publication, the issue of cross-trial comparison raises the problem of worldwide standardization of oncology clinical trials, and, more widely, clinical research in oncology. The best example in this field is the increasing number of biomarker-based prognostic and predictive studies aiming to define which subset of patients may actually benefit best from a specific drug or therapeutic strategy ( Fig. 60.6 ). One of the major issues encountered in these studies is the reproducibility of published results, that is, the stability of the biomarkers across different series of patients, across different geographic areas, and over time. Once the question of variability of molecular assays is fixed across different laboratories, an issue that is not trivial, limiting the confusion bias is a major step of the analysis. Multivariate analyses aim to control for stratification factors of the trial and for the confounding clinical or pathologic characteristics influencing survival in the univariate analysis at a given p value (<0.1 or <0.2, depending on the study). However, the strategy of such multivariate analyses is rarely comparable within multiple trials or series, often making such comparisons hazardous.