Hematopoietic Stem Cell Transplantation
Mary C. Territo
I. PRINCIPLES
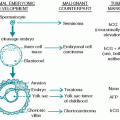
A. Hematopoietic stem cell transplantation (HSCT) is an important treatment option for an increasing number of malignant and nonmalignant disorders that are listed in Table 37.1. HSCT has been used in malignant diseases for the fol lowing situations:
1. To restore marrow function for the patient following the administration of very high doses (myeloablative/immunoablative) of chemotherapy with or without radiotherapy (CT/RT) to kill off tumor cells. The following requirements apply when using this approach to treat tumors:
a. The tumor must have a steep dose—response curve so that escalating doses of drug results in increased tumor killing.
b. The drugs that give that steep dose—response curve must have the bone marrow as their main dose-limiting toxicity (since HSCT will not protect any of the other organ toxicity).
c. The types of tumors that are usually treated in this manner include primarily the hematologic malignancies (leukemias, lymphomas, myelomas) but also germ cell tumors, neuroblastoma, and selected other solid tumors.
2. To replace deficient or defective hematopoietic cells for diseases such as aplastic anemia and congenital hematologic, immunologic, and metabolic disorders
3. To effectively administer adoptive immunotherapy against tumor cells (the graft-vs.-tumor effect [GVT])
B. The choice of the type of transplant that is performed and the type of conditioning therapy used will depend on the disease being treated, the clinical status of the patient, and the donor cells that are available.
C. Outcomes of transplantation will depend on multiple factors including age of the patient, stage of disease, disease risk factors, prior therapies, comorbid conditions, and the type of conditioning therapy used. For allogeneic transplants, donor relationship, HLA matching, and donor cell dose (for cord blood) are also important variables impacting on outcome.
II. STEM CELL SOURCES.
Hematopoietic stem cells (HSCs) normally reside in the bone marrow, with only rare HSCs circulating in the blood. HSCs can be found in increased numbers in the blood during recovery from chemotherapy-induced cytopenias and can also be mobilized from the bone marrow into the blood with agents such as granulocyte colony-stimulating factor (GCSF) or plerixafor, which cause release of the stem cells from their niche in the bone marrow. Umbilical cord blood is a very rich source of HSCs that can also be used for transplantation.
A. Bone marrow (BM) is collected in the operating room under general or spinal anesthesia. Multiple aspirations (of 5 to 10 mL each) are obtained to a desired target dose of about 3 × 108 nucleated cells per kilogram of recipient weight (about 1 to 1.5 L for an adult). The collection is then filtered to remove any bone
particles, can be processed to deplete red blood cells (RBCs) or plasma if needed for ABO-incompatible transplants, and can then be cryopreserved for later use or directly infused intravenously into the patient. This was the original product used for HSCT.
particles, can be processed to deplete red blood cells (RBCs) or plasma if needed for ABO-incompatible transplants, and can then be cryopreserved for later use or directly infused intravenously into the patient. This was the original product used for HSCT.
Table 37.1 Uses for Hematopoietic Stem Cell Transplantation | ||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
B. Peripheral blood stem cells (PBSCs). Donors are first given GCSF to mobilize the HSC into the blood (for autologous donors, chemotherapy is frequently given prior to the GCSF). PBSCs are then obtained in the nucleated cell fraction of the blood by apheresis. Multiple collections may be required to reach the target dose (1 to 5 × 106 CD34-positive cells per kilogram of patient weight). The cells can then be cryopreserved for later use or directly infused intravenously into the patient. This product engrafts a little faster than BM, has a similar incidence of acute graft versus host disease (GVHD) as BM, but has a greater occurrence of chronic GVHD.
C. Umbilical cord blood (UCB) cells are obtained from the umbilical vein of the placenta after the umbilical cord has been severed from the newborn. This blood is rich in HSC, and the lymphocytes are naive. This product results in less GVHD than either BM or PBSC but is slower to engraft.
D. Manipulation. Stem cell sources can be manipulated in a variety of ways depending on the intent of the transplant. Some of the manipulations used include depletion of T cells or lymphocyte subsets, enrichment of CD34-positive cells, and stem cell expansion. Hematopoietic stem cells are also being engineered to augment tumor killing, infection control, or replace genetic deficiencies.
III. TYPES OF HSCT
A. Autologous transplant. The patient’s own cells are used as the HSC source. This approach is primarily used to permit the administration of very high doses (myeloablative) of CT/RT to kill tumor cells. The advantage of this approach is that you do not have to search for an allogeneic donor and there is no GVHD. The disadvantage is that you may have residual tumor cells in the graft and you achieve no graft-versus-tumor effect from the graft.
B. Allogeneic transplant. The HSCs are obtained from someone other than the patient. The donor of the HSCs must be matched by HLA tissue typing with the patient. Genes for the HLA antigen system are found on chromosome 6. HLA typing is performed for the class I antigens (A, B, and C) and the class II DR antigens to identify properly matched donors. The advantage of this approach is that the product has normal stem cells, which are free of tumor or abnormal cells. In addition to allowing recovery after myeloablative CT/RT, allogeneic HSCs can be used to replace deficient or defective stem cells
(as used for aplastic anemia or genetic deficiencies). Allogeneic transplantation also provides adoptive immunotherapy against tumor cells of the recipient (GVT effect), and a reduction in the intensity of the conditioning therapy is sometimes used with GVT as the major goal of the transplant. The disadvantages of allogeneic transplantation are that you need to find an appropriately matched donor and that the patient is at risk for GVHD.
(as used for aplastic anemia or genetic deficiencies). Allogeneic transplantation also provides adoptive immunotherapy against tumor cells of the recipient (GVT effect), and a reduction in the intensity of the conditioning therapy is sometimes used with GVT as the major goal of the transplant. The disadvantages of allogeneic transplantation are that you need to find an appropriately matched donor and that the patient is at risk for GVHD.
1. Related donors. The best donor is usually a sibling with the same two HLA haplotypes as the recipient (matched on both chromosomes for HLA A, B, C, and DR; an “8 of 8 HLA match”). Only about 30% of patients will have an identifiable sibling donor. Identical twin (syngeneic) donors are the best donors immunologically but have a higher risk of relapse (less graft-vs.-tumor effect) after transplant. Rarely, HLA-identical family members other than siblings can be identified. Partially matched or haploidentical related donors (only matched for one of the HLA chromosomes [4 of 8 HLA match]) have been used at some centers. These transplants require additional immunodepletion of the graft and have delayed immune reconstitution.
2. Unrelated donors. There are large registries of individuals around the world who have volunteered to donate HSCs for unrelated patients in need of a transplant. With sensitive DNA-based typing, HLA-matched unrelated donors can be found for many patients. The chances of finding an appropri ate unrelated donor for an individual patient depends on the specific HLA typing of the patient and varies with different ethnic groups. Transplants using unrelated donor HSCs have some increased risk of GVHD but are an appropriate treatment option if no matched related donors are available. The use of unrelated UCB requires less stringent HLA compatibility but may be limited by cell dose.
IV. CONDITIONING THERAPY.
Patients are given high doses of CT/RT prior to the transplant. For autologous transplants, the therapy is aimed at getting the greatest tumor killing while ignoring the myeloablative toxicity of the agents but is limited by toxicity to other organs. Immune ablation (to allow for engraftment of the foreign HSCs) is necessary for allogeneic transplants, in addition to tumor killing. Many different “conditioning protocols” have been used with similar outcomes depending on the clinical situation of the patient. Examples of some standard conditioning regimens follow.
A. Total-body irradiation and cyclophosphamide (Cy)
1. Total-body irradiation. Patients receive 12 Gy given in 8 fractions (1.5 Gy each) on days minus 7 to minus 4.
2. Cy, 60 mg/kg/d in normal saline solution, is given IV over 1 hour for 2 days (on days minus 3 and minus 2). Patients also receive mesna, 60 mg/kg/d by constant IV infusion beginning with the start of Cy and continuing until 24 hours following completion of Cy.
3. Rest on day minus 1.
4. HSCT on day 0
B. High-dose busulfan and Cy
1. Busulfan (Busulfex) at a dose of 0.8 mg/kg of ideal body weight or actual body weight (whichever is lower) is administered IV over 2 hours every 6 hours for 4 days for a total of 16 doses on days minus 7 to minus 4. Phenytoin (300 mg/d and adjusted for therapeutic blood levels) should be given prophylactically to prevent seizures beginning on the day prior to starting the busulfan and continued until 2 days after busulfan is completed.
2. Cy (to start after busulfan finishes) at a dose of 60 mg/kg/d is given in normal saline solution IV over 1 hour daily for 2 days (on days minus 3 and minus 2).
Patients receive mesna, 60 mg/kg/d by constant IV infusion, beginning with the start of Cy and continuing until 24 hours following completion of Cy.
Patients receive mesna, 60 mg/kg/d by constant IV infusion, beginning with the start of Cy and continuing until 24 hours following completion of Cy.
3. Rest on Day minus 1
4. HSCT on Day 0
C. High-dose BEAM (BCNU/etoposide/cytarabine/melphalan)
1. BCNU (carmustine), 300 mg/m2 in 500 mL normal saline, is given IV over 2 hours on day minus 7.
2. Etoposide, 100 mg/m2, is given IV over 2 hours every 12 hours for 8 doses on days minus 6, minus 5, minus 4, and minus 3.
3. Cytarabine, 200 mg/m2, is given IV over 1 hour every 12 hours for 8 doses on days minus 6, minus 5, minus 4, and minus 3.
4. Melphalan, 140 mg/m2, is given IV over 1 hour on Day minus 2.
5. Rest on day minus 1
6. HSCT on day 0
D. On day 0, donor BM, PBSC, or UCB is administered IV without a filter. Patients should be premedicated with acetaminophen (650 mg PO), diphenhydramine (Benadryl, 50 mg PO or IV), and hydrocortisone (50 mg IV) 30 minutes prior to HSC infusion. Benadryl (50 mg), epinephrine (1:10,000; 10 mL), and hydrocortisone (100 mg) should be at bedside for standby IV use. Oxygen with a nasal cannula setup should also be on standby in the room during the stem cell infusion.
V. SUPPORTIVE CARE
A. All blood products (except the HSC) should be irradiated with 1.5 Gy (to prevent transfusional GVHD) as soon as the conditioning regimen is initiated.
1. RBC transfusions. Packed RBCs should be given to maintain the hematocrit at 27% (or higher if clinically indicated).
2. Platelet transfusions.




Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree