Objective
Modulation of insulin delivery
Reduction of severity and/or duration of hypoglycaemia
Suspension of insulin delivery at low glucose threshold
Hypoglycaemia prevention
Suspension/reduction of insulin delivery when hypoglycaemia is predicted
Control to range
Modulation of insulin delivery outside target range to reduce hypoglycaemic and hyperglycaemic excursions
Overnight glucose control
Modulation of insulin delivery for nocturnal glucose control
Closed-loop system with meal/exercise announcement
Modulation of insulin delivery when the control algorithm is aware of exercises and meals as announced by user; prandial insulin boluses are administered by user
Fully closed-loop system
Modulation of insulin delivery when the control algorithm is unaware of meals, exercise, stress and other lifestyle disturbances that affect glucose control; glucagon or other hormones may be co-administered to reduce the risk of hypoglycaemia
The concept of closed-loop insulin delivery was proposed by Kadish over five decades ago [47]. The Biostator, the first commercially available device, was introduced in the late 1970s utilising intravenous glucose sampling and intravenous insulin infusion. With advances in subcutaneous insulin pump therapy and sensor technology, the focus moved to combining body-worn and not implantable devices to deliver insulin and measure glucose in the subcutaneous tissue. Current closed-loop systems consist of three components: a real-time CGM, an insulin pump and a control algorithm (Fig. 14.1).
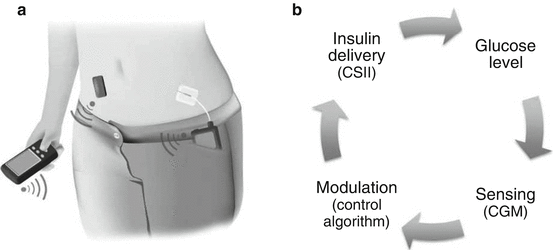

Fig. 14.1
Closed-loop insulin delivery. (a) A sensor (black rectangle) transmits information about interstitial glucose levels to a hand-held controller (black box) which runs a control algorithm and interacts with the user. An insulin pump (device in the pocket) delivers a rapid-acting insulin analogue subcutaneously. Insulin delivery is modulated by the control algorithm. The communication between the system components is wireless. The control algorithm can also reside within the insulin pump (Adapted from Hovorka [43]). (b) The closed loop mimics the physiological feedback normally provided by the beta cell
Two main categories of control algorithm have been employed, the proportional-integral-derivative (PID) controller [48, 49], a classic feedback control mechanism, and the model predictive controller (MPC) [50]. Other clinically evaluated approaches include controllers based on fuzzy logic [51] or a combination of MPC and PID for insulin and glucagon co-delivery [52]. Apart from the insulin-only systems, bi-hormonal systems have evolved which deliver both insulin and glucagon [44].
Initial single- or multiday/night studies in children and adolescents under controlled laboratory settings suggested improved glycaemic control during overnight closed loop [50, 53–55] and day-and-night closed loop [56, 57] and an unchanged [56, 57] or reduced [50, 53, 54] risk of hypoglycaemia when compared to sensor-augmented pump therapy.
Investigations in transitional settings, incorporating remote monitoring and supervision by research staff on-site in hotels [58] or at diabetes camps [59–61] demonstrated promising results in children and adolescents with regard to improved glucose control and hypoglycaemia reduction using insulin-only [59, 61] and bi-hormonal systems [60].
Recently, first short- to midterm at-home studies have been performed. In adolescents, three- [62] to six-week [63] home use of overnight closed-loop systems with remote monitoring [63] or unsupervised [62] was shown to be feasible, safe and effective. The time glucose was in the target range was significantly increased compared to sensor-augmented pump therapy [62, 63], including fewer nights with glucose levels in the hypoglycaemic range [62] and reduced time spent in hypoglycaemia [63]. In a 7-day free-living evaluation of a 24/7 closed-loop system with manual prandial insulin in suboptimally controlled children and adolescents, closed loop significantly reduced mean glucose and increased time in target without increasing the risk of hypoglycaemia [64].
In a quantitative and qualitative psychosocial analysis of experiences of closed-loop trial participants, adolescents and their parents reported positive themes such as reassurance/peace of mind, ‘time off’ from diabetes demands, improved overnight control leading to improved daily functioning and diabetes control and improved sleep. The key negative themes are related mainly to technical difficulties, intrusiveness of alarms and size of the devices. Overall, for adolescents, benefits of a closed-loop system seemed to outweigh practical challenges [65].
14.6 Outlook
Closed-loop insulin delivery systems are progressing fast from bench to clinical practice. Over the past 5 years, closed-loop approaches such as threshold suspend and predictive low glucose suspend have been integrated into commercial insulin pumps.
Results of studies investigating more advanced closed-loop systems in laboratory settings and short- to midterm outpatient testing have generated excitement stimulating further innovation to improve component-level and system-level performance and system usability on the way towards safe and efficacious hands-off fully closed-loop systems.
The absorption rate of currently available rapid-acting insulin is still not fast enough to effectively control postprandial glucose excursions, apparent in post-meal performance of fully closed-loop system without meal announcement [49]. Postprandial hyperglycaemia is considered a major problem in the paediatric population [66], exacerbated by a longer duration of insulin action in adolescents [67]. Ancillary technologies such as site warming [68] and co-administration of hyaluronidase [69] accelerate absorption and action of rapid-acting insulin analogues. Premeal pramlintide or glucagon-like peptide-1 analogue may limit post-meal glucose excursions [70, 71].
Bi-hormonal closed loop may provide additional benefit and lead to a reduced risk of hypoglycaemia [52, 60]. With respect to overnight glucose control, insulin-only closed-loop systems may be sufficiently safe [72]. The present unavailability of room-temperature stable glucagon is a limitation, as well as unavailability of dual chamber pumps, and results from studies evaluating chronic delivery of subcutaneous glucagon are required. Developments in these fields are taking place.
At present, multiple devices are used in most closed-loop prototypes to receive signals and compute and control insulin delivery, and bulkiness, size and weight compromise user-friendliness, particularly in youth [73]. The system complexity including communication between devices increases the risk of system failures which should be minimised to facilitate adherence and use in outpatient settings. Alongside the complexity and size of current prototypes, the number of skin perforations needed for glucose sensing, insulin and, in case of bi-hormonal systems, glucagon delivery is a major barrier to practical use, especially in younger children. Developments of single-port technologies are ongoing and could address wear issues [74].
Wider application of closed-loop systems will be supported by the establishment of training resources for users, carers and healthcare professionals, support network to manage technical issues and integrated data management. Collaboration with the medical device industry and healthcare insurance companies is needed to assure availability, affordability and reimbursement.
Closed-loop systems and interventions to preserve beta-cell function may serve as a bridge to a cure for type 1 diabetes until stem-cell therapy or other curative approaches become reality. Long-term evaluations of closed-loop systems and improvements of closed-loop components and their usability are priority to support gradual integration into and wider use of commercially available systems. Following threshold suspend and predictive low glucose suspend, a single-hormone closed-loop system directing insulin overnight and between meals with manual delivery of partial or full prandial boluses might be the next commercially available closed-loop technology [75]. More comprehensive approaches are expected to follow.
References
1.
2.
Group DCCT (1994) Effect of intensive diabetes treatment on the development and progression of long-term complications in adolescents with insulin-dependent diabetes mellitus: diabetes control and complications trial. J Pediatr 125:177–188CrossRef
3.
Acerini CL, Williams RM, Dunger DB (2001) Metabolic impact of puberty on the course of type 1 diabetes. Diabetes Metab 27:S19–S25PubMed
4.
Cryer PE (2008) The barrier of hypoglycemia in diabetes. Diabetes 57:3169–3176CrossRefPubMedPubMedCentral
5.
6.
7.
Barnard K, Thomas S, Royle P et al (2010) Fear of hypoglycaemia in parents of young children with type 1 diabetes: a systematic review. BMC Pediatr 10:50CrossRefPubMedPubMedCentral
8.
9.
Gaudieri PA, Chen R, Greer TF et al (2008) Cognitive function in children with type 1 diabetes: a meta-analysis. Diabetes Care 31:1892–1897CrossRefPubMedPubMedCentral
10.
Barnea-Goraly N, Raman M, Mazaika P et al (2014) Alterations in white matter structure in young children with type 1 diabetes. Diabetes Care 37:332–340CrossRefPubMedPubMedCentral
11.
Maahs DM, Horton LA, Chase HP (2010) The use of insulin pumps in youth with type 1 diabetes. Diabetes Technol Ther 12(Suppl 1):S59–S65PubMed
12.
Misso ML, Egberts KJ, Page M et al (2010) Continuous subcutaneous insulin infusion (CSII) versus multiple insulin injections for type 1 diabetes mellitus. Cochrane Database Syst (1)Rev:CD005103. http://www.ncbi.nlm.nih.gov/pubmed/20091571
13.
14.
Heinemann L (2009) Insulin pump therapy: what is the evidence for using different types of boluses for coverage of prandial insulin requirements? J Diabetes Sci Technol 3:1490–1500CrossRefPubMedPubMedCentral
15.
Jones SM, Quarry JL, Caldwell-McMillan M et al (2005) Optimal insulin pump dosing and postprandial glycemia following a pizza meal using the continuous glucose monitoring system. Diabetes Technol Ther 7:233–240CrossRefPubMed
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree