Fig. 3.1
Active insulin treatment with high insulin doses at diagnosis was associated to higher C-peptide in T1D children (Ludvigsson Thesis 1976)
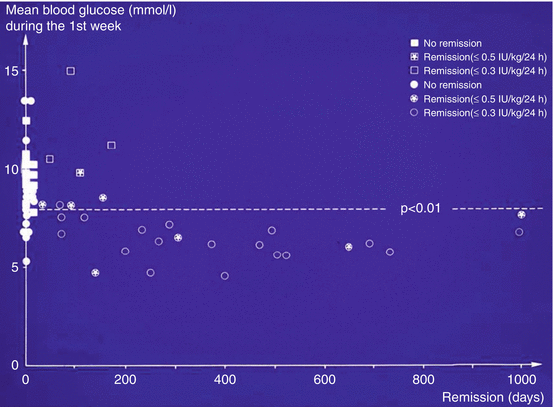
Fig. 3.2
Patients with active insulin treatment which pressed down blood glucose to a low mean during the first week after diagnoses more often got a long partial remission with stable blood glucose parallel to low insulin requirement
With this background knowledge, my discussions with Lise Heding (1984) encouraged studies showing that insulin treatment could be used to prevent autoimmune diabetes in experimental animals [24]. Insulin treatment might not only cause beta cell rest and thereby recovery of beta cell function but also decrease of autoantigen presentation, which might dampen an ongoing autoimmune process. It was shown that presentation of the so-called 64kD [25], later shown to be GAD, glutamic acid decarboxylase [26], increased dramatically when beta cells were stimulated by increasing glucose concentrations [27]. Furthermore, hard-working beta cells could also be sensitive to oxidative stress and inflammatory mechanisms [28].
This and other supports lead to studies in humans suggesting that insulin treatment could be used to prevent T1D [29]. The encouraging results lead to the Diabetes Prevention Trial using insulin SC and also peroral insulin. The results were negative. Insulin SC did not prevent T1D in that regimen used [4]. However, peroral insulin might have had an effect. Although the study failed to reach the primary endpoint, post hoc analyses using the inclusion criteria first planned for the peroral arm of DPT, with IAA > 80 RA, then there was a significant effect [30]. Thus, with inconclusive results a new trial became necessary and is now ongoing. Other ways beside sc or oral to present insulin as an autoantigen have been tried. There are arguments for the presentation of the antigens on the mucosa. However, intranasal administration of insulin had no effect on the prevention of T1D when given to young high-risk children [31].
Several data still suggest that insulin is central to autoimmunity in T1D, and therefore there are more studies using insulin. A combination of a diabetes-specific antigen and the metabolically inactive insulin B-chain fragment, which contains an epitope recognized by the immune system, has been combined with Freunds adjuvant. Twelve newly diagnosed T1D adults were randomized to either a single dose of this “vaccine” or placebo in a double-blind pilot study [32]. There was no significant effect on C-peptide, but quite interesting effects on T-regulatory cells, which is encouraging to further studies. New projects are ongoing where the aim is to combine proinsulin peptides with IL-10 and present to the immune system.
Studies continue to try other versions of insulin and/or proinsulin fractions as autoantigens in an effort to create tolerance [33].
The idea to preserve beta cell function by beta cell rest at diagnosis of T1D was reconsidered decades after our first observations in the 1970s. Thus, diazoxide was used to block insulin secretion and give real beta cell rest. That treatment did lead to increased C-peptide values for some time but the effect was transient [34].
In summary several facts suggest that active insulin treatment with good metabolic control leads to a positive circle where the remaining beta cells get better conditions to recover and survive. Thus, in all studies trying any forms of intervention, an important basis is good insulin treatment, as physiological as possible.
3.3 Other Ways to Preserve Beta Cell Function
Several attempts with immune interventions t to preserve residual beta cell function have been made, but so far with limited benefits or with unacceptable adverse effects [35–42]. However, in most recent years, a number of new interventions have appeared or been improved, and we should hopefully see a number of different trials in the near future. The approaches can be divided into different areas as shown below.
3.3.1 Immunosuppression
Cyclosporin has been regarded as the proof of concept [36], and although its adverse effects were too severe for single-drug treatment, cyclosporin has returned as a possible therapy in combination with other drugs.
AntiCD3 treatment has been proposed since the 1980s, but not until recent years, treatment with monoclonal antibodies has shown transient efficacy, both with teplizumab (formerly called hOKT3gamma1(Ala-Ala)) and otelixizumab (ChAglyCD3) [43, 44], with at least 6–12 months delay of decline in beta cell function. The difference in C-peptide created during this short effect persists, and later follow-up has shown that, especially in those patients with the best insulin secretion at diagnosis, the difference between treatment arm and placebo remains 4–5 years later [45, 46]. The mechanism of action is not fully understood. It is possible that monoclonal antibodies against CD3 deplete or marginate effector T-cells, in a similar way as the action attributed to rituximab (antiCD20 mAb) or alemtuzumab (antiCD52 mAb).
AntiCD3 treatment, depending on dose and regimen, may lead to adverse events and risks. A trial with teplizumab when the patients were aimed to receive three cycles of teplizumab 6 months apart had to be stopped already after ten subjects were included, six with active treatment, who all experienced cytokine release syndrome and lymphopenia, and two of them with serious adverse events [46]. However, with lower dose in the so-called Protegé trial, teplizumab caused no more adverse events than seen in the placebo-treated group and not any serious infections as could have been feared [47]. The preservation of C-peptide was quite impressive, at least in patients treated within 6 weeks from diagnosis, those with rather good residual insulin secretion and children and adolescents [48]. The effect was even more clear at 2 years of follow-up [49]. Thus, with adequate doses, there seem to be clinically important effects with justifiable risks and adverse events. Less adverse events also decreases the problem to interpret the results, as otherwise severe and typical adverse events might reveal for the patients and treating teams who has active treatment or not. Expectation that a patient participates in an active arm may influence the treatment, not least because of the attitude of the patient even when the physician tries to avoid bias, as change of treatment and lifestyle may influence residual insulin secretion [49]. Thus, with lower but adequate doses, these problems have been overcome. Still new studies are needed to find dosing and treatment design with as few and mild adverse events as possible but hopefully even better efficacy. This will make intervention with antiCD3 interesting, either as single-drug therapy, but probably even more so in combination therapies with other drugs.
AntiCD20 monoclonal antibodies (rituximab) directed against the B-cells should actually have been useless according to the paradigm that T1D is a pure T-cell disease. But antiCD20 administration gave a significant C-peptide preservation [50]. However, the effect was limited and disappeared already within a year. During the first infusion, 93 % of the patients given rituximab had adverse reactions as compared with 23 % of those given placebo, and it cannot be totally excluded that this might have influenced the attitude of patients and diabetes teams, thereby also influencing the results. In any case this trial has been important by pointing to the role of both T- and B-cell function and possible importance of combination therapies.
Tumour necrosis factor-alfa (TNF-alfa) may play an important role for the autoimmune process leading to T1D. Etanercept is a recombinant soluble TNF-alfa receptor fusion protein that binds in a way that TNF-alfa is cleared from the circulation and its biological activity is thereby blocked. Etanercept has been used with good effect in several autoimmune diseases such as psoriasis and juvenile rheumatoid arthritis. A pilot study has been performed in children with new onset of T1D [51]. In a double-blind placebo-controlled trial, patients were given either etanercept 0.4 mg/kg body weight (max 25 mg) or placebo twice a week. Because of difficulties to recruit patients, only 18 patients were included. However, the effect was impressive! From baseline to 24 weeks, the change in C-peptide area under the curve showed a 39 % increase in the actively treated group compared to a decrease of 20 % in the placebo group, and this was associated with a corresponding decrease of insulin dose (18 %) in the etanercept group compared to an increase of insulin dose (23 %) in the placebo group. There were no serious adverse events, although the etanercept group showed a slight increase of mild to moderate adverse events, e.g. twice as often cold symptoms, three cases of paraesthesia and some cases with abdominal pain. Further studies that are needed will hopefully soon start. Thus, there are plans for studies combining etanercept both with GAD-alum vaccinations, with the hope to prolong the effect, and also with vitamin D which may have several supportive effects as it contributes to Th2 deviation, improve dendritic cell function and both protect beta cells and increase insulin sensitivity.
Interleukin-2 is supposed to be another important part of the autoimmune process and monoclonal antibodies (daclizumab) that binds to CD25. The alfa subunit of IL-2 expressed on activated lymphocytes has shown to be useful in induction therapy before islet transplantation, and it has also been used in treatment of several autoimmune disorders such as multiple sclerosis and uveitis. Daclizumab has been tried in newly diagnosed T1D in combination with mycophenolate mofetil (MMF), which has potent cytostatic effects on lymphocytes [52]. However, the result was negative. The treatment caused serious adverse events in many patients, especially the combination therapy (34 % of the patients), but also in the group treated with MMF alone (16 % of the patients). As many as 23/126 patients had to stop the treatment because of adverse events such as elevated liver enzymes and EBV positivity. Thus, this rather dangerous treatment did not preserve beta cell function, nor did it decrease insulin requirement or HbA1c. Ustekinumab which interferes with IL-2 and thereby CD12 and CD23, which seems effective in several autoimmune diseases [53], might be of interest also in type 1 diabetes.
IL-1 has also been regarded as an important cytokine in the destructive process in T1D [54] although not only as part of the autoimmune process but rather as an important part of the inflammatory process to which also the glucose toxicity contributes when metabolism deteriorates. Efficacy of IL-1 antagonists was shown in short-term experiments in type 2 diabetes [55]. However, in T1D the treatment with IL-1 inhibitor, as well as IL-1 receptor blocker, failed [56]. There were adverse events but no efficacy. Still there are theoretical reasons to believe that it may not be sufficient to stop the autoimmune process, but the inflammatory process should also be stopped. Therefore, there may still be a place for blocking of IL-1, as a complement to other therapies, also in T1D.
A dramatic approach to stop the immune process is to kill most of the effector cells by heavy cytostatic treatment and then save the patient by an autologous stem cell transplantation. Such treatment has been performed [57], even in younger patients, which raises serious ethical questions [58]. It can be questioned if the prognosis of T1D is severe enough to justify that sort of very heavy and dangerous intervention. But the treatment seems to show that heavy immune suppression does work. The results suggest that such heavy treatment can stop the autoimmune process, at least for some time, and that this effect saves residual insulin secretion. Effective but more safe methods are needed.
3.3.2 Immunostimulation
Linomide was used with remarkably good efficacy to stop the autoimmune process in many different diseases such as multiple sclerosis but also T1D [40]. It was an immunostimulatory drug aimed for cancer treatment but showed to prevent several autoimmune diseases in experimental animals. It had quite good effects also in human autoimmune diseases. This illustrates that suppression of the immune system is not the only way of decreasing autoimmunity, but a new balance via stimulation of the immune system may also work.
Ingested interferon-alfa has an immunomodulatory effect, although the mechanisms are unknown. It has been used with some positive effects in multiple sclerosis without apparent toxicity. Oral ingestion of interferon-alfa has been tried to preserve beta cell function in children and adolescents with T1D [59]. The treatment had a significant effect with preservation of C-peptide (area under the curve) after 12 months of follow-up. There was no effect on insulin dose or HbA1c. One serious adverse event occurred in the treatment group (Staphylococcus septicaemia), but otherwise there were no adverse events. Further studies are needed to confirm and develop this treatment.
Adding stem cells from the umbilical cord might be regarded as another effort to stimulate or modulate the immune system. The theoretical basis for such treatment is speculative, and it is a weakness that open studies have been performed without controls [60]. However, as pilot study, this may have to be accepted. So far there is no evidence of any effect, neither on the autoimmune process nor on the residual insulin secretion, but further studies are needed, and it should then be possible to solve the practical and ethical problems to make such studies both blinded and controlled with sham transfusions.
3.3.3 Immunomodulation
The autoimmune process includes different steps, of which presentation of antigen is important. Instead of trying to block certain cells, one way of modulating the immune response may be to present antigen(s) in a way that the immune system turns away from a destructive reaction into tolerance.
Already in the 1980s, the paediatricians Iron Cohen et al. worked with the use of heat shock proteins [61], and later one peptide, DiaPep277, has been tried with intriguing results. Thus, it looked as if a very easy SC administration of DiaPep277, causing no adverse events, leads to beta cell preservation in adults [62]. However, the result was dependent on sporadic outliers and could not be confirmed when tested in diabetic children and adolescents [63]. Immunological effects [64] seemed to be promising enough for further studies, and a review of performed studies suggested optimism [65]. However, a phase III trial gave results which are quite difficult to interpret [66]. Although there was a significant preservation of C-peptide seen after glucagon stimulation, there was no such effect seen after the more clinically relevant mixed-meal tolerance test. In addition no effect on the immune system was seen. Taken together these results cause scepticism until further results can support one or the other view. This is even more true since treatment with Diapep277 is questioned; several publications have been retracted because of fraud of research!
In accordance with the use of allergens in immunotherapy of allergy to create tolerance, it seems appropriate to try creating tolerance against autoantigens in autoimmunity [67]. In type 1 diabetes, we have several autoantigens, and some have been used in trials to prevent diabetes. The use of insulin has been described earlier in this paper. Administration of rather large doses of insulin SC every day as in the Diabetes Prevention Trial [4] can perhaps not be regarded as immunotherapy but rather beta cell support. This did not work as prevention. Oral insulin is an immune intervention and might have had some effect in a prevention trial (see above).
GAD-alum treatment is so far the only autoantigen administration that has shown efficacy in some, but not all, studies. Already in the dose-finding study of LADA patients, the small group of patients receiving 20 mikrog GAD-alum (Diamyd) twice with an interval of 1 month suggested beta cell preservation [68], which still persisted 5 years later, without any treatment-related adverse events [69]. In a phase II trial on diabetic children and adolescents with recent onset diabetes [70], the pre-specified endpoint fasting C-peptide was significantly improved first after 30 months, and there was still an effect after 4 years in those with <6 months of diabetes duration at treatment [71]. The treatment was extremely simple, very well tolerated by the patients, and has so far shown no treatment-related adverse events. There were very convincing immunological results which supported the clinical data [72–77]. However, a European phase III trial failed to reach primary endpoint after 15 months [78]. There was quite an impressive effect in certain countries and especially in males, but as the effect was minimal in Sweden from where most of the patients were recruited, the total study failed. Possible reasons for this difference between phase II and phase III have been discussed. In the phase II [71], no other vaccination was allowed close to the GAD treatment, while in the phase III trial influenza vaccination was allowed [78]. The Swine flu and its vaccination coincided in time with the GAD treatment. In Sweden, the vaccine contained squalene, which has been suspected to contribute to autoimmune reactions, and it can be noticed that the flu vaccination caused an epidemic of narcolepsy, an autoimmune disease, in Sweden and Finland, where such vaccine was used. Later follow-up of those few patients in the European phase III trial who had passed 30 months before termination of the trial showed a significant preservation of C-peptide in those patients treated with two doses of 20 mikrog GAD-alum [79]. An American phase III trial was terminated prematurely with no results available, and a phase II trial performed by TrialNet with three doses given to T1D patients aged 4–45 years failed [80]. The phase II trial [71] had proven the concept, and new studies are needed to test dosing, intervals, duration of treatment and combinations of other treatment modalities [39]. New studies have started combining GAD-alum with vitamin D and ibuprofen, and further trials using GAD-alum in combination with etanercept is ongoing.
3.3.4 Beta Cell Protection
Instead of intervening in the autoimmune process, another approach has been to try to protect the beta cells.
Nicotinamide has been tried both in prevention trials (3) and in intervention at the onset of T1D [81] with minimal or no effect.
As mentioned above, daily insulin injections were tried to support the beta cells in the DPT trial aiming at the prevention of type 1 diabetes, but this had no effect [4].
Another way of protecting the beta cells and causing beta cell rest is to give diazoxide which blocks insulin secretion. As mentioned above in children and adolescents with newly diagnosed type 1 diabetes, this treatment as expected postponed the insulin secretion, and the maximal function was delayed. But the C-peptide AUC was just the same in the treatment group compared to placebo [34]. A Norwegian study showed that treatment with diazoxide in adult patients had no effect neither on C-peptide nor the immune parameters measured, while there was a difference in insulin requirement which is difficult to explain [82].
Vitamin D contributes to Th2 deviation of the immune system and also improves function of dendritic cells important for autoantigen. Furthermore, vitamin D seems to protect beta cell function and may also improve insulin sensitivity. There are inconclusive suggestions that lack of vitamin D might increase the incidence of type 1 diabetes [83]. Treatment with 1alfa,25-dihydroxyvitamin D3 in a rather high was tried in a double-blind placebo-controlled trial including 40 young adult patients with type 1 autoimmune diabetes with a duration less than 2 months. There was no effect on C-peptide, insulin dose or metabolic parameters [84]. Thus, as a single drug, vitamin D may be insufficient, but it should be and is tried in combination therapies.
3.3.5 Beta Cell Regeneration
The general opinion was earlier that when somebody gets diabetes, there is no longer any capacity of the beta cells to regenerate. In recent years some studies have suggested that the paradigm may be wrong and that beta cells certainly can regenerate (10.11). And studies of the pancreas from dead T1D patients in the nPOD material have confirmed that in many patients there are a large number of islets with beta cells [9].
GLP-1 might stimulate beta cell regeneration [85]. Therefore, this drug has been used in T1D but so far the results are meagre. In one trial, the authors tried a GLP-1 agonist (exenatide) in combination with monoclonal antibodies interfering with IL-2 (daclizumab) given to patients with long-standing T1D, but with some residual insulin secretion, to see whether the treatment could improve C-peptide as a sign of more functioning beta cells. The result was negative [86].
Efforts to discover therapeutic targets for enhancing islet neogenesis have led to the discovery of a peptide called INGAP (islet neogenesis-associated protein), which seems to have pancreatic regenerative capacity. When INGAP has been administered to animals, their beta cell mass has increased with reversal of hyperglycaemia, and hopefully INGAP has regenerating potential in humans. Daily introduction of INGAP or placebo has been tried in a double-blind randomized trial in both type 1 and type 2 diabetic patients [87]. A per-protocol analysis showed increased arginine-stimulated C-peptide during the treatment period, but this effect was not seen 30 days after the end of treatment, which one would have expected if the results should be interpreted as a sign of increased beta cell mass. Furthermore, the drop-out rate was high, mainly because of adverse effects on the injection site in the actively treated group, and intention-to-treat analyses showed no effect on C-peptide.
3.4 We Need More and Better Clinical Trials!
Studies on autoimmune diabetes have to a great extent been performed on experimental animals. Although of great help to elucidate mechanisms and create new ideas, we have learnt that there is a huge gap between autoimmune diabetes in mice and men. There are more than 500 ways to prevent autoimmune diabetes in animals, but none of these work in humans! Clinical studies on human beings are necessary. Unfortunately such studies are much too rare. There are several probable reasons:
Many doctors seem to be satisfied with the existing treatment and more enthusiastic of the improving modern technical devices, even though these do not mean cure.
Patients and their parents/relatives get the message that they can “live a normal life”, so why then bother and take the risk and burden of participating in studies. Too late many of them will find, unfortunately, that the life was far from normal, and those unhappy who do not manage to get a good metabolic control may meet serious complications.
The scientific society seems to be fixed to certain paradigms, and nothing can be tried unless proven in the right order with enough studies in experimental animals (even though we know that they may be rather irrelevant for the effect in humans) and with the right opinion leaders in agreement to be able to get funding.
Authorities put up rules, endpoints and requirements, which sometimes makes it very difficult to proceed.
The big companies may be more interested in producing drugs or devices which can be sold to large populations for very long time. Cure of diabetes is probably not a good affair.
Clinical trials could and should be done. There are reasonably well-founded ideas we know enough to act. On the other hand, we have of course to be cautious both in our design of studies and in our interpretations of clinical trials as there are several problems:
T1D may seem to be a well-defined disease in relation to many other diseases such as allergy, asthma, joint diseases, celiac disease, etc., but still it is not always clear how diabetes should be classified. All T1D patients do not have autoantibodies at diagnosis, and all do not have risk of HLA type. Furthermore, although C-peptides rarely reach extreme values above 1.0 nmol/l, even children may have a broad range of residual C-peptide making it sometimes difficult to differ between type 1, type 2 and MODY [88]. Furthermore, phenotypic type 2 diabetes may have autoantibodies and/or other signs of diabetes-related autoimmunity. Thus, sometimes it is difficult to be sure when we deal with pure T1D. This becomes especially evident when trials include patients with different ethnic origins, with a large age range, especially obese adults, as type 2 or LADA becomes much more common among young adults diagnosed with insulin-dependent diabetes. The degree of autoimmunity may differ, shown, for example, in different numbers and titres of autoantibodies, which might influence the process as well as response to the treatment.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree