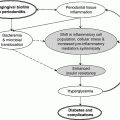
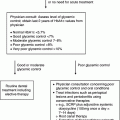
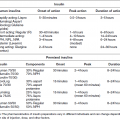
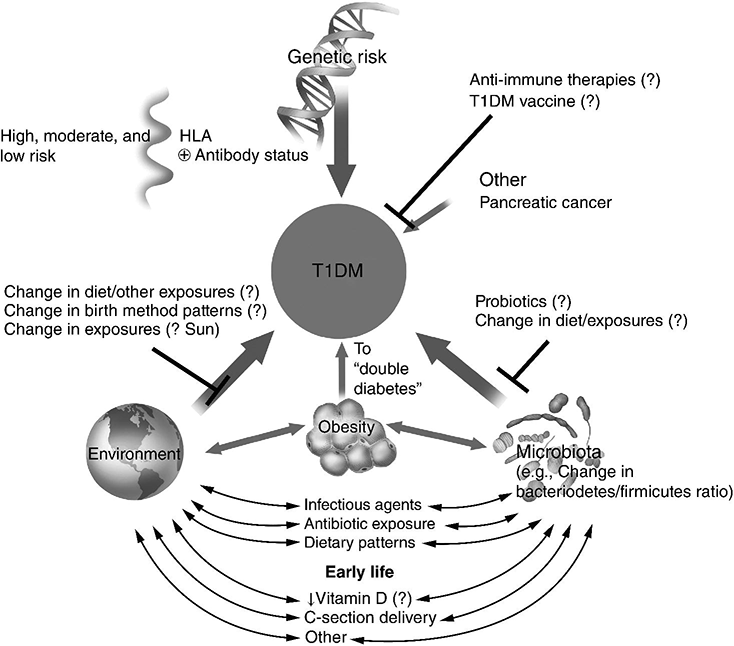
Chapter 1 Ravichandran Ramasamy, PhD and Ann Marie Schmidt, MD The defining characteristics of diabetes, irrespective of the precise etiology, relate to the presence of hyperglycemia. The American Diabetes Association (ADA) has set forth specific criteria for the definition of diabetes. In the ADA guidelines, the following are necessary for the diagnosis of diabetes: (1) hemoglobin A1c (HbA1c) equal to or greater than 6.5% OR (2) fasting plasma glucose equal to or greater than 126 mg/dl OR (3) two-hour plasma glucose equal to or greater than 200 mg/dl during an oral glucose tolerance test (OGTT) (glucose load containing 75 grams anhydrous glucose dissolved in water) OR (4) in a patient with classic symptoms of diabetes or during a hyperglycemic crisis, a random glucose of equal to or greater than 200 mg/dl suffices to diagnose diabetes [1]. In this chapter, we will review the major types of diabetes and the etiologic factors that are known to or are speculated to contribute to these disorders. Furthermore, we will take the opportunity to present an overview of emerging theories underlying the pathogenesis of type 1 and type 2 diabetes. Types 1 and 2 diabetes constitute the vast majority of diabetes cases. Interestingly, both of these types of diabetes are on the rise worldwide [2, 3]. In addition to types 1 and 2 diabetes, we will also discuss gestational diabetes. Often a harbinger to the ultimate development of frank type 2 diabetes in the mother, this form of diabetes is potentially dangerous to both the mother and the developing fetus. Finally, we will discuss the syndromes known as MODY or maturity onset diabetes of the young. The disorders underlying MODY have very strong genetic components and are due to mutations in multiple distinct genes. The greatest long-term danger of diabetes, irrespective of the etiology, lies in the potential for complications. The complications of the disease are insidious, deadly, and difficult to treat or reverse; hence, there is great urgency to identify specific means to prevent or mitigate these most common types of diabetes. Type 1 diabetes accounts for approximately 5–10% of all cases of diabetes [1]. The countries with the highest incidence of type 1 diabetes include Finland and Sardinia [4]. Type 1 diabetes is usually diagnosed in childhood, hence the original classification “juvenile onset diabetes.” Indeed, type 1 diabetes accounts for more than 90% of diabetes diagnosed in children and adolescents. Given that the disease is often diagnosed in adults, however, even into advanced age, the term “type 1 diabetes” has been adopted to more accurately reflect the diversity of affected ages. In type 1 diabetes, the primary etiology is due to a cellular-mediated autoimmune-mediated destruction of the β cells of the pancreas. Traditionally, in subjects with type 1 diabetes, autoantibodies may be detected that reflect the underlying attack against these cells [5]. These include autoantibodies to insulin, to GAD65, and to IA-2 and IA-2β (the latter two are tyrosine phosphatases). These antibodies are often detected up to years before the diagnosis of type 1 diabetes [6]. In most subjects with type 1 diabetes, one or more of these antibodies is evident. Indeed, in vulnerable subjects, such as first-degree relatives of affected individuals, the presence of these autoantibodies is often, but not always, a harbinger of the eventual diagnosis of diabetes. Hence, these antibody profiles may be used to predict the risk of diabetes in the siblings and relatives of affected subjects with type 1 diabetes [6]. More than forty years ago, type 1 diabetes was found to have very strong links to the human leukocyte antigen (HLA)-encoding genes [7]. The largest study to address this issue was known as the Type 1 Diabetes Genetics Consortium (T1DGC). This group was composed of an international collaboration and amassed more than 14,000 samples [8]. By far, the greatest association to type 1 diabetes was found in the HLA, particularly in the HLA DR-DQ haplotypes. Furthermore, other genes found to have strong genetic association were in polymorphisms identified in the insulin gene [9]. The researchers of T1DGC earlier reported that beyond these two associations, two other loci were found to have odds ratios (ORs) greater than 1.5, and included PTPN22 and IL2RA [9]. However, the ORs for these genes were relatively much lower than that of the HLA region, consistent therefore with the overall strong role of the HLA in the susceptibility to type 1 diabetes. A number of groups have published the results of genome wide association studies (GWAS) in type 1 diabetes and identified more than 40 potential susceptibility loci in the disease [11]. Candidate genes identified in this approach included those encoding IL10, IL19, IL20, GLIS3, CD69, and IL27; these are all genes strongly linked to the immune/inflammatory response [10]. In their report, Bergholdt and colleagues integrated the data from these GWAS studies and translated them to a more functional level, that is protein-protein interactions and, finally, they tested their relevance in human islets and in a β cell line, INS-1 cells (rat insulimona-derived cells) [11]. First, they performed a meta-analysis of the type 1 diabetes genome wide Association studies that were available. From these, they identified 44 type 1 diabetes non-major histocompatibility complex (MHC) low density (LD) regions with significance; these regions contained more than 395 candidate genes. They then performed network analysis studies with the intention to more deeply probe network connections and protein-protein interactions. From this work, 17 protein networks were identified (which contained 235 nodes) containing at least two genes from different type 1 diabetes LD regions [11]. To follow up on these findings, human islets were exposed to pro-inflammatory cytokines and comparisons were made between the treated and untreated human islets (retrieved from eight donors). From this, the following genes were found to be significantly impacted by the cytokine stimulation in the human islets: IL17RD, CD83, IFNGR1, TRAF3IP2, IL27RA, PLCG2, MYO1B, and CXCR7. Interestingly, the study design suggested that perhaps these traditionally inflammation-associated factors were being produced by pancreatic β cells and not necessarily solely by immune cells. To test this specific point, rat INS-1 cells were treated with cytokines and the above eight genes were examined. Indeed, all but IL27ra were identified in the stimulated INS-1 cells [11]. In the case of cultured INS-1 cells, no immune cells are present, therefore suggesting the interesting possibility that these factors may be produced both by islet β cells themselves as well, likely, by infiltrating inflammatory cells. Examples of non-HLA genes linked to type 1 diabetes are illustrated in Table 1.1. Table 1.1 Examples of non-HLA type 1 diabetes-associated loci. There is strong evidence that links the pathogenesis of type 1 diabetes to immune-mediated mechanisms of β cell destruction, including the detection of insulitis, the presence of islet cell autoantibodies, activated β cell-specific T lymphocytes and, as considered above, association of the disease with a restricted set of class II major histocompatibility alleles [12]. Importantly, the rate of the development of type 1 diabetes after the appearance of autoantibodies may be quite variable, reflecting perhaps the contribution of protective mechanisms (such as CD4 + -T regulatory cells and other regulatory cells such as invariant natural killer T [NKT] cells). Such protective factors may differ among individuals, thereby possibly accounting for the variable progression of damaging autoimmunity and the appearance of diabetes. The diagnosis of type 1 diabetes, often made by the appearance of diabetic ketoacidosis [13], is linked to the absence or near absence of plasma C-peptide (N-terminus fragment of insulin that is used to monitor the ability to produce insulin) [14]. It has been suggested that particularly in adults, residual β cell function may be retained for years after the appearance of autoantibodies without manifestation of ketoacidosis. In the sections to follow, we consider some of the specific factors that have been linked to the pathogenesis of type 1 diabetes. As discussed above, the incidence of type 1 diabetes is on the rise at a rate of 3–5% per year that is doubling every 20 years. This is occurring particularly in very young children and is present more often in subjects bearing the low risk alleles [15, 16]. What accounts for these findings? Certainly, genetic risk cannot explain the overall rise in this disorder over relatively short time periods, thereby placing a spotlight on so-called “environmental” factors. For example, it has been suggested that acute infections such as those that are bacterial or viral in nature may precipitate the disease. After such an acute onset, subjects may often enter so-called “honeymoon” periods during which time hyperglycemia abates and the subjects do not require insulin for survival. Examples of viruses linked to type 1 diabetes include cytomegalovirus, coxsackie B, mumps, rubella, Epstein-Barr virus, rotavirus, and varicella zoster virus [17]. An intriguing example of an association between an environmental trigger and type 1 diabetes was speculated to have occurred in Philadelphia in 1993. During the first six months of that year, a substantial rise in the incidence of type 1 diabetes among children was observed. It had been noted that in the two years prior to this event, an outbreak of measles had occurred in the same location, thereby raising the hypothesis that the viral infection stimulated factors that caused type 1 diabetes to emerge in vulnerable children [18]. In the human intestine, it is estimated that more than 100 trillion bacteria reside and colonize the organ [19]. Far from being a passive factor in the host, these bacteria critically interface with the immune and metabolic systems. Studies have suggested that specific classes of bacteria may exert effects on the immune system. For example, Bacteroidetes were shown to reduce intestinal inflammation [20]. Segmented filamentous bacteria were suggested to induce Th17 immune responses [21]. Th17 immune responses are usually linked to the clearance of extracellular pathogens during periods of infection; Th17 T cells produce major cytokines that induce inflammation such as IL6 and IL8 [22]. In animal models, interference with the normal gut microbiota has impacted the incidence of type 1 diabetes. For example, raising two major mouse and rat models of type 1 diabetes in germ-free or altered flora environments resulted in the animals developing insulitis and type 1 diabetes at accelerated rates [23, 24]. In contrast, feeding type 1 diabetic-vulnerable animals antibiotics significantly delayed or prevented type 1 diabetes [25]. Based on these considerations, the hunt is on to identify the specific phyla of bacteria that display adaptive/anti-type 1 diabetes impact. So called “probiotics” might one day be identified as treatments to alter the course of type 1 diabetes development, such as the protective effects shown by treatment of type-1-diabetes-vulnerable rats with Lactobacillus johnsonii [26]. In the context of the microbiome, it is interesting that type 1 diabetes may appear more frequently in individuals born by Cesarean section vs. natural deliveries [27]. It was shown that in the earliest time of life, the gut microbiome constituents differ in these two states with skin vs. vaginal microbes, respectively, reflecting the major microbiota in subjects born by these two methods. Hence, via Cesarean birth, there is a delay in the colonization of the gut with organisms such as Bacteroides, Bifidobacterium, and Lactobacillus; the extent to which this might account for increased type 1 diabetes is not clear [28]. The possibility that the distinct phyla of bacteria may influence the types of immune/inflammatory cells in the gut is under consideration as a contributing factor in type 1 diabetes. In this context, type 1 diabetes manifests with an increased number of intestinal inflammatory cells in parallel with reduced numbers of FoxP3 + CD4 + CD25+ T lymphocytes [28]. Hence, it is possible that alteration of the gut microbiota might lead to alterations in immune cell patterns in the gut. In studies in Finnish subjects with type 1 diabetes, experimental analyses have shown that within the gut microbiome, there is a change in the ratio of two key phyla of bacteria—an increased percentage of Bacteroidetes in parallel with a lower percentage of Firmicutes [29]. Whether this association is linked mechanistically to type 1 diabetes has yet to be clarified. 16S sequencing and metagenomics are current strategies under way to determine if there are actual mechanistic links between alterations in the gut microbiome and the susceptibility to type 1 diabetes. Vitamin D, or 1,25-dihydroxyvitamin D3 (1,25(OH)2D3), has been linked at multiple levels to the pathogenesis of type 1 diabetes. Most importantly, vitamin D plays immunomodulatory roles in cells that express the vitamin D receptor (VDR). Included among such cells are antigen presenting cells, activated T cells, and pancreatic islet β cells [30]. Studies have shown that administration of vitamin D or analogues may exert protection against type 1 diabetes in non-obese diabetic (NOD) mice [31]. Experimental studies to discern the underlying mechanisms showed that administration of 1,25(OH)2D3 reduced inflammatory cytokine (such as IL6) production in parallel with increased regulatory T cells. On the contrary, mice deficient in 1,25(OH)2D3 were shown to be at higher risk of developing type 1 diabetes [32]. What is the evidence in human subjects linking vitamin D to type 1 diabetes? Insights into this question became evident in the study of vitamin D receptor (VDR) polymorphisms. The gene encoding the VDR is located on chromosome 12q12-q14 in the human and single nucleotide polymorphisms (SNPs) have been shown to alter the function of the receptor. The results of studies examining these SNPs have yielded contrary data but the largest meta-analysis to date showed that one of the VDR polymorphisms, BsmI, was associated with significantly increased risk of T1D but other SNPs, including FokI, ApaI, and TaqI, did not display a significant association with T1D [33]. It remains possible that the VDR locus is not itself the disease affecting locus; rather, the VDR may in fact be a marker locus in linkage equilibrium with the true disease locus. Certainly greater functional studies on the SNPs and vitamin D actions are essential to mechanistically link the SNPs to pathological function of the receptor and associations with the pathogenesis of T1D. What about the levels of vitamin D? Multiple studies in different countries have addressed this question and suggest that lower levels of vitamin D might be related to type 1 diabetes. For example, studies in Switzerland, Qatar, North India, the northeastern United States, and Sweden suggested that levels of vitamin D were lower in type 1 diabetic subjects vs. control subjects. In contrast, in the sun-enriched state of Florida no differences in vitamin D levels were noted between type 1 diabetic subjects and their unaffected first degree relatives and control subjects [30]. Interestingly, support for the North to South incidence of type 1 diabetes emanates from the fact that sun exposure, which is strongly linked to latitude, has possible relationships with type 1 diabetes. Specifically, a number of observational studies have suggested increased type 1 diabetes prevalence in the northern, less sun-exposed latitudes vs. more sun exposed regions. In the EURODIAB study, the incidence of diabetes was found to be higher in the northern region study centers vs. the southern centers, with the exception of Sardinia. Sardinia is considered to be in the southern region but it reported higher rates than those observed in neighboring southern region sites [34, 35]. Not taken into account in these studies are the genetic variations and other vulnerabilities and associations with type 1 diabetes, such as affluence (the latter associated with type 1 diabetes) [36]. The above considerations suggest that supplementation with vitamin D might be protective in type 1 diabetes. When a meta-analysis of multiple observational studies was performed, the results suggested that the incidence of type 1 diabetes was reduced by up to 29% in subjects given supplementation with vitamin D [37]. It is notable, however, that in these studies, concerns regarding many factors, such as reporting of vitamin D levels, doses of vitamin given, and the absence of documentation of vitamin consumption, as examples, limited the overall interpretability of these studies. Hence, a prospective randomized clinical trial is definitely needed to rigorously address these questions and establish possible causality between vitamin D and type 1 diabetes. At this time, no specific answer is available to unequivocally address this issue. Despite these caveats, however, it is essential to address this issue as supplementation with vitamin D should be feasible. In the sections above, we discussed some of the major factors impacting the etiology of type 1 diabetes. Of late, the issue of “double diabetes” has emerged; this term, first employed to describe this concept in 1991, suggests that there is an emergence of insulin resistance in subjects with type 1 diabetes [38, 39]. For example, in type 1 diabetic subjects with obesity or in whom even very high levels of exogenous insulin did not achieve euglycemia, insulin resistance was speculated to be present [38, 39]. The Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications (DCCT/EDIC) study suggested that a family history of type 2 diabetes significantly predicted excess weight gain in type 1 diabetic subjects [40]. Thus, the degree of peripheral insulin resistance might result from genetic and/or environmental factors (such as energy intake and physical activity). In fact, in the Pittsburgh cohort of the Epidemiology of Diabetes Complications (EDC) study, there is evidence that the prevalence of obesity has risen significantly in type 1 diabetic subjects, similar to the findings reported in the general population. From 1987 to 2007, this study showed that the prevalence of obesity rose seven-fold and that the prevalence of overweight rose 47% [41]. Although some of these changes might be attributable to insufficient glycemic control in the past decades, the overall premise is that the general increase in obesity/overweight has also impacted the type 1 diabetic subject population. Finally, it is plausible that the development of insulin resistance might be accounted for, in part, by the route of administration of therapeutic exogenous insulin. When insulin is administered by the subcutaneous route, this has been associated with relative peripheral hyperinsulinemia together with hepatic hypoinsulinemia. Such a regimen might ultimately lead to reductions in peripheral insulin-mediated glucose uptake and increased hepatic glucose production [42]. It remains to be seen which factors may underlie the observed insulin resistance in type 1 diabetes and how these might best be managed in type 1 diabetes. In summary, the incidence of type 1 diabetes is on the rise. As Figure 1.1 illustrates, there are multiple contributing factors. Although genetic factors are a major underlying cause, emerging evidence suggests that subjects with traditionally lower genetic risk alleles are being diagnosed with type 1 diabetes. These considerations strongly implicate so-called “environmental” factors in the multiple steps beyond genetic risk that are required before frank type 1 diabetes results. Insights into the interactions between the host and microbiome with respect to modification of genetic risk highlight the complexity of the factors that may significantly modify type 1 diabetes risk. Figure 1.1 Contributory factors to the development of type 1 diabetes. Multiple factors, from genetic risk to environmental influences and perhaps the interface with the gut microbiome, contribute to the pathogenesis of type 1 diabetes. Given that in the vast majority of subjects, genetic risk/antibody status may be discerned and tracked, novel interventions hold promise, ultimately, for the prevention of type 1 diabetes. Dangers in type 1 diabetes, however, include the possible influence of obesity on the rise in “double diabetes.” Efforts to minimize possible contributory risks for type 1 diabetes are essential. Type 2 diabetes is the most prevalent form of diabetes, accounting for up to 90–95% of diagnosed cases of diabetes, and is on the rise [1]. The International Diabetes Foundation (IDF) reported that in the age range of 20–79 years, approximately 285 million adults suffer from diabetes, a number which is expected to rise to approximately 438 million in the year 2030 [2]. In fact, about 90–95% of these cases will be in the type 2 diabetes classification. Older nomenclature referred to this form of diabetes as “non-insulin dependent” or “adult-onset diabetes.” In this form of diabetes, at least early in the course of the disease, subjects display insulin resistance with a “relative” deficiency of insulin. However, in the later stages of disease, some subjects are not able to produce sufficient amounts of insulin to compensate for the hyperglycemic stress [1]. This reflects underlying dysfunction of the pancreatic β cell. In type 2 diabetes, ketoacidosis seldom occurs; where it does occur, it may be precipitated by events such as infections. In cases in which very high levels of glucose are present, subjects may present with coma [43]. In general, the risk of developing type 2 diabetes rises with age and is associated with obesity and diminishing physical activity. Type 2 diabetes occurs more frequently in women who displayed gestational diabetes (GDM) during their pregnancies. Further, epidemiological evidence suggests that the incidence of type 2 diabetes is rising in childhood and adolescence, presumably due to increased obesity and reduced physical activity [44]. In type 2 diabetes, there is a very strong association with genetic factors. Many studies have addressed this issue and will be considered in the sections that follow. The genetics of type 2 diabetes must take into account two key underlying etiologies of the disease, that is, β cell function (insulin secretion) and insulin resistance [1]. In the pre-GWAS era, the strong genetic contribution to type 2 diabetes was determined via family and twin studies [45]. From these efforts, a major gene found to be linked to type 2 diabetes included CAPN10 (first described in a Mexican-American population) [46]. Others found that regions within chromosomes 5 and 10 were linked to type 2 diabetes, including within the latter, the TCF7L2 gene [47, 48]. Multiple independent studies confirmed that SNPs in TCF7L2 were linked to type 2 diabetes. The association of this gene with type 2 diabetes was confirmed in multiple distinct populations [49, 50]. Candidate gene approaches also identified PPARG and KCNJ11 (the latter a potassium inwardly rectifying channel subfamily J member 11) as susceptibility genes for type 2 diabetes [51, 52]. It was not until the GWAS era that more modern and effective approaches were used to identify susceptibility genes in type 2 diabetes. The first reported GWAS in type 2 diabetes was performed in a French cohort and was composed of 661 cases and 614 control subjects. The number of SNP loci covered in this study was 392,935. From this study, the following genes were identified as association signals in type 2 diabetes: SLC30A8, HHEX, LOC387761, and EXT2, and the study validated the association of the disease with TCF7L2 [53]. Following this study, the Icelandic company deCODE Genetics and its colleagues confirmed the association of type 2 diabetes with SLC30A8 and HHEX and added CDKAL1 [54]. Following this, three collaborating groups (Wellcome Trust Case Control Consortium/United Kingdom type 2 Diabetes Genetic consortium, the Finland-United States Investigation of NIDDM [FUSION] and the Diabetes Genetics Initiative [DGI]) published the findings confirming the association of type 2 diabetes with SCL30A8 and HHEX and added newly discovered associations with CDKAL1, IGF2BP2, and CDKN2A/B [55–57]. Following these discoveries, the need to increase sample size led to the above groups combining efforts to form the Diabetes Genetics Replication and meta-analysis, or DIAGRAM, consortium. Upon testing of an additional 4,549 cases and 5,579 controls, an additional five loci were discovered including JAZF1, CDC123/CAMK1D, TSPAN/LGR5, THADA, and ADAMSTS9 [58]. By the continued addition of new subjects into these studies, an additional 12 new loci were reported in 2010 [59].
Etiology of diabetes mellitus
Introduction
Type 1 diabetes
Genetics of type 1 diabetes
Locus
Description
Comments
PTPN22
Protein tyrosine phosphatase, non-receptor type 22
Modulation of T and B cell function
INS
Insulin
Deficient in type 1 diabetes
IL2RA
Interleukin-2 receptor, α
T lymphocyte function
IL10
Interleukin-10
Immunoregulation Inflammation
IL19
Interleukin 19
Immunity/inflammation
GLIS3
Gli-similar 3 protein
Pancreatic β cell generation Insulin gene expression Modulation of pancreatic β cell apoptosis
TRAF3IP2
TRAF3 interacting protein 2
Implicated in IL17 signaling Interacts with members of Rel/NF-κB transcription factor family
PLCG2
Phospholipase C, γ 2
Leukocyte signal transduction NK cell cytotoxicity
CCR5
CC-chemokine receptor 5
Major co-receptor for HIV entry into cells Immune cell recruitment
MYO1B
Myosin 1B
Cell membrane trafficking and dynamics
Pathogenesis of type 1 diabetes
Type 1 diabetes and the environment: infectious agents
Type 1 diabetes: the microbiome
Type 1 diabetes: vitamin D
Type 1 diabetes and insulin resistance
Type 1 diabetes: summary
Type 2 diabetes
Genetics of type 2 diabetes
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree