Chapter 7 Dana Wolf, DMD, MS and Evanthia Lalla, DDS, MS Periodontitis is a chronic inflammatory condition that is initiated by bacteria and results in the destruction of the tissues that anchor and support the teeth in the oral cavity. As reviewed in Chapter 6, it is well established that individuals with diabetes mellitus are at high risk for periodontitis. Moreover, the current thinking about the relationship between these two disease entities is that it is bi-directional [1]. Not only does diabetes mellitus negatively impact periodontal status, but periodontitis also adversely affects glycemic control and other outcomes in diabetic patients. In this chapter, we summarize the evidence that explores the potential effects of periodontitis on the diabetic state, including glycemic control, development of complications, and incident diabetes, and briefly describe possible mechanisms that lend biologic plausibility to this aspect of the association between the two diseases. A number of longitudinal observational studies have assessed whether periodontitis negatively affects glycemic outcomes in patients with diabetes. One of the first and most widely cited reports on glycemic control in particular was based on data collected from the Pima Indians of the Gila River Indian Community in Arizona [2]. The prevalence of type 2 diabetes mellitus is very high in this population. Taylor et al. used data collected from residents of this community for the National Institute of Diabetes, Digestive and Kidney Diseases (NIDDK) follow-up study and a related oral health study to test the hypothesis that severe periodontal disease confers risk for poor glycemic control. Subjects were dentate individuals aged 18–67 who had had a diagnosis of non-insulin-dependent diabetes and a glycosylated hemoglobin A1 (HbA1) less than 9% at baseline. Severe periodontitis at baseline was defined as either the presence of radiographic bone loss of 50% or more on one or more teeth, or a maximum of 6 mm of clinical attachment loss on one of six Ramfjord index teeth. Most subjects had one follow-up exam two years after baseline and 17 subjects had an additional follow-up exam at four years after baseline. The authors adjusted for multiple covariates and found that severe periodontitis at baseline was associated with increased risk for poor glycemic control (HbA1 greater than 9%) at follow-up. Other investigations have focused on the relationship between periodontitis and diabetic complications. In one of the first such studies, Thorstensson et al. divided type 1 diabetic subjects into 39 pairs of those with severe periodontitis (cases) and those with gingivitis or mild periodontitis (controls) [3]. Cases and controls were matched for age, sex, and diabetes duration. Over the median follow-up time of six years, cases were found to have a significantly higher prevalence of proteinuria and cardiovascular complications including stroke, transient ischemic attack, angina, myocardial infarct, and intermittent claudication compared to controls. Saremi et al. assessed how periodontal disease affected overall and cardiovascular disease (CVD) mortality in subjects of the Gila River Indian Community with type 2 diabetes [4]. Data from 628 subjects who were part of the NIDDK follow-up study and who had undergone at least one dental examination were analyzed. Over an average of 11 years of follow-up, 204 of the subjects died. Among individuals with severe periodontal disease, the death rate from ischemic heart disease was 2.3 times higher and the death rate from diabetic nephropathy was 8.5 times higher than among individuals with no, mild, or moderate periodontal disease after adjustment for age, sex, and duration of diabetes. In the same population of Pima Indians with type 2 diabetes, Shultis et al. investigated the effect of periodontitis on the development of overt nephropathy (defined as macroalbuminuria) and end-stage renal disease (ESRD) [5]. Of the 529 subjects included, 20% had no or mild periodontitis at baseline, 38% had moderate periodontitis, 22% had severe periodontitis, and 20% were fully edentulous. Over the follow-up period of up to 22 years (median of 9.4 years), 193 subjects developed macroalbuminuria and 68 developed ESRD. After adjusting for various confounders, the incidence of these diabetic complications was higher in subjects who had severe periodontitis or who were edentulous at baseline. The authors concluded that severe periodontitis and edentulism predict the development of overt nephropathy and ESRD in patients with type 2 diabetes. Li et al. assessed the association between tooth loss and subsequent mortality in subjects with type 2 diabetes [6]. Ten thousand eight hundred and fifty-eight subjects who were part of the large cohort study Action in Diabetes and Vascular Disease: Preterax and Diamicron Modified-Release Controlled Evaluation (ADVANCE) trial counted the number of teeth in their mouths and reported the number of days their gums had bled over the preceding year. After controlling for several potential confounders, it was found that both the presence of fewer teeth and complete edentulism were significantly associated with an increased risk of death from all causes, CVD and non-CVD, over a five-year period. It is important to note that although tooth loss can occur for different reasons, periodontal disease is a major cause of tooth loss in adults. There have been two studies to date which have examined whether periodontitis is associated with the development of type 2 diabetes. Demmer et al. used data from the first National Health and Nutrition Examination Survey (NHANES I) and its Epidemiologic Follow-up Study (NHEFS) to test the hypothesis that baseline periodontal status could predict incident type 2 diabetes [7]. A total of 9,296 non-diabetic subjects who had completed a baseline dental examination and had at least one follow-up evaluation were included in the analysis. Dentate subjects were classified into categories of periodontal disease severity based on the Periodontal Index (a clinical index in which higher scores represent increased levels of disease). Over a follow-up period of 17 ± 4 years, 817 cases of incident diabetes were found. The adjusted odds ratios for incident diabetes in periodontal index categories 1 and 2 were not elevated, whereas those in categories 3, 4, and 5 were 2.26, 1.71, and 1.50, respectively. The authors speculated that the substantially elevated odds at intermediate periodontal index levels might have been a consequence of the fact that examiners assigned the lesser of two periodontal index values when periodontal findings were equivocal. The authors concluded that periodontal disease at baseline could predict incident type 2 diabetes. Limitations of this study were the use of an outdated clinical index to assess periodontal status and the unavailability of laboratory measures to exclude undiagnosed diabetes at baseline. In contrast to the findings from the NHANES I data, a large prospective study out of Japan failed to demonstrate an association between periodontitis and incident diabetes [8]. Ide et al. analyzed 5,848 civil service officers aged 30–59 who were diabetes-free (verified with a negative fasting plasma glucose test result at baseline). Periodontal status was defined again with a rather imprecise method, using the Community Periodontal Index of Treatment Needs (CPITN), an index which examines only certain teeth in the mouth [9]. Based on a positive fasting plasma glucose test result, 287 new cases of diabetes were identified over a mean follow-up period of 6.5 years (range: 2–7 years). There was a statistically significantly increased risk of developing diabetes among those with moderate and severe periodontitis at baseline, but the association did not hold after adjustment for various confounders. There are other reports in the literature on diabetes-free subjects which have examined the impact of periodontal disease on longitudinal changes in HbA1c [10] and the development of glucose intolerance [11], but enrolled subjects did not develop frank diabetes over the course of these trials. Several intervention studies aimed at testing the hypothesis that periodontal therapy may result in improved glycemic control in patients with diabetes have been published since the 1990s. Unfortunately, many of the reports are based on uncontrolled studies with small sample sizes and short duration of post-treatment follow-up. The two most recent systematic reviews which analyzed data from available randomized controlled trials (RCT) and controlled clinical trials (CCT) were published in 2010 [12, 13]. Table 7.1 summarizes the studies included in the meta-analyses of these two reviews. Table 7.1 Intervention studies testing the impact of periodontal therapy on glycemic control in diabetes included in the two most recent meta-analyses [12, 13]. 1 Studies included in both meta-analyses. CCT = controlled clinical trial; RCT = randomized control trial; DM = diabetes mellitus; T = treatment group; C = control group; F/u = follow-up; OHI = oral hygiene instruction; SRP = scaling and root planing; HbA1c = hemoglobin A1c; FPG = fasting plasma glucose. The first, by Teeuw et al. [13], sought publications that met the following inclusion criteria: The authors identified five studies that met the inclusion criteria [14–18], with a total of 382 patients with type 2 diabetes (199 in the intervention groups and 183 in the control groups). Sample sizes of the individual studies ranged from 44 to 165 subjects. The intervention groups received mechanical periodontal therapy (scaling and root planing) with [14–16] or without [17, 18] adjunctive local or systemic antibiotics. All five studies identified assessed changes in HbA1c after periodontal therapy. Two of the five studies showed statistically significant improvement in glycemic control in cases (as measured by a decrease in HbA1c levels) compared to controls [17, 18]. The other three studies showed a non-statistically significant decrease in HbA1c in the treatment group [14–16]. Three of the five studies also reported changes in fasting plasma glucose (FPG) after therapy [14, 15, 17]. Two of the studies showed a non-statistically significant decrease in FPG after treatment [15, 17], while one showed a non-significant increase [14]. The data from the individual studies were pooled for two separate meta-analyses using either change in HbA1c (all five studies, 382 subjects) or FPG (3 studies, 145 subjects) as the main outcome variable. Mean changes in HbA1c in the treatment groups ranged from -1.90 to -0.14% while in the control groups they ranged from -0.40 to +0.31%. The weighted mean difference (WMD) in HbA1c between test and control before and after periodontal therapy was -0.40% (95% CI: -0.04 to -0.77%) indicating a statistically significant HbA1c reduction in the treatment group. Mean changes in FPG ranged from -3.63 to +19.00 mg/dl for the treatment groups versus -3.00 to +1.22 mg/dl for the controls. The WMD of mean FPG differences between test and control groups was a non-significant increase by 2.30 mg/dl (95% CI: -13.64 to +18.24 mg/dl).
The influence of periodontal disease on glycemic control in diabetes
Longitudinal observational studies
Treatment studies
Publication
Study design
Intervention
Main outcome variables and results
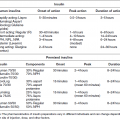
Stewart et al. 2001 [18]
CCT; subjects with type 2 DM;
T – n = 36
C – n = 36
F/u – 3 months
T: SRP, extraction of hopeless teeth
C: None, dental status of controls unknown
HbA1c
T: ∆ -0.8 (P = 0.0001)
C: ∆ -1.9 (P = 0.02)
Statistically significant difference in change between T and C
Kiran et al. 2005 [17] 1
RCT, subjects with type 2 DM;
T – n = 22
C – n = 22
F/u – 3 months
T: OHI, SRP
C: None
HbA1c
T: ∆ -0.86; C: ∆ +0.31
Statistically significant difference in change between T and C
FPG
T: ∆ -3.96; C: ∆ +1.22
Statistically significant difference in change between T and C
Promsudthi et al. 2005 [15]
CCT; subjects with type 2 DM;
T – n = 27
C – n = 36
F/u – 3 months
T: OHI, SRP, systemic doxycycline
C: None
HbA1c
T: ∆ -0.19; C: ∆ +0.12
Non-statistically significant difference in change between T and C
FPG
T: ∆ -3.63; C: ∆ +0.2
Non-statistically significant difference in change between T and C
Jones et al. 2007 [16]1
RCT; DM type not reported
T – n = 82
C – n = 83
F/u – 4 months
T: SRP, systemic, chlorhexidine gluconate rinses
C: “usual dental care”: participants did not alter dental care routine. Some used dental services, others did not.
HbA1c
T: ∆ -0.65
C: ∆ -0.49
Non-statistically significant difference in change between T and C
Yun et al. 2007 [23]
RCT; subjects with type 2 DM;
T – n = 23
C – n = 23
F/u – 16 weeks
T: SRP, systemic doxycycline
C: systemic doxycycline
HbA1c
T: ∆ -0.77
C: ∆ -0.58
Between group (T and C) analysis not reported
Katigiri et al. 2009 [14]
RCT; subjects with type 2 DM;
T – n = 32
C – n = 17
F/u – 1, 3, 6 months
T: OHI, SRP, locally administered minocycline ointment
C: OHI
HbA1c
T: Statistically significant reduction at 1 month but not at 3 or 6 months
C: No statistically significant change at any time point
FPG
No statistically significant change at any time point
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree