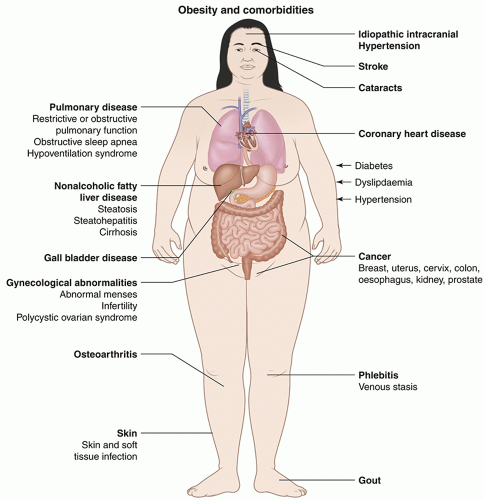
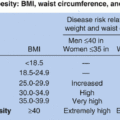
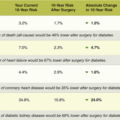
Most asymptomatic adults do not need routine screening for
CAD/
CVD. However, as described in the 2013 American Heart Association/American College of Cardiology/The Obesity Society (
AHA/
ACC/TOS) Guideline for the Management of Overweight and Obesity in Adults, patients with obesity should undergo risk assessment for cardiovascular risk factors, including lipids, blood pressure measurement, and fasting blood glucose. A waist circumference measurement is recommended for individuals with
BMI 25 to 34.9 kg/m
2 to provide additional information on risk. It is unnecessary to measure waist circumference in patients with
BMI ≥35 kg/m
2 because the waist circumference will likely be elevated and will add no additional risk information. Cut points of (≥88 cm [≥35 inches] for women and ≥102 cm [≥ 40 inches] for men) are indicative of
increased cardiometabolic risk.
19 While there is little evidence to support routine exercise testing in asymptomatic adults,
ACC/
AHA guidelines suggest that exercise electrocardiogram (
ECG) testing may be of benefit in patients with multiple risk factors for
CAD in the following situations: as a guide to risk reduction therapy; men ≥45 years of age and women ≥55 years of age who are sedentary and planning to begin a vigorous exercise program; or patients who are involved in occupations linked to public safety.
20 If stress testing reveals an abnormal result, referral to a cardiologist and aggressive treatment is needed with secondary prevention measures (e.g., statin therapy, aspirin, tight blood pressure control). The use of computed tomography (CT) to obtain a coronary artery calcium (
CAC) score may be useful to further risk stratify and to guide a discussion regarding statin therapy in adults at borderline (5% to < 7.5%) or intermediate (7.5% to < 20%) 10-year atherosclerotic cardiovascular disease (
ASCVD) risk.
Chapter 3 contains an in-depth discussion of how cardiovascular risk factors can be used to risk stratify patients with obesity.