Shwachman–Diamond syndrome (SDS) is an inherited neutropenia syndrome associated with a significant risk of aplastic anemia and malignant transformation. Multiple additional organ systems, including the pancreas, liver, and skeletal and central nervous systems, are affected. Mutations in the Shwachman-Bodian-Diamond syndrome (SBDS) gene are present in most patients. There is growing evidence that SBDS functions in ribosomal biogenesis and other cellular processes. This article summarizes the clinical phenotype of SDS, diagnostic and treatment approaches, and novel advances in our understanding of the molecular pathophysiology of this disease.
Key points
- •
Many exciting advances in our understanding of SDS have occurred in the past few years.
- •
Our understanding of the natural history and spectrum of disease, diagnosis, and therapy remain limited.
- •
Ongoing basic and clinical investigations on larger numbers of patients are crucial to better tie together our evolving comprehension of molecular function and clinical manifestations of this multiorgan disease to affect the treatment of patients with this rare disorder.
Introduction
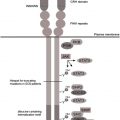
Shwachman–Diamond syndrome (SDS) is a rare autosomal recessive multisystem disorder characterized by congenital anomalies, exocrine pancreatic dysfunction, bone marrow failure, and predisposition to myelodysplasia (MDS) and leukemia, particularly acute myeloid leukemia (AML). In addition, growth, heart, liver, central nervous system, skeletal system, and the immune system may also be affected. The incidence of SDS is indirectly approximated at 1:77,000. Around 90% of patients with SDS harbor mutations in the Shwachman-Bodian-Diamond syndrome ( SBDS ) gene located on chromosome 7q11. SBDS encodes a novel protein involved in ribosomal maturation and implicated in additional functions, such as cell proliferation and mitosis, as well as in the stromal microenvironment.
Although the majority (90%) of patients clinically diagnosed with SDS harbor mutations in the SBDS gene, phenotype varies widely between patients and even within the same individual over time, posing challenges for diagnosis and treatment. Owing to the rarity of this disorder, the natural history of SDS remains poorly defined and controlled clinical studies to direct therapy are lacking. Thus, current management is largely based on case series and consensus reports. Longitudinal clinical studies are needed to define the diagnostic criteria, phenotypic range, and molecular pathophysiology of SDS to identify risk factors for medical complications and guide therapeutic interventions.
This review highlights recent advances in the understanding of the clinical manifestations and molecular pathogenesis of SDS. The reader is referred to prior excellent reviews for a general overview of SDS.
Introduction
Shwachman–Diamond syndrome (SDS) is a rare autosomal recessive multisystem disorder characterized by congenital anomalies, exocrine pancreatic dysfunction, bone marrow failure, and predisposition to myelodysplasia (MDS) and leukemia, particularly acute myeloid leukemia (AML). In addition, growth, heart, liver, central nervous system, skeletal system, and the immune system may also be affected. The incidence of SDS is indirectly approximated at 1:77,000. Around 90% of patients with SDS harbor mutations in the Shwachman-Bodian-Diamond syndrome ( SBDS ) gene located on chromosome 7q11. SBDS encodes a novel protein involved in ribosomal maturation and implicated in additional functions, such as cell proliferation and mitosis, as well as in the stromal microenvironment.
Although the majority (90%) of patients clinically diagnosed with SDS harbor mutations in the SBDS gene, phenotype varies widely between patients and even within the same individual over time, posing challenges for diagnosis and treatment. Owing to the rarity of this disorder, the natural history of SDS remains poorly defined and controlled clinical studies to direct therapy are lacking. Thus, current management is largely based on case series and consensus reports. Longitudinal clinical studies are needed to define the diagnostic criteria, phenotypic range, and molecular pathophysiology of SDS to identify risk factors for medical complications and guide therapeutic interventions.
This review highlights recent advances in the understanding of the clinical manifestations and molecular pathogenesis of SDS. The reader is referred to prior excellent reviews for a general overview of SDS.
Clinical manifestations
Owing to the rarity of this syndrome, our understanding of the full spectrum of clinical disease in SDS remains incomplete. The current knowledge was summarized recently in an updated clinical consensus guideline. The classical clinical scenario describing SDS includes exocrine pancreatic dysfunction and bone marrow failure ( Box 1 ). Skeletal abnormalities may include metaphyseal dysplasia, flared ribs, thoracic dystrophies, and osteopenia. Neurocognitive deficits have been described. While the exocrine pancreatic dysfunction in SDS is well described, a distinctive abnormal hepatic phenotype in these patients has also been reported. Progression and evolution of bone marrow disease remains a major source of morbidity and mortality in these patients. Registries and clinically annotated biosample repositories for SDS are poised to expand our knowledge of this disease and its myriad of developmental effects through systematic and longitudinal studies leading to more disease-specific interventions.
Biallelic mutations in SBDS or clinical Shwachman–Diamond syndrome: one criteria from Category I and II
Category I
Low levels of trypsinogen (age <3 years) or low pancreatic isoamylase levels (age >3 years)
Low levels of fecal elastase
Supportive features:
Pancreatic lipomatosis
Elevated 72-hour fecal fat excretion and absence of intestinal pathologic condition
Category II
Hypoproductive cytopenias
Neutropenia (absolute neutrophil count <1500)
Anemia or idiopathic macrocytosis
Thrombocytopenia (<150,000)
Bone marrow examination with any of the following:
Myelodysplasia
Leukemia
Myelodysplasia syndrome
Hypocellularity for age
Cytogenetic abnormalities
Supporting features
First-degree or second-degree blood relative with Shwachman–Diamond syndrome
Personal history of
Congenital skeletal abnormalities consistent with chondrodysplasia or a congenital thoracic dystrophy
Height 3% or less, of unclear cause
Deficiency in 2 or more fat-soluble vitamins (A, 25-OHD, and E).
Hematologic manifestations
Patients with SDS are at risk for cytopenias secondary to marrow failure. Neutropenia is reported in 88% to 100% of patients and can be either intermittent or persistent, with variable severity. Anemia and thrombocytopenia have also been reported in most patients, although both are often intermittent or asymptomatic. Elevated hemoglobin F levels can also be seen in a subset of patients. Severe aplastic anemia with trilineage cytopenias may also develop in a subset of patients. The French Severe Chronic Neutropenia Registry recently evaluated the hematologic complications in their cohort of 102 genetically diagnosed patients with SDS and found 41 patients (40%) with hematologic complications including transient severe cytopenias. Of these patients, 21 (20.6%) presented with definitive persistent cytopenias (anemia with hemoglobin levels <7 g/dL or profound thrombocytopenia with platelets <20 g/L), in 9 of whom the condition was classified as malignant and in another 9 as nonmalignant, and in 3, the condition progressed from nonmalignant to malignant. Prognostic factors reported with severe cytopenias in this cohort included early age at diagnosis and hematologic parameters.
Reports of progression to MDS or AML in patients with SDS have varied. Previously, the Severe Chronic Neutropenia International Registry (SCNIR) had reported a rate of 1% per year of MDS or AML in patients with SDS, with an overall incidence of 8.1% in 37 patients with SDS in 10 years. The French registry reported a rate of transformation to MDS or AML of 18.8% at 20 years and 36.1% at 30 years in a cohort of 55 patients with SDS. Some of this discrepancy arises from differences in the definition of MDS. More recently, the Canadian Inherited Bone Marrow Failure Study (CIBMFS) registry reported a cumulative transformation rate of 18% in 34 patients with SDS. This result is in contrast to other recent reports from the NIH registry (17 patients) and the Israeli registry (3 patients) in which no patient developed MDS or AML. Although it is difficult to draw conclusions from such small numbers of patients, this discrepancy may be partly due to the age of these cohorts. The median age of transformation for patients with SDS was 19.1 years in the French group and 20 years in the Canadian cohort, whereas the NIH and Israeli cohorts had median ages of 14 and 4 years, respectively, at the time of report. Transformation rates reported by the SCNIR for patients with severe congenital neutropenia (SCN) are 11.8% at 10 years, whereas the rates for fanconi anemia (FA) and dyskeratosis congenita (DC) by the age of 50 years as reported by the NIH are 40% and 30%, respectively, for myelodysplasia (MDS), and 10% for both for AML. The Diamond Blackfan Anemia Registry reports that patients with diamond blackfan anemia (DBA) are less likely to transform with cumulative incidence of AML of 5% by the age of 46 years, with incidence increasing only after the age of 40 years. Together, these data suggest that the risk of malignant transformation in patients with SDS is significant, especially with respect to some of the other inherited marrow failure syndromes, but occurs with less frequency and longer latency than in patients with Fanconi anemia. Published reports of solid tumors in patients with SDS are rare thus far. There are only 2 cases in the literature, one of bilateral breast cancer in a 30-year-old woman with SDS and another of dermatofibrosarcoma in a 20-year old-woman, which had been present and slowly growing for approximately 3 years at diagnosis.
It has long been known that patients with SDS may develop characteristic cytogenetic clones in the absence of overt MDS or AML and that these abnormalities may persist over time without progression or malignant evolution. Recent reports suggest that a common cytogenetic abnormality seen in patients with SDS, del(20)(q11), is not associated with a high risk of malignant transformation. Another characteristic cytogenetic anomaly in SDS that can come and go over years of time without progression to MDS/AML is isochromosome i(7)(q10). Maserati and colleagues reported on clonal changes in 22 new patients with SDS and 14 cases of follow-up of previously reported cases. Of the 36 cases, 16 demonstrated clonal changes, all of which involved either chromosome 7 or 20. Chromosome 7 abnormalities included isochromosome [i(7)(q10) ( n = 10)], [add(7)(p?) ( n = 1)], and a long arm deletion [del(7)(q22q23) (n = 1)]. All 6 clones involving chromosome 20 abnormalities initially involved del(20)(q11); however, 2 evolved into subclones, which had acquired additional cytogenetic abnormalities. All 5 patients with del(20)(q11) demonstrated loss of the common MDR established in patients with MDS who did not have SDS. Over the course of the study (range of follow-up being between 1 month and 9 years), 8 patients had stable clones, 4 demonstrated increasing clonal involvement, and 1 had diminished clonal involvement, irrespective of the initial type or size of clonal abnormality. Despite these additional cytogenetic abnormalities and the loss of a region commonly deleted in MDS/AML, the only patient to progress to MDS or require bone marrow transplantation carried the add(7)(p?) abnormality, and no patient progressed to AML. In addition, the appearance of clonal abnormalities seemed to be age related, with increased frequency of clonal changes seen with increasing age.
Furthermore, Crescenzi and colleagues evaluated the bone marrows of 2 patients with SDS and del(20)(q11) who had been followed up over a 6- to 7-year period without development of MDS/AML. In these patients, there was no acquisition of additional cytogenetic changes associated with MDS/AML despite increasing clonal population in 1 patient. By fluorescence in situ hybridization, del(20)(q11) was seen to be present in totipotent hematopoietic stem cells as well as downstream myeloid and lymphoid lineages, indicating preservation of the capacity to differentiate even in the face of this cytogenetic abnormality.
Historically, clinical observations have also demonstrated an increased frequency of infection beyond that attributable to simple neutropenia in patients with SDS. Sepsis is one of the most common fatal infections in SDS, often associated with neutropenia. However, patients with SDS also have susceptibility to recurrent bacterial, viral, and fungal infections. Dror and colleagues prospectively studied immune functions in this population. B-cell defects (less number of circulating B cell, low levels of IgG and IgG subclasses, and deficient antibody production) and T-cell defects (low Cd3 + /CD4 + cell subpopulations and decreased T-lymphocyte proliferation) were described in most patients with SDS studied. Universally abnormal neutrophil chemotaxis was also reported as described previously. However, in contrast to other neutrophil chemotaxis disorders, patients with SDS retain the ability to form purulent abscesses and empyema.
Gastrointestinal manifestations
Exocrine pancreatic dysfunction is a classic feature of SDS resulting from severe depletion of pancreatic acinar cells. The majority (>90%) of patients with SDS are diagnosed with pancreatic dysfunction in the first year of life, often in the first 6 months. Clinical manifestations range widely from severe dysfunction with significant nutrient malabsorption, steatorrhea, and resultant failure to thrive, to completely asymptomatic. Despite these findings, clinical symptoms in many patients with SDS spontaneously improve with age for reasons that remain unclear. In as many as 50% of patients, pancreatic enzyme supplementation can be stopped by the age 4 years based on evidence of normal fat absorption, although enzyme secretion deficits remain. A recent study of parotid acinar function in 16 patients with SDS compared with 13 healthy controls and 13 patients with cystic fibrosis or fibrosing pancreatitis found parotid acinar dysfunction. Both serum pancreatic and parotid isoamylase levels were lower in patients with SDS than in healthy controls, whereas pancreatic isoamylase levels were lower in other disease controls than in normal controls. Secreted parotid amylase levels were also lower in patients with SDS than in healthy controls, whereas the levels in disease controls were comparable to those in normal controls. These findings suggest a more generalized defect in acinar cell function in patients with SDS. In addition, a recent study by Shah and colleagues of histologic changes in gastrointestinal mucosal biopsies of 15 symptomatic patients with genetically confirmed SDS demonstrated that more than 50% showed varying degrees of duodenal inflammation by histology. This result suggests that there may be an enteropathic component in addition to the pancreatic exocrine failure contributing to the symptoms in some patients with SDS.
Although the pancreatic manifestations of SDS are well known, patients with SDS often have other gastrointestinal involvement, most notably in the liver. A recent longitudinal study of 12 Finnish patients with SDS further characterized the hepatic manifestations of SDS, confirming previous reports of elevated levels of transaminases and hepatomegaly in younger patients with SDS that resolve with age. In addition, this study found that a majority (58%) of patients had elevated levels of bile acids. Of these patients, 3 had longitudinal bile acid measurements, and all had repeatedly elevated levels, although intermittently in 2, raising the concern for persistent cholestasis. Longitudinal examination of hepatic imaging revealed hepatomegaly only in young patients (younger than 3 years). Interestingly, all 3 patients older than 30 years had developed hepatic microcysts that were readily apparent on imaging studies.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree