Abstract
The bone marrow (BM) extracellular matrix (ECM) is often inconspicuous, and hence neglected in a normal BM trephine biopsy (BMB), but it has essential functions in the homeostasis of bone and the maturation of haematopoiesis (see Chapter 2). The ECM surrounds the bony trabeculae, which are covered by osteoblasts and osteoclasts, the latter an essential component of the stem cell niche, and embeds the maturing haematopoietic cells. It is rich in hyaluronic acid, type III collagen (reticulin), fibronectin and laminin.
Introduction
The bone marrow (BM) extracellular matrix (ECM) is often inconspicuous, and hence neglected in a normal BM trephine biopsy (BMB), but it has essential functions in the homeostasis of bone and the maturation of haematopoiesis (see Chapter 2). The ECM surrounds the bony trabeculae, which are covered by osteoblasts and osteoclasts, the latter an essential component of the stem cell niche, and embeds the maturing haematopoietic cells. It is rich in hyaluronic acid, type III collagen (reticulin), fibronectin and laminin.
Marrow fibre production by stromal cells is a balance between collagen production and resorption, regulated by cytokines such as TGF-beta, FGF and PDGF. These cytokines derive from both haematopoietic and stromal cells [1]. The stromal cells in the BM include mesenchymal stem cells in the vascular niche, which can differentiate into adipocytes, chondrocytes and osteoblasts, depending on ECM composition, structure and oxygenation. Disturbance of cytokine balance or oxygenation can directly or indirectly lead to extensive necrosis, fibrosis, bone resorption or osteosclerosis. Stromal changes in a BMB can therefore be seen in a wide spectrum of reactive and neoplastic diseases, both systemic and localized to the marrow. In this chapter we will focus on necrosis, extracellular depositions and fibrosis, and the distinction from artefacts.
Necrosis, Oedema and Haemorrhage
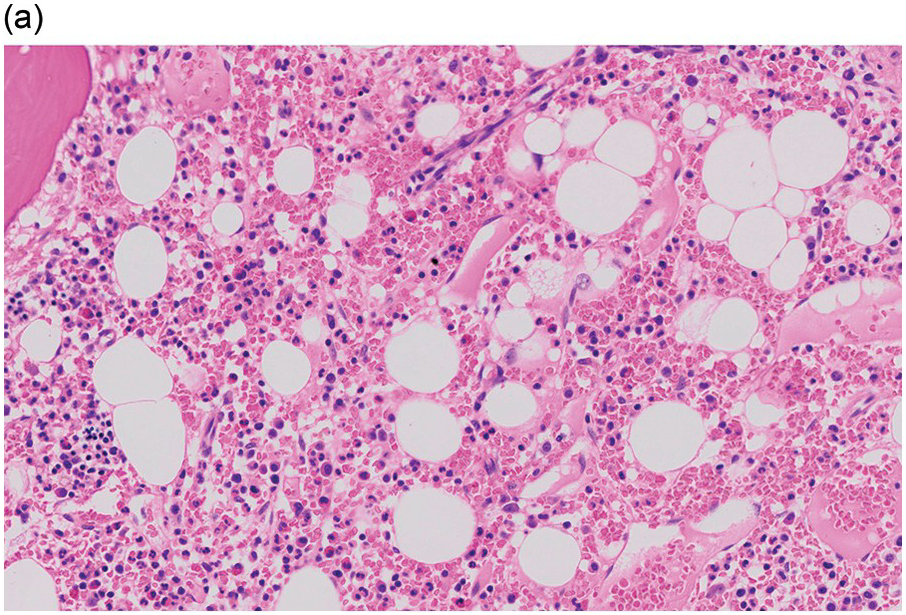
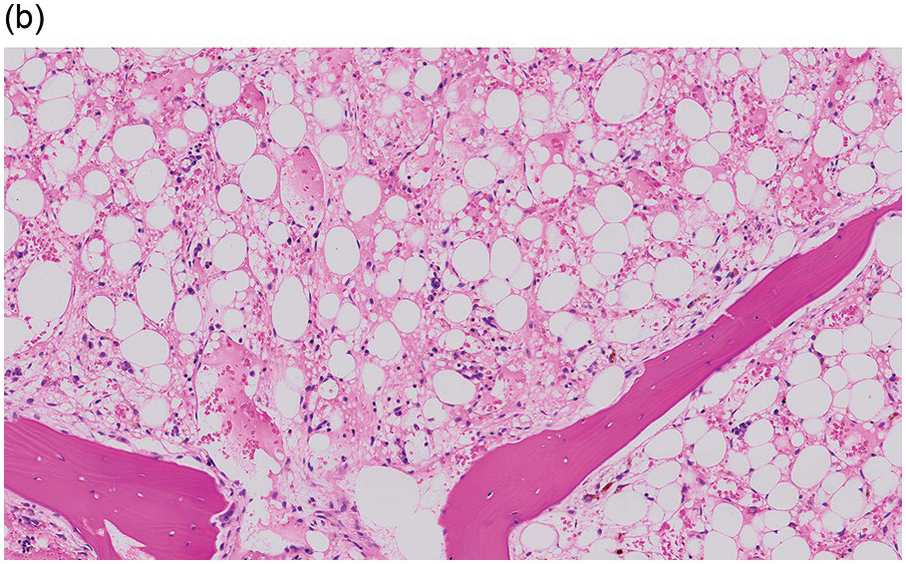
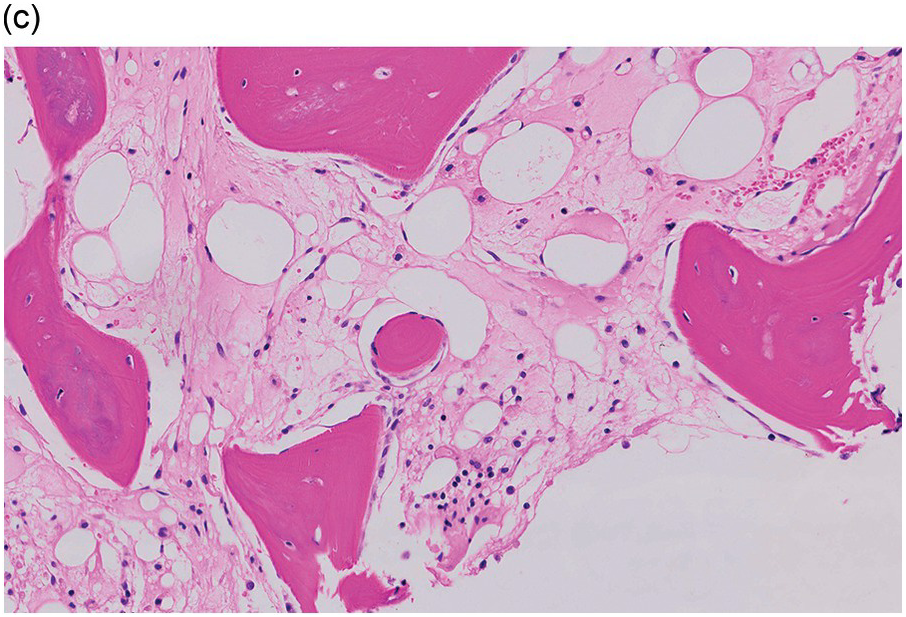
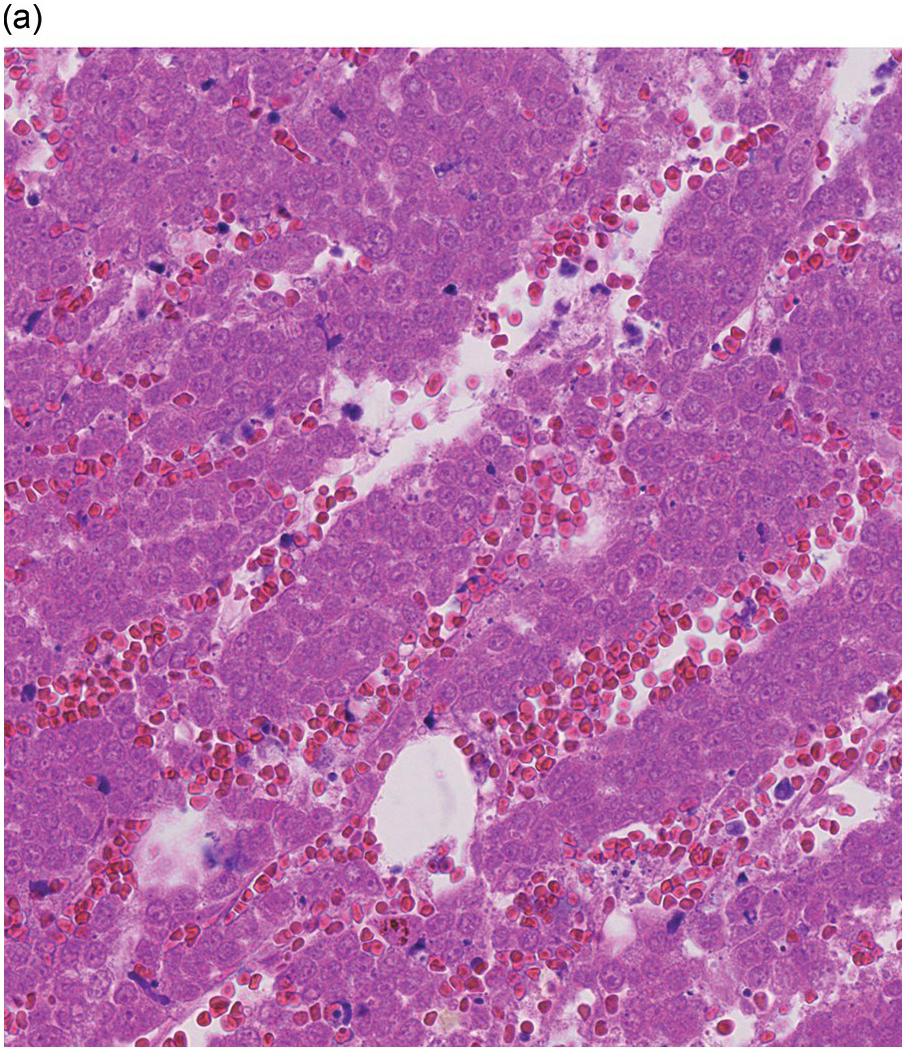
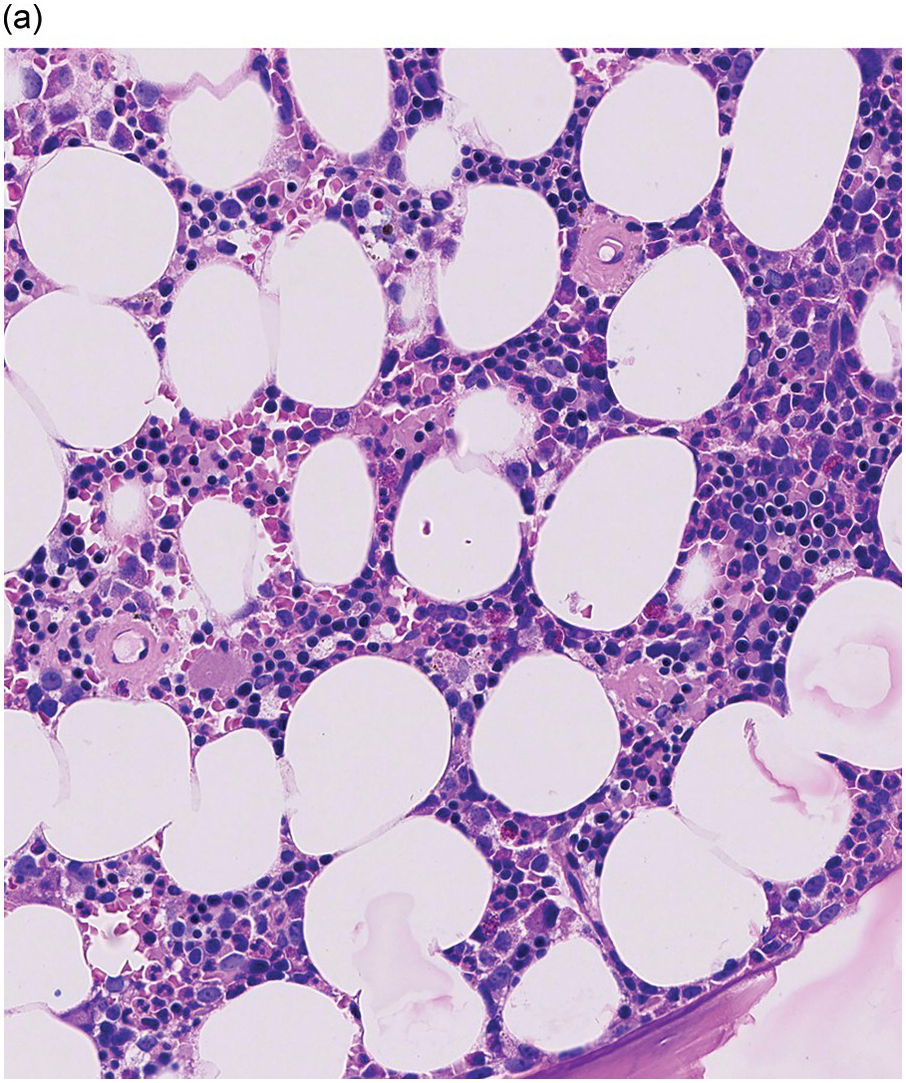
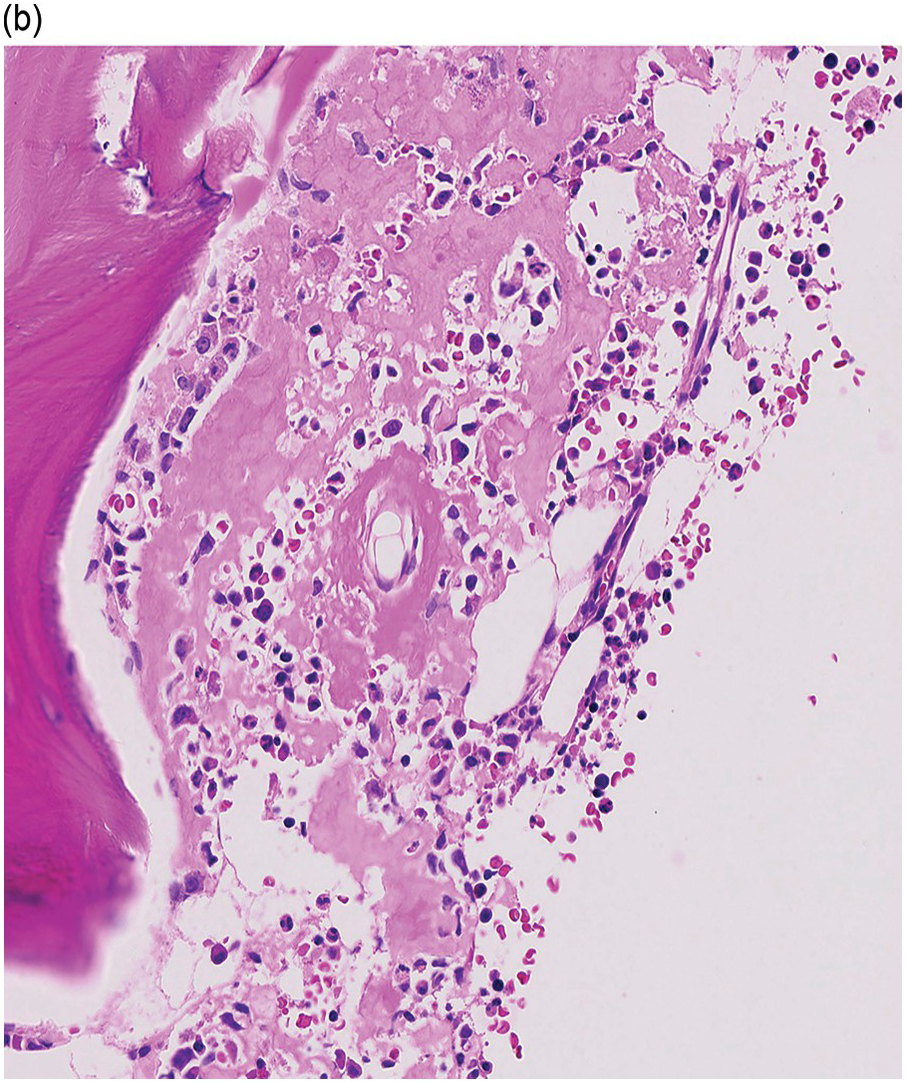
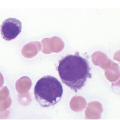
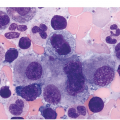
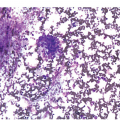
The pathogenesis of oedema, haemorrhage and necrosis in the BM is not known exactly but can be assumed to result from vascular damage. A severely compromised circulation of oxygen and nutrients to BM or tumour cells, caused by damage to the sinusoidal endothelium in chemotherapy or radiotherapy [2], inflammation, or vascular occlusion as in sickle cell crisis [3] can eventually lead to confluent areas of coagulation necrosis. While oedema and haemorrhage present in an H&E stain as a pale eosinophilic matrix with dispersed red blood cells and vital haematopoietic cells with staining nuclei (Figure 3.1a, b, c), coagulation necrosis appears as large areas of homogeneous, intense eosinophilic colouration, frequently with remnants of cellular outlines (ghost cells) or nuclear debris (Figure 3.2a). Structural elements such as blood vessels and fibres are often lost in necrosis and the bony trabeculae can be avital as well, resulting in loss of nuclear staining of osteoclasts, osteoblasts and osteocytes (so-called empty lacunae). The underlying causes can be numerous, both malignant and benign (Table 3.1) [7]. The microvascular damage in chemotherapy and radiotherapy leads to interstitial oedema and haemorrhage, while suppression of haematopoietic cell proliferation and cell death lead to marrow hypoplasia (Figure 3.2b, c). The acute phase of oedema, haemorrhage and cell death can result in fibrinoid necrosis with a more granular eosinophilic appearing matrix than in coagulative necrosis. Cellular reconstitution of necrotic marrow is accompanied by increased numbers of foamy macrophages, regeneration of fat cells, which may be multivacuolated, and often fibrosis. Bone marrow necrosis clinically presents with fever, bone pain, fatigue and jaundice, while laboratory findings are pancytopaenia and high LDH and alkaline phosphatase levels [5, 6]. Fat embolism is a rare complication of extensive BM necrosis [3].
Table 3.1 Diseases that can present with localized or general necrosis, serous atrophy or fibrosis of the bone marrow.
| Serous atrophy | Necrosis | Fibrosis | ||
|---|---|---|---|---|
| Infections | Parvovirus Visceral leishmaniasis HIV, AIDS Tuberculosis Other granulomatous infections | X | X X X X X | X X X X |
| Autoimmune disease | Systemic lupus erythematosus Sjögren syndrome Systemic sclerosis Primary autoimmune myelofibrosis | X | X X X X X | |
| Myeloid neoplasm | Chronic myeloid leukaemia Myeloproliferative neoplasm Myelodysplastic syndrome Acute myeloid leukaemia Acute megakaryoblastic leukaemia MDS/MPN overlap neoplasia Systemic mastocytosis | X | X X X | X X X X X X X |
| Lymphoproliferative disease | Acute lymphoblastic leukaemia Hairy cell leukaemia Non-Hodgkin lymphoma Plasma cell myeloma Hodgkin lymphoma | X X X X | X X X X X | X X X X X |
| Solid tumours | Carcinoma metastasis | X | X | X |
| Genetic | Osteopetrosis Grey platelet syndrome | X X | ||
| Metabolic disturbance | Hyperparathyroidism Osteomalacia Diabetes mellitus (type I) Hypothyroidism Liver cirrhosis | X X X | X X X | |
| Malnutrition, deficiencies | Alcoholism Anorexia nervosa Iron deficiency Vitamin D deficiency | X X X | X | |
| Drugs | Haematopoietic growth factor(mimics) Interferons Cytotoxic agents Tyrosine kinase inhibitor | X X | X X X X | X X |
| Vascular, ischaemic | Pulmonary arterial hypertension Sickle cell disease Haemolytic uremic syndrome Antiphospholipid syndrome Diffuse intravascular coagulation Previous biopsy | X X X X X | X X X |
Epidemiology
The reported frequency of severe necrosis, involving more than 50% of the BMB, is less than 1 to 10% of trephines, depending on the patient selection [4–7]. Solid or haematological malignancies are the most common underlying diseases.
Serous Atrophy or Gelatinous Transformation
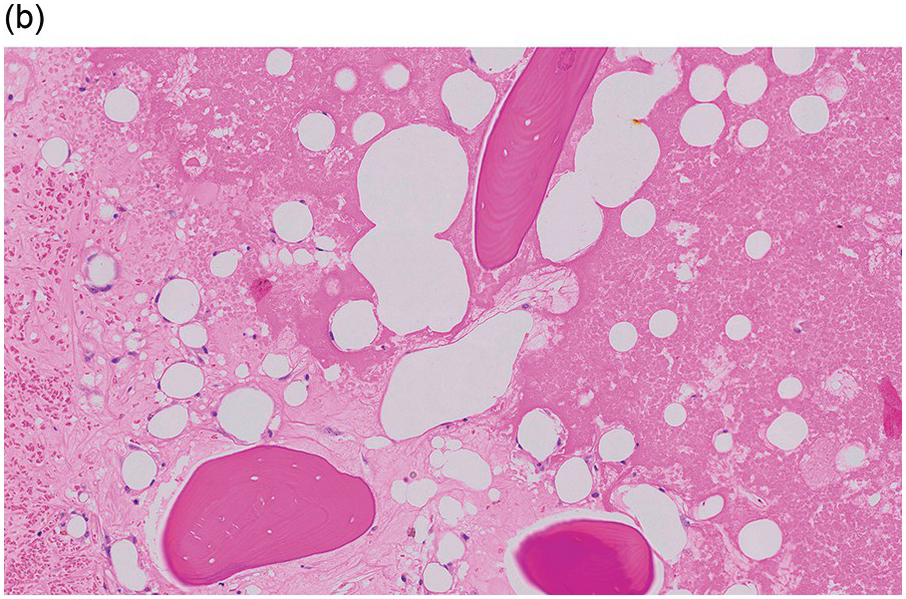
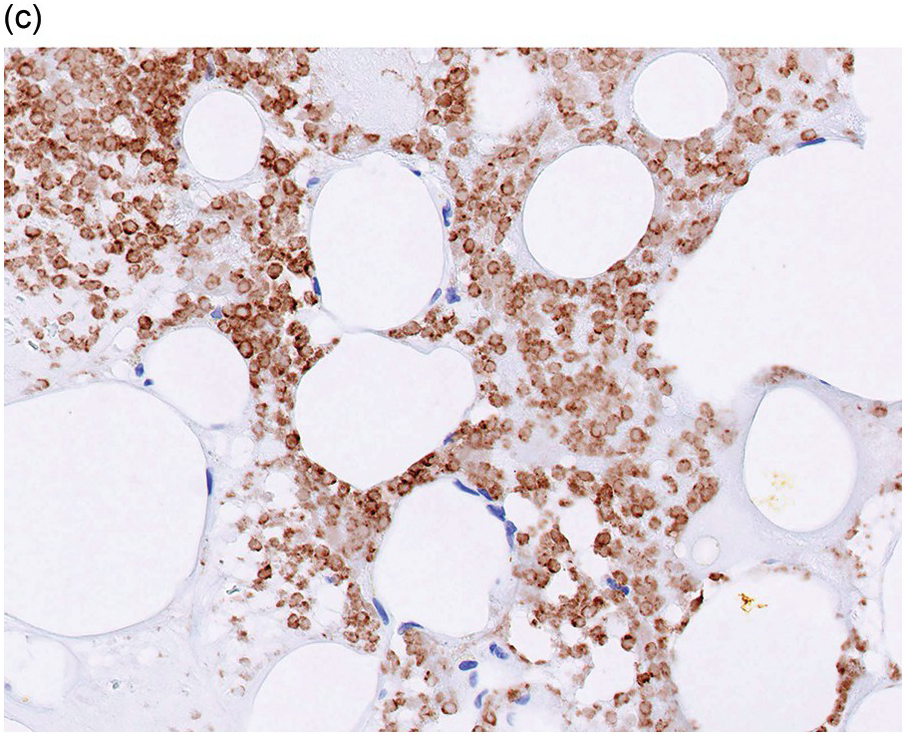
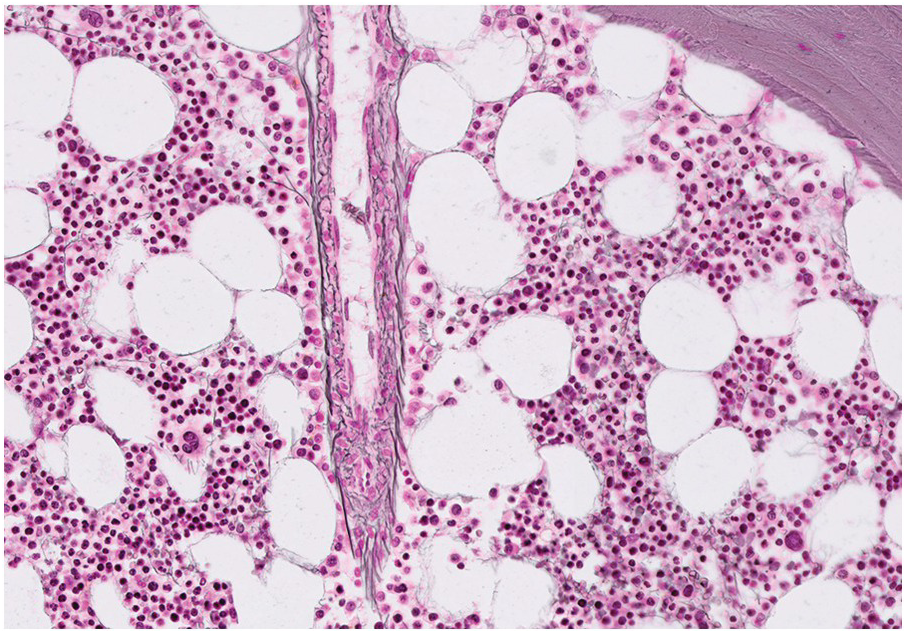
Serous atrophy, also referred to as gelatinous transformation of the BM, consists of focal replacement of fat and haematopoietic cells by eosinophilic amorphous extracellular material consisting of acid mucopolysaccharides, rich in hyaluronic acid, that can be demonstrated by Alcian blue stain at pH 2.5 (Figure 3.3a–c) [8]. Overall cellularity is decreased, while fat cells show a decrease in size but not in number [9]. Serous atrophy is an indicator of serious generalized, usually chronic, illness and is probably due to a systemic or local decrease of nutrient supply in diseases such as malnutrition, vitamin deficiency, anorexia nervosa, alcoholism, gastrointestinal dysfunction, malignancy (lymphoma, plasma cell myeloma or metastatic carcinoma), systemic lupus erythematosus (SLE), chronic infections (tuberculosis, AIDS), diabetes mellitus, chronic heart failure and treatment of myeloid neoplasms with chemotherapy or imatinib (Table 3.1) [8, 10, 11]. Clinical symptoms are anaemia, weight loss and cachexia.
Figure 3.3 Serous atrophy with a hypocellular homogeneous matrix (a). Alcian blue stains the mucopolysaccharides intense blue (b). S100 immunohistochemistry highlights the membranes of the atrophic fat cells (c).
Epidemiology
While the reported frequency in an Indian series is almost 5%, mainly in children [10], in Western reports serous atrophy is seen in less than 0.5% of trephine biopsies, mainly in young adults [8]. Bone marrow core biopsy of patients with anorexia nervosa shows serous atrophy in half of the cases [9].
Differential Diagnosis
In contrast to the hypoplasia that is common in serous atrophy, oedema shows eosinophilic fluid accumulation in the ECM between preexisting haematopoietic cells of normal cellularity and distribution. The appearance of amyloid deposition is described below.
Amyloid Deposition
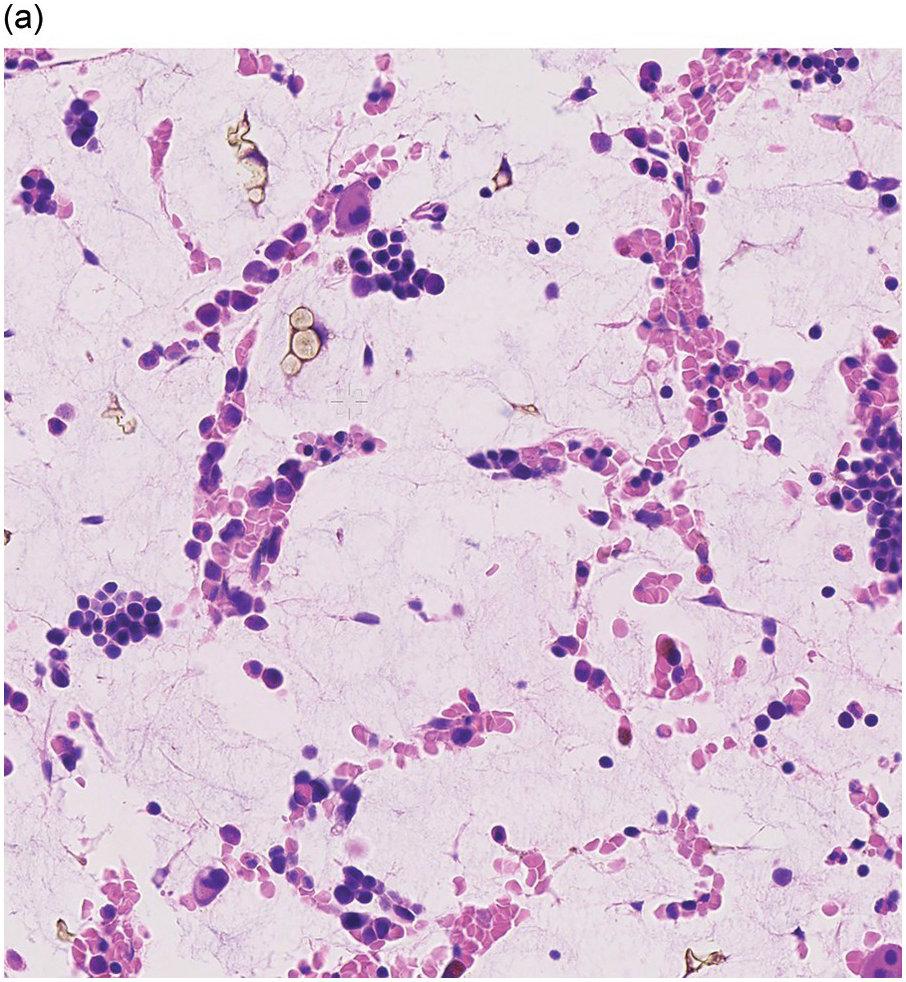
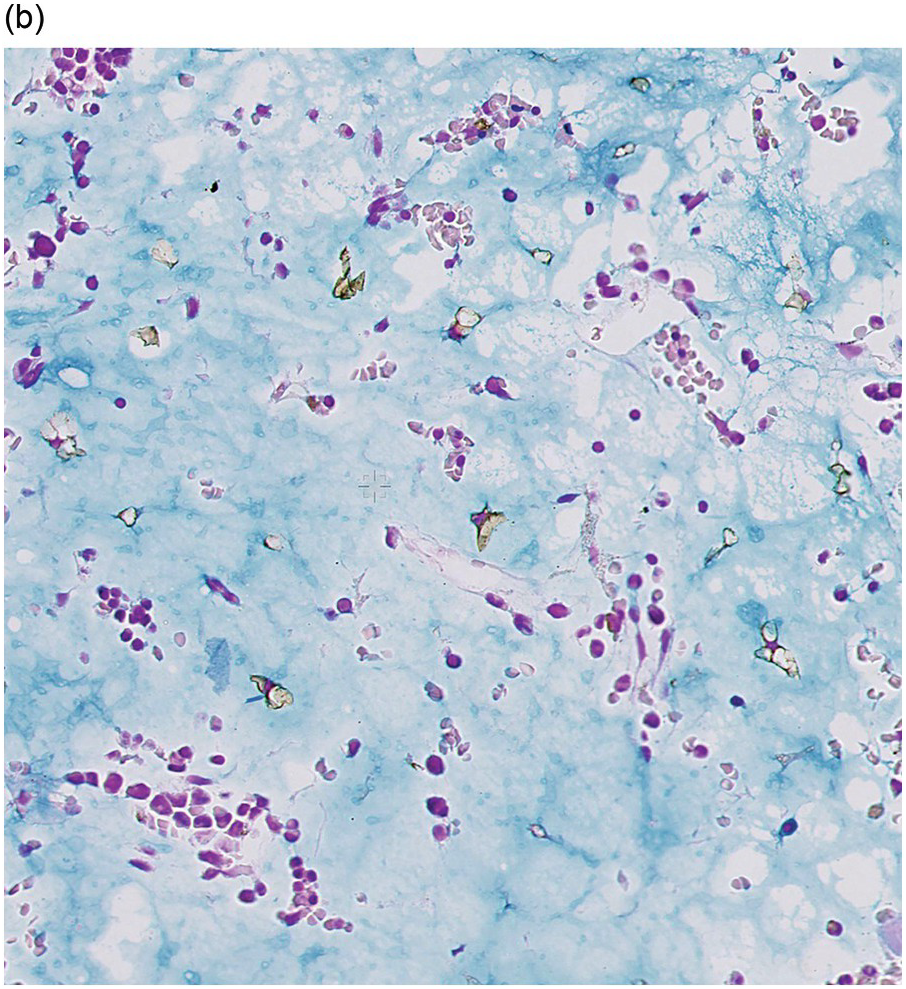
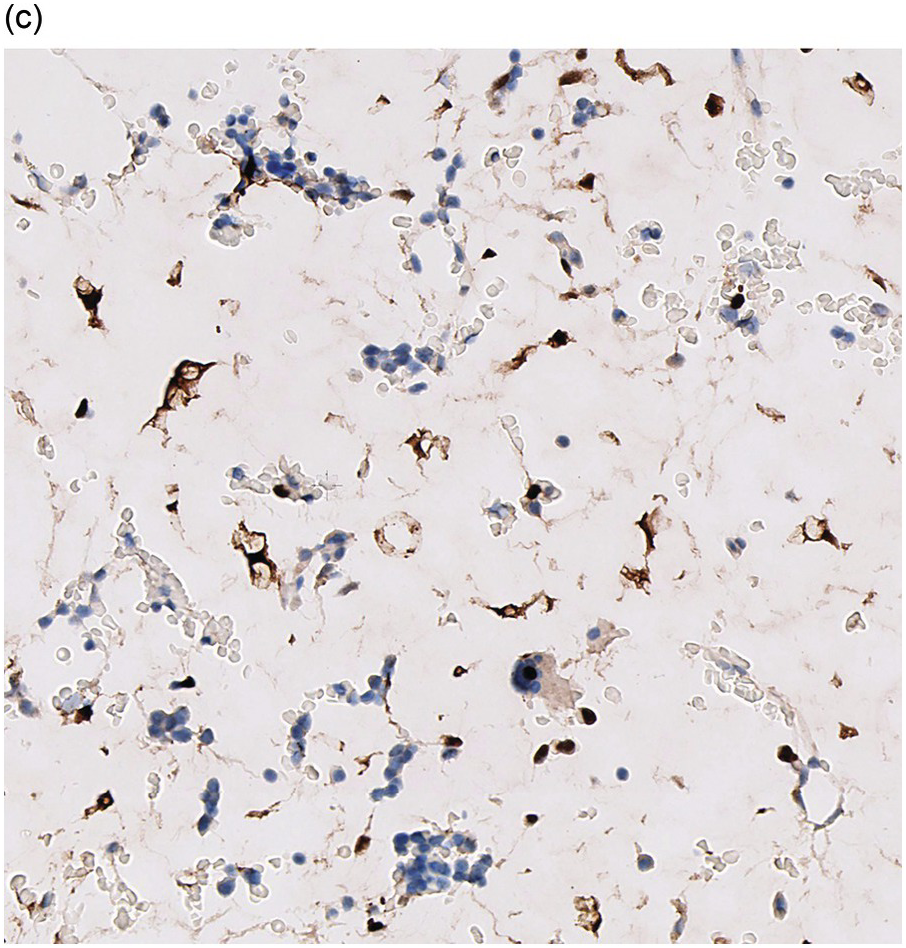
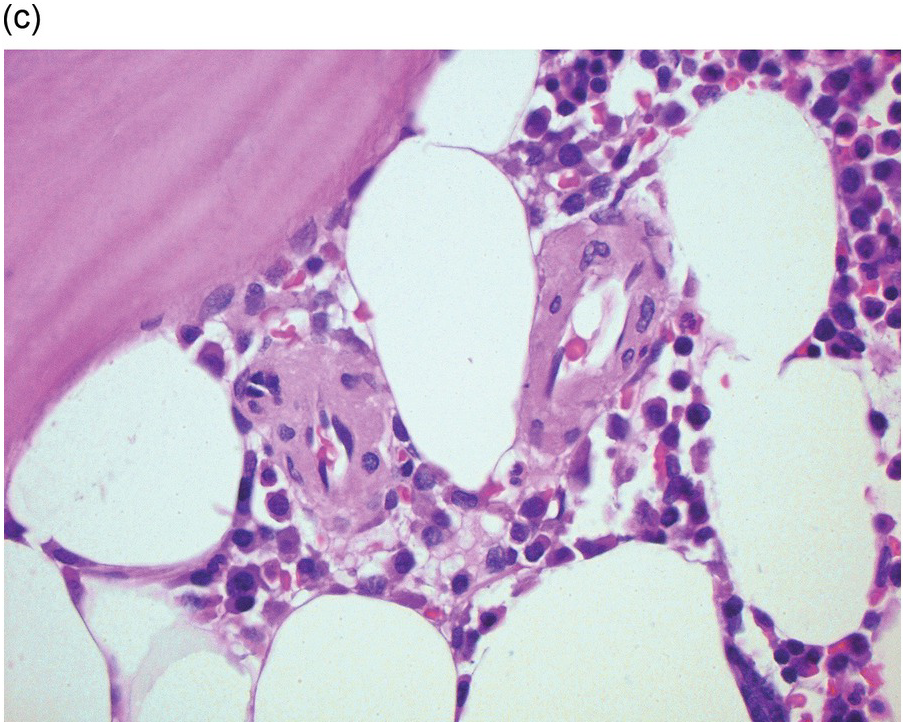
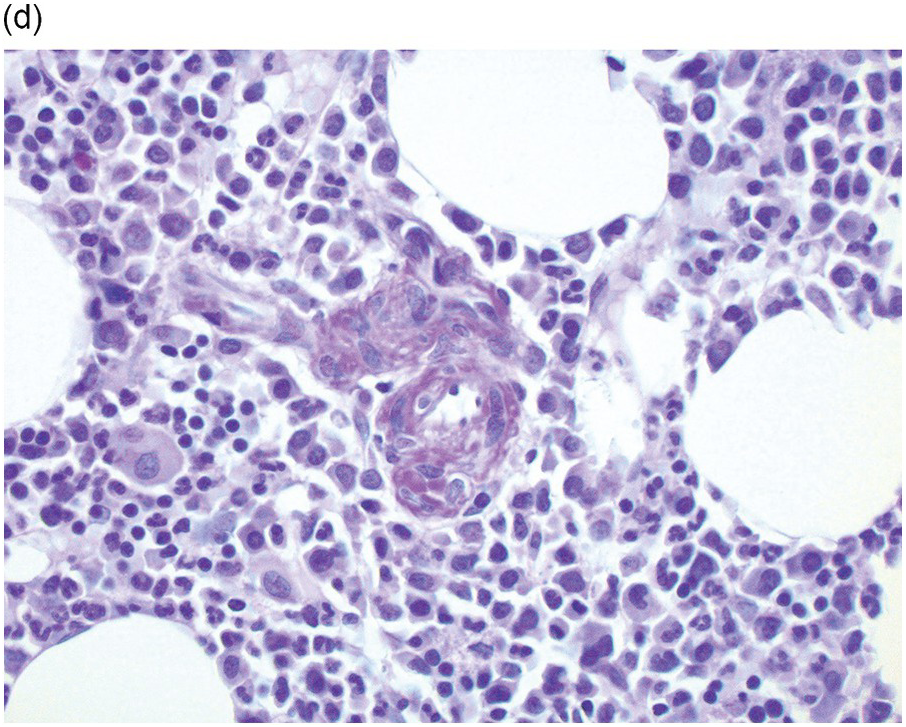
Amyloidosis is characterized by deposits of eosinophilic amorphous, PAS-positive material in vascular walls or the ECM (Figure 3.4a,b). These are highlighted by a Congo red stain and show apple-green birefringence when the Congo red-stained deposits are viewed with polarized light. Fluorescence microscopy increases sensitivity of Congo red-stained amyloid detection [12]. Immunohistochemistry for AA amyloid, immunoglobulin heavy and/or light chains and transthyretin can help to determine the nature of the amyloid.
Figure 3.4 Amyloid deposition in small vessels (a) and as interstitial aggregates (b). Amyloid-like (c) PAS-positive (d) immunoglobulin depositions occurring in rare non-amyloid light chain deposition disease.
Amyloid consists of naturally occurring misfolded proteins that arrange in insoluble sheets. Although amyloid is mainly deposited extracellularly, sporadic cases with spindle-shaped intracellular inclusions have been described [13]. Macrophages can accumulate at the edge of large amyloid depositions.
Many different proteins have been identified in a variety of clinical presentations of amyloidosis [14]. In the BM this is almost entirely limited to deposition of monoclonal immunoglobulins, light chains (AL amyloidosis) or, less frequently, heavy chains that acquire their unique structural features due to specific mutations. An exception is the deposition of amyloid consisting of β2-microglobulin in almost 20% of patients after more than ten years of dialysis, although in the iliac crest β2-microglobulin deposition is localized predominantly to the periosteum, not to the marrow [15]. Other non-immunoglobulin amyloid depositions in BM biopsies consist of inflammatory related AA-amyloid in nearly 80% of patients with familial Mediterranean fever [16], and deposition of mutant transthyretin in 30 to 40% of patients with transthyretin (ATTR) cardiomyopathy [17].
The aetiology of immunoglobulin-derived amyloidosis is usually a plasma cell clone: plasma cell myeloma, AL amyloidosis, plasmacytoma or, less frequently, Waldenström’s macroglobulinaemia/lymphoplasmacytic lymphoma or other lymphoproliferative disorders (see Chapter 16) [18].
Epidemiology
Light chain amyloidosis is the most common systemic amyloidosis, with an incidence of ten patients per million per year. Amyloid deposits are present in 60% of the BMB at diagnosis in light chain amyloidosis, mainly in blood vessel walls [18]. In 1 to 40% of plasma cell myeloma patients without systemic amyloidosis, amyloid deposits can nevertheless be detected in the BM by a Congo red stain [13, 19].
Differential Diagnosis
Amyloid-like PAS-positive immunoglobulin depositions without green birefringence of Congo red-stained tissue and lacking amyloid fibrils by electron microscopy, occur in rare non-amyloid light chain deposition disease (Figure 3.4c,d) [20].
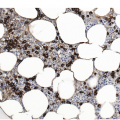
Myelofibrosis and Osteosclerosis
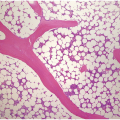
The normal BM stroma harbours a framework of fine reticulin fibres (type III collagen) that surround the bone, blood vessels and fat cells, and can be highlighted as thin black fibres with silver impregnation stains (Figure 3.5). Pathologic fibrosis ranges from a loose network of reticulin fibres to dense sclerosis, consisting of type I collagen deposition. Collagen type I is not normally present in the BM and stains bluish-green in a trichrome stain. Several soluble factors such as TGF and PDGF are implicated in the initiation and maintenance of myelofibrosis, but active TGF-beta plays a central role [1, 21]. Inactive TGF-beta produced by megakaryocytes is abundantly present in the ECM [22]. After activation by reactive oxygen species, proinflammatory cytokines and proteases, that can be released by a wide range of reactive and neoplastic processes, TGF-beta leads to increased production of collagens I, III, IV and V, and bone morphogenic proteins, and decreased synthesis of tissue inhibitors of metalloproteinase (TIMP) [1, 21]. Since collagen synthesis is a dynamic process, myelofibrosis can decrease after treatment of the underlying disease [23–26].