Key points
- 1.
Women who present with a breast mass require triple assessment , which comprises physical examination, breast imaging with mammography and ultrasound, and pathologic or cytologic evaluation. Discordant results require further investigation and careful follow-up.
- 2.
Breast cancer screening recommendations beyond established guidelines are informed by individual risk assessment .
- 3.
Breast-conserving therapy , which includes lumpectomy and adjuvant radiation therapy, and mastectomy share equivalent survival. Surgical decision making is informed by whether breast conservation is thought to be feasible with acceptable cosmesis as well as by patient preference and other factors. Reconstructive options should be discussed with all patients who elect to undergo mastectomy.
- 4.
Women with invasive cancer require axillary staging , which is achieved with sentinel lymph node biopsy (SLNB) or with axillary lymph node dissection (ALND). Clinically node-negative (cN0) patients should undergo SLNB; ALND is required for women who have any positive nodes in the setting of mastectomy or have three or more positive nodes or gross extra-nodal extension in the setting of breast conservation.
- 5.
BRCA gene testing and genetic counseling should be offered to women who meet testing criteria. Surveillance programs for BRCA carriers should include annual clinical breast examination in conjunction with imaging with mammography and MRI. Chemoprevention for BRCA2 gene carriers and risk-reducing surgery, including bilateral mastectomy and bilateral salpingo-oophorectomy, should be discussed with all BRCA mutation carriers.
Introduction
Breast cancer remains the most common malignancy among American women, comprising 15.2% of incident cancer cases in the United States. The American Cancer Society estimates that approximately 281,550 incident cases of invasive breast cancer in females in addition to approximately 48,000 incident cases of ductal carcinoma in situ (DCIS) will be diagnosed in 2021. Approximately 43,600 women are expected to die from breast cancer, making breast cancer the second-leading cause of cancer-related death after lung cancer.
The lifetime risk among women of developing breast cancer is 12.5% (1 in 8); the lifetime risk of dying from breast cancer is 3.6% (1 in 28). Recent advances in screening and systemic therapies have been credited with improved disease-free and overall survival. This decline is thought to be secondary to the increased use of mammographic screening with early detection of breast cancer and the use of effective adjuvant therapies.
This chapter presents an overview of breast cancer screening, diagnosis, and treatment of benign and malignant conditions of the breast. Some of the many factors that have led to improved disease-specific survival include early screening and improvement in adjuvant therapies as well as decreased rates of hormone replacement therapy (HRT) as a result of the Women’s Health Initiative (WHI) reports.
Anatomy of the breast
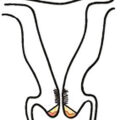
The adult breast lies between the second and sixth ribs in the vertical plane, and between the sternal edge (medially) and mid-axillary line (laterally). The average breast measures 10 to 12 cm in diameter and 5 to 7 cm in thickness. It is concentric, with a lateral projection into the axilla named the axillary tail of Spence ( Figs. 11.1 and 11.2 ).
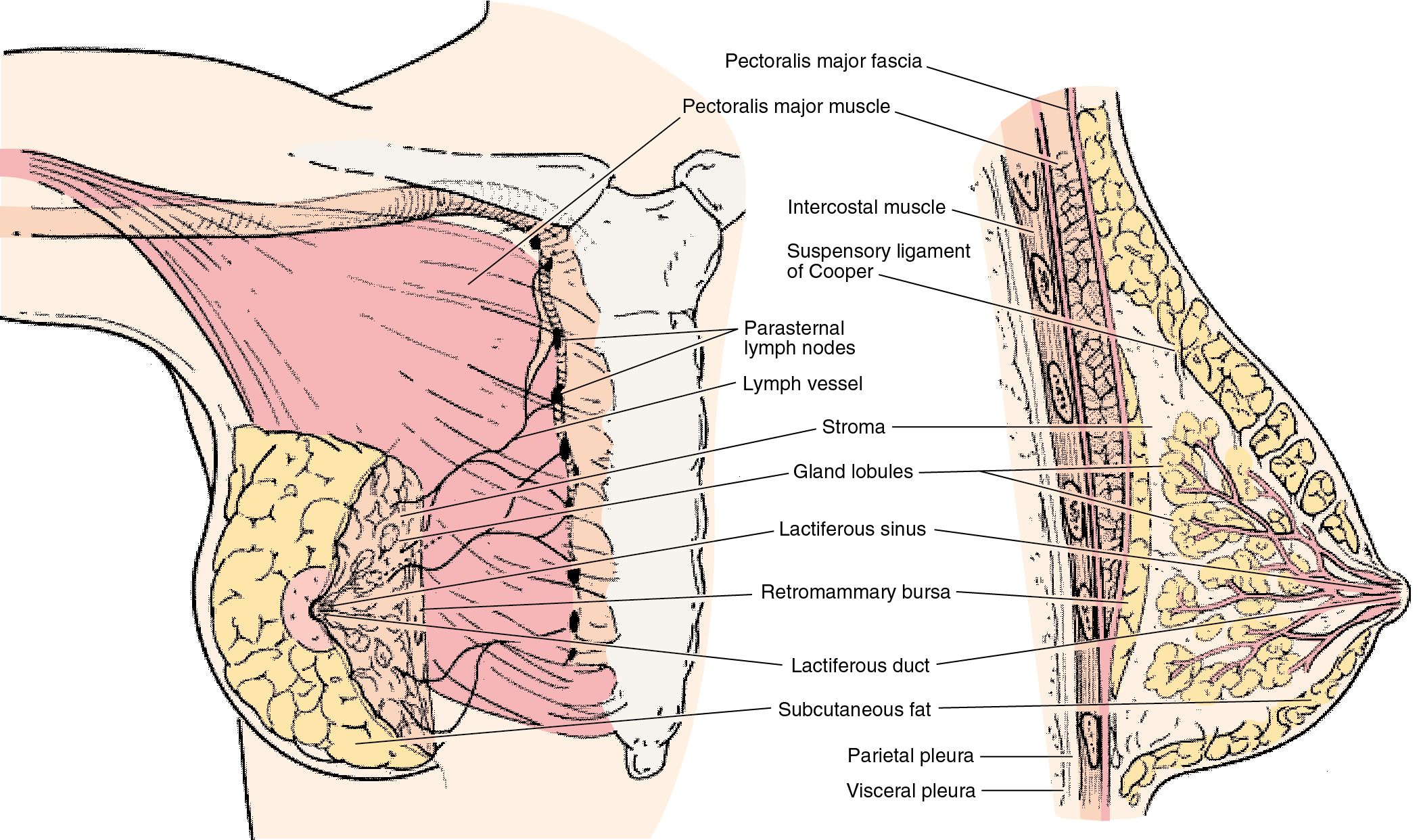
The breast consists of three major structures: skin, subcutaneous fatty tissue, and breast tissue (parenchyma and stroma). The skin contains hair follicles, sebaceous glands, and eccrine sweat glands. The glandular breast is divided into 15 to 20 segments (lobes) that are separated by connective tissue and converge at the nipple in a radial arrangement. These lobes are made up of 20 to 40 lobules, which in turn consist of 10 to 100 alveoli (terminal duct lobular units). Five to 10 major milk-collecting ducts drain each segment and open at the nipple into subareolar lactiferous sinuses.
The superficial pectoral fascia envelops the breast; the undersurface of the breast lies on the deep pectoral fascia. The superficial pectoral fascia is continuous with the superficial abdominal fascia of Camper. Cooper suspensory ligaments between these fascial layers provide support for the breast. The retromammary bursa comprises the space between the deep layers of the superficial fascia of the breast and the deep investing fascia of the pectoralis.
The epidermis of the nipple (mammary papilla) and areola is pigmented and wrinkled, and consists of keratinized, stratified squamous epithelium that contains smooth muscle fibers in dense connective tissue that enable erection of the nipple. Ruffini-like bodies and the end bulb of Krause within the nipple transmit stretch, pressure, and temperature sensation.
While the areola is generally devoid of hair follicles, it has sweat glands and Montgomery glands, also called accessory areolar glands. Montgomery glands are intermediate-stage sebaceous glands capable of secreting milk; they represent an intermediate stage between the sweat and the mammary glands.
The principal blood supply of the breast is derived from the internal thoracic (mammary) and lateral thoracic arteries, and their tributaries. Approximately 60% of the breast, primarily the medial and central breast, is supplied by the anterior perforating branches of the internal mammary artery. About 30% of the breast, mainly the upper outer quadrant, is supplied by the lateral thoracic artery. The second to fourth posterior intercostal arteries also give off tributaries called the mammary branches. The superficial veins follow the arteries and drain through perforating branches of the internal thoracic vein, tributaries of the axillary vein, and perforating branches of posterior intercostal veins. The veins anastomose circumferentially around the nipple forming the circulus venosus.
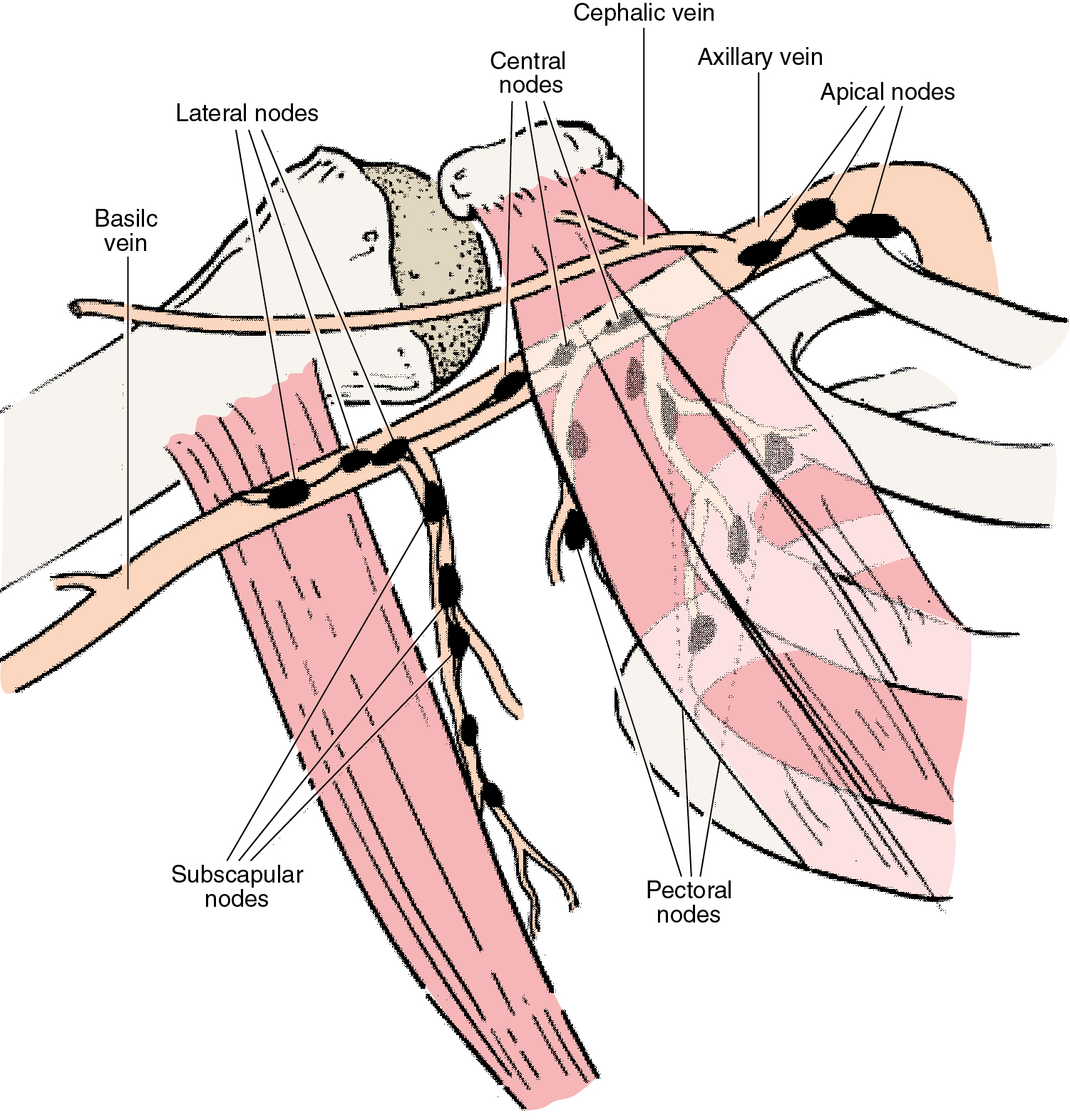
The papillary plexus of breast lymphatics is confluent with the subepithelial lymphatics over the surface of the body. These valveless lymphatic channels communicate with subdermal lymphatic vessels and converge with Sappey’s subareolar plexus. The subareolar plexus receives lymphatic vessels from the nipple and the areola, and communicates by way of the vertical lymphatic vessels analogous to those connecting the subepithelial and subdermal plexus elsewhere in the body. Lymph flows unidirectionally, from the superficial to the deep plexus, and from the subareolar plexus to the perilobular and deep subcutaneous plexus via the lymphatic vessels of the lactiferous ducts. The lymphatic flow from the deep subcutaneous and intramammary lymphatic vessels moves centrifugally toward the axillary and internal mammary lymph nodes. It is estimated that approximately 97% of the lymph flows to the axillary nodes and that 3% flows to the internal mammary chain.
Axillary lymph nodes may be divided into three anatomic levels defined by their relationship to the pectoralis minor muscle. Level I nodes are the axillary vein lymph nodes that lie along the axillary vein from the lateral extent of the pectoralis minor muscle to the latissimus dorsi muscle. In this area, deep to the floor of the axilla, subscapular nodes that lie on the subscapularis muscle are identified. Level II nodes are designated by their location behind the pectoralis minor muscle. Medial to the pectoralis minor are level III nodes, which include apical or infraclavicular nodes and are adjacent to Halstead’s ligament. Interpectoral nodes are called Rotter’s nodes.
Embryology and development of the breast
The mammary glands are highly specialized skin derivatives of ectodermal origin. The epithelial ridge that develops into breast tissue undergoes a series of proliferations to form the lactiferous ducts. Paired mammary ridges extend from the bilateral axillae to the medial thighs on the ventral surface of the embryo starting at the fourth week of gestation. All but the pair at the fourth intercostal space involute subsequently. The remaining ridges proliferate, forming the primary mammary bud, which forms a solid diverticulum into the underlying dermis in response to signals from the underlying mesoderm. As the primary mammary bud branches, it forms secondary buds and gives rise to lobules. Placental hormones induce canalization of the mammary buds, forming the ductal network which converges into a mammary pit. Inward proliferation of the ectoderm induces the formation of a nipple with smooth muscle fibers that are surrounded by areolar ectoderm.
After birth, breast tissue remains dormant until adolescence, when estrogen produces a proliferation of ductal epithelium and progesterone produces rapid growth of the acini. Breast growth and development, however, are not completely dependent on estrogen and progesterone levels. Complete functional development also requires insulin, cortisol, thyroxine, growth hormone, and prolactin. Minor deficiencies in any one of these hormones can be mitigated by an excess of prolactin.
Pregnancy and breast feeding
Increasing amounts of estrogen, progesterone, and human placental lactogen stimulate growth of functional breast tissue during pregnancy. Estrogen production from the fetus modulates progesterone production, uteroplacental blood flow, mammary gland development, and fetal adrenal gland function. By the 20th week of pregnancy, most of the estrogen excreted in maternal urine comes from fetal androgens, and 90% of maternal estriol is derived from fetal precursors. Serum prolactin rises from non-pregnant levels of 10 ng/mL to term levels of 200 ng/mL. Additionally, decidual endometrial cells produce high levels of prolactin in the amniotic fluid, which approach over 100 times higher than serum levels in maternal or fetal blood during early pregnancy.
Estradiol and prolactin-inhibiting factor modulate prolactin secretion. Elevated levels of estradiol parallel those of prolactin and indicate that estriol may be responsible for increases in prolactin; however, high levels block its physiologic effects. Prolactin levels are increased by both reduced post-partum serum estrogen levels and suckling-induced suppression of prolactin-inhibiting factor. If breast feeding does not occur, serum prolactin levels decrease to non-pregnant levels in about 1 week. The release of oxytocin from the posterior pituitary gland affects contraction of the duct system, stimulating the delivery of milk to the nipples; however, after 3 to 4 months of breast feeding, suckling appears to be the only stimulus required for lactation.
Benign breast conditions
Fibrocystic change
Fibrocystic change is the most common benign breast condition in women and is not a risk factor for subsequent development of cancer. It encompasses a histologic spectrum of changes in the breast, some of which are normal and some of which are abnormal. Fibrocystic change is most common in premenopausal women between the ages of 20 and 50 years as the result of fluctuating hormone levels. It is typically bilateral and associated with mastodynia (breast pain). Most women will report symptoms during the premenstrual phase of the cycle, which suggests that progesterone may play a role in the development and symptoms of cystic alterations in breast tissue. However, the proportional effect of both estrogen and progesterone on the cause of benign breast conditions is unclear.
Mastodynia results from breast stromal edema, ductal dilation, and associated inflammatory response. An increase in breast size is also frequently reported. The differential diagnosis for mastodynia includes other conditions affecting the anterior chest wall, such as intercostal neuralgia, myalgia, and chronic costochondritis. Women with large, pendulous breasts may have stretching of Cooper ligaments, which is also associated with mastodynia. They may also experience neck, back, and shoulder pain.
In the initial management of fibrocystic change, it is of paramount importance to rule out any underlying breast cancer with appropriate imaging and work-up. Symptomatic women may benefit from counseling regarding proper selection and fitting of a brassiere, as improved mechanical support may relieve breast pain. The effectiveness of dietary interventions is uncertain. A low-fat diet was associated with a substantial improvement in mastalgia symptoms when 21 patients with severe mastopathy were randomized to a diet containing 15% fat intake or a general diet containing 36% fat intake. Although many women report that a reduction in caffeine intake alleviates their breast pain, clinical studies have not shown consistent findings. In an uncontrolled study, 61% of women with breast pain who substantially decreased caffeine intake for 1 year had decreased pain or complete relief. Studies with small numbers of patients suggested that vitamin E is potentially beneficial in fibrocystic change. While evening primrose oil (gammalinolenic acid) has been widely advocated as an option, studies have failed to show a benefit relative to placebo.
Hormonal manipulation may alleviate mastodynia but is associated with other side effects. There is no consensus with regards to which of these is superior. Danazol, an androgen, was the only medication approved by the U.S. Food and Drug Administration (FDA) for treatment of mastodynia, but as of 2018 is no longer FDA approved for this indication. While danazol results in alleviation of breast pain in most patients at a dose of 200 mg daily, its utility is limited by its androgenic side effects, including weight gain, menstrual irregularities, voice changes, acne, hair loss, depression, and vasomotor symptoms. Some patients experience improvements in adverse effects by restricting use of danazol to the luteal phase of the menstrual cycle while achieving similar relief from mastodynia.
The selective estrogen receptor (ER) modulator tamoxifen, which is commonly used for chemoprevention and treatment of breast cancer, is effective in reducing pain in 71% to 96% of women with cyclic mastalgia, and 56% of women with non-cyclical mastalgia, in controlled trials. While tamoxifen achieves similar pain relief at 10 and 20 mg daily, the lower dose is better tolerated with a more favorable side-effect profile. Potential adverse effects include vasomotor symptoms, vaginal dryness, weight gain, and arthralgia, as well as increased risk of venous thromboembolism and endometrial carcinoma. For some women, the side effects lead to discontinuation.
Proliferative changes
Proliferative changes include hyperplasia as well as adenosis. Hyperplasia is proliferation of ductal epithelium and results in the layering of the cells; atypia may be associated with this proliferation. There are two types of atypical hyperplasia based on microscopic appearance: atypical ductal hyperplasia (ADH) and atypical lobular hyperplasia (ALH). Both lesions are associated with an increased relative risk (RR) of approximately four for development of a later invasive breast cancer.
The American Society of Breast Surgeons issued a consensus guideline on management of borderline or high-risk lesions to help guide clinical decision making. The management of these lesions depends on imaging-pathology concordance and presence of atypia. Surgical excision is recommended because some studies have reported rates approaching 10% to 20% of finding invasive or in situ carcinoma in conjunction with ADH found on core biopsy, and because distinguishing between ADH from DCIS can be challenging on core needle biopsy. Lower rates of upgrade, as low as less than 5%, have been reported among women with low-risk features, including the absence of mass lesion on imaging, removal of greater than 50% of the mammographic lesion, absence of necrosis, and involvement of two or fewer terminal duct lobular units. The absence of prospective data that validate these criteria limit routine omission of surgical excision of ADH.
In the case of ALH, recent studies have suggested the invasive cancer “upgrade” rate is lower (less than 3%) after surgical excision of ALH in the setting of imaging-histologic concordance. Therefore, if radiologic findings are concordant or the findings are incidental, excision may not be warranted. All patients with atypical hyperplasia warrant careful clinical and radiological follow-up.
Adenosis is a benign proliferative lesion caused by changes in the acini in the distal mammary lobule. Sclerosing adenosis refers to the dense, fibrotic tissue surrounding these small ducts. These lesions may present as a palpable mass in women in their 30s and 40s, and generally do not require excision.
Papillomas are benign papillary lesions with branching fibrovascular cores surrounded by epithelium. They are associated with serosanguineous nipple discharge in 25% to 50% of patients and typically present as a small palpable mass adjacent to the areola. Case series of patients with solitary intraductal papillomas without atypia have demonstrated low rates of upgrade, less than 5%. As a result, small papillomas without atypia that are concordant with imaging may be observed; however, papillomas with atypia warrant excision as atypia has been associated with an unacceptably high rate of upgrade to carcinoma, which approached 37% in a recent meta-analysis of 34 studies including 2236 non-malignant breast papillary lesions diagnosed at core needle biopsy. Papillomas that are not excised require close clinical and imaging surveillance.
Complex sclerosing lesions
Complex sclerosing lesions have been described by a variety of names, including sclerosing papillary proliferation, non-encapsulated sclerosing lesion, indurative mastopathy, and radial scar. They are important because they may simulate carcinoma on mammographic, gross, and microscopic examinations. These lesions are typically less than 1 cm in diameter. On gross examination, they are irregular, gray or white, and indurated with central retraction. On microscopic examination, the lesion has a stellate configuration and consists of a central, fibrotic core containing entrapped glandular elements. The surrounding breast tissue typically shows varying degrees of intraductal hyperplasia and adenosis. The significance of this lesion relative to subsequent development of carcinoma is controversial, though the rate of upgrade has been reported to be as high as 25%. Local excision of these lesions is the treatment of choice, though observation may be considered for small radiologic-pathologic concordant lesions that are completely removed or well-sampled with core needle biopsy.
Fibroadenoma
Fibroadenomas are benign fibroepithelial tumors and the most common lesion found in women younger than 25 years of age. They will persist during the menstrual years of a woman’s life, but regression after menopause has been reported. Patients typically present with a mobile, smooth, painless, palpable mass. Ultrasound examination along with physical examination can help make the diagnosis.
Mammographically, fibroadenomas typically appear as round, oval, or lobulated masses with circumscribed margins. In older women, they can have a rim of coarse calcifications. Fine-needle aspiration (FNA) will reveal benign ductal epithelial cells and elongated dense stromal cells. Microscopically, fibrous tissue comprises the majority of the fibroadenoma. Carcinomas arising from fibroadenomas are rare.
Fibroadenomas can be followed without the need for complete surgical excision. This can be achieved with physical examination or ultrasound examination if they are not palpable. However, surgical excision should be performed if the mass continues to enlarge, the results of FNA or core biopsy are inconclusive or yield atypia, there is an increased cellularity, stromal mitoses or overgrowth, there is presence of nuclear pleomorphism, or the patient desires surgical excision.
Table 11.1 lists proliferative breast disease and breast cancer risk. Table 11.2 shows breast diagnoses grouped by cancer risk.
| Characteristic | Relative Risk (95% Confidence Interval) |
|---|---|
| Proliferative disease, no atypia | 1.3 (0.69–11.9) |
| Complex fibroadenoma a | 1.46 (0.53–14.0) |
| Atypical hyperplasia | 2.53 (1.01–6.3) |
| Neither proliferative disease nor complex fibroadenoma | 1.27 (0.89–11.8) |
a Contains cysts, sclerosing adenosis, epithelial calcification, or papillary apocrine changes.
| Cancer Risk | Diagnosis |
|---|---|
| No increased risk | Adenosis, sclerosing or florid Apocrine metaplasia Cysts, macro, or micro Duct ectasia Fibroadenoma Fibrosis Hyperplasia (mild) Mastitis Periductal mastitis Squamous metaplasia |
| Slightly increased | Hyperplasia, moderate or florid Papilloma, solid or papillary, with fibrovascular core |
| Moderately increased | Atypical hyperplasia Ductal Lobular |
Phyllodes tumor
Phyllodes tumors are uncommon, locally aggressive fibroepithelial tumors. Previously referred to as cystosarcoma phyllodes, this name contributed to confusion in understanding this entity. Although very similar to a fibroadenoma, the stromal component is hypercellular with increased pleomorphism and mitotic activity. Phyllodes tumors can occur in women of any age, but more commonly occur in premenopausal women. They are classified as benign, borderline, or malignant based on assessment of four features: stromal cellular atypia, mitotic rate, presence of infiltrative tumor margins, and presence of stromal overgrowth.
Malignant phyllodes tumors are rare among premenopausal women. Incomplete excision is a major determinant for local recurrence. Treatment is total surgical excision with margins of 1 cm, though narrower margins are accepted for benign phyllodes tumors. Because axillary involvement is rare even among malignant phyllodes tumors, axillary surgery is not indicated.
Adenoma
Adenoma of the breast is a well-circumscribed tumor composed of benign epithelial elements with sparse, inconspicuous stroma; this feature differentiates adenomas from fibroadenomas in which the stroma is an integral part of the tumor. Adenomas may be divided into two major groups: tubular adenomas and lactating adenomas. Tubular adenomas in young women are well-defined, freely movable nodules that clinically resemble fibroadenomas. Lactating adenomas manifest as one or more freely movable masses during pregnancy or the postpartum period. They are grossly well-circumscribed and lobulated; on cut section, they appear tan and softer than tubular adenomas. On microscopic examination, these lesions have lobulated borders and are composed of glands lined by cuboidal cells with secretory activity identical to the lactational changes normally observed in the breast tissue during pregnancy and during the postpartum period.
Because of the pregnancy and lactation-related changes resulting in increased breast vascularity, extreme caution should be taken during surgery with any invasive breast procedure given the elevated risk of postoperative hematoma formation. Moreover, the lactating breast is predisposed to postoperative infection because milk is a good culture medium. Because of the significant risk of infection and milk fistula, the patient who is lactating should cease lactating before biopsy is performed.
Superficial thrombophlebitis
Superficial thrombophlebitis, also known as Mondor’s disease of the breast, is an uncommon benign inflammatory process. While it can occur spontaneously, superficial thrombophlebitis is usually associated with breast trauma, breast surgery, or pregnancy. It is a thrombophlebitis of the thoracoepigastric vein, which drains the upper-outer quadrant of the breast. Patients present with acute pain and a linear, tender fibrotic band with skin retraction over the distribution of the thoracoepigastric vein. Treatment is conservative, with analgesics and application of heat, and typically resolves in 1 to 3 weeks. Skin retraction superficial to the area of inflammation can remain if the inflammation is extensive. Biopsy is not necessary.
Mastitis
Mastitis usually occurs in relation to lactation. It can occur in non-puerperal periods in association with galactorrhea. Skin organisms, Staphylococcus aureus, and Streptococcus species may cause infection of the nipple and breast ducts. The presence of milk in the ducts can provide an excellent medium for infection.
Women with mastitis may continue to breast feed. Antibiotic therapy with dicloxacillin sodium (500 mg qid) or penicillin G is indicated. If there is not a response, an abscess that may require surgical drainage must be excluded. Inflammatory carcinomas can mimic mastitis, and if no resolution of infection is noted despite continued antibiotics, a skin biopsy may be indicated.
Galactoceles are milk-filled cysts. They are usually tender and present after the abrupt termination of breast feeding. Aspiration of the cyst is often necessary for symptomatic relief. If re-accumulation occurs, however, surgical excision may be required to avoid infection.
Duct ectasia
Duct ectasia is a condition that usually occurs in perimenopausal or postmenopausal women. Patients present with a tender, hard, erythematous mass adjacent to the areola in association with burning, itching, or a pulling sensation in the nipple area. A thick, greenish-black discharge may be present.
The pathogenesis of this condition has not been fully established; however, it is thought that periductal inflammation leads to periductal fibrosis, which subsequently results in ductal dilation. The etiology of the inciting inflammatory response remains poorly understood, however. Histologic evaluation demonstrates dilated, distended terminal-collecting ducts obstructed with inspissated lipid-containing epithelial cells and phagocytic histiocytes. This process tends to occur in a segmental fashion, extending from the involved nipple area to adjacent ducts. Occasionally, a small subareolar abscess forms at the base of the nipple.
Younger women can have inflammation of the ducts in the region of the nipple, which may produce fissures and fistulae from the nipple ducts to the skin at the areolar margin. Prior periductal mastitis may lead to squamous metaplasia of the lactiferous ducts. Keratin formed in the ducts can accumulate and form a subareolar abscess. This condition is associated with smoking, and excision of the area usually is necessary.
Fat necrosis
Breast trauma and surgery may incite fat necrosis. Patients may present with a palpable irregular firm mass and skin retraction that can mimic carcinoma. Multiple calcifications can be seen on mammography.
The histology is active chronic inflammatory cells, with lymphocytes and histiocytes predominating. In the later stages, a collagenous scar is noted, with “oil cysts” or free lipid material released by lipocyte necrosis. Fat necrosis is a benign process and is not associated with an increased risk of subsequent carcinoma.
Nipple discharge
Nipple discharge has been reported in 10% to 15% of women with benign breast disease and in 2% to 3% of those with carcinoma. Galactorrhea presents as bilateral, milky nipple discharge consisting of lipid droplets; the condition is usually idiopathic, but can be found after discontinuation of oral contraceptives or as a persistent discharge after pregnancy. Plasma prolactin levels should be measured, as prolactin-producing pituitary adenomas can also cause galactorrhea.
Nipple discharge is classified according to its appearance as milky, green, bloody, serous, cloudy, or purulent. The drainage should be classified according to whether it is unilateral, bilateral, spontaneous, or recurrent. This information is obtained at the time of a thorough history and physical examination. Additionally, the presence of a mass should also be investigated. The risk of cancer is increased when the discharge is unilateral from a single duct, when it occurs in a postmenopausal patient, or when a mass is present. Diagnostic imaging should include a mammogram and ultrasound with MRI at the discretion of the treating physician. Lactiferous duct exploration and excision may be required.
The most common cause of unilateral, spontaneous nipple discharge is mammary-duct ectasia. The classic reddish-brown discharge is often mistaken for a blood discharge and is thought to result from an increase in glandular secretions, with the production of an irritating lipid fluid that can produce a nipple discharge.
The next most common cause of a multicolored, sticky nipple discharge is non-puerperal mastitis. The transient subtype is associated with periareolar inflammation, whereas the persistent subtype involves inflammation in deeper portions of the breast. If the inflammation develops into an inflammatory mass, surgical excision and drainage is necessary. Medical management with local care, avoidance of all nipple manipulation, and administration of non-steroidal antiinflammatory agents and anti-staphylococcal antibiotics is often successful when infection is suspected.
Bloody nipple discharge warrants surgical evaluation. Intraductal papillomas are the most common cause of bloody nipple discharge. During the breast examination, physicians should look for an associated periareolar mass. The examination consists of gently and carefully palpating the subareolar region to identify the pressure point that produces the discharge. It is important to reproduce the discharge and demonstrate the breast quadrant from which it emanates. All significant nipple discharges warrant referral for tissue biopsy. Although a mass is usually present when the discharge is a result of cancer, there is no palpable mass in 13% of cancers with nipple secretions. Bloody discharge occurring in the third trimester of pregnancy may be regarded as physiologic, however, and does not require intervention unless persistent for several months after delivery. There are no contraindications to breast feeding in these patients. Cytology of the discharge is not routinely recommended because of a reported low sensitivity for detection of cancer (approximately 27%). While galactography (injecting radiopaque contrast into the discharging duct and then performing mammography) offers better visualization of small intraductal papillomas, it cannot differentiate between benign and malignant lesions. Surgical excision is still necessary. Mammography has a 9.5% false-negative rate and a 1.6% false-positive rate for detecting cancer in patients with a nipple discharge. Table 11.3 reveals characteristics of nipple discharge.
| Color | Likely Cause | Percentage Caused by Cancer |
|---|---|---|
| Milky (galactorrhea) | Pituitary adenoma, pregnancy, oral contraceptives | Rare |
| Green, yellow, sticky | Ductal ectasia | Rare |
| Clear, watery | Ductal carcinoma | 30–50 |
| Bloody, sanguineous | Fibrocystic changes, ductal papillomas | 25 |
| Pink, serosanguineous | Fibrocystic changes, ductal papillomas | 10 |
| Yellow, serous | Fibrocystic changes, ductal papillomas | 5 |
| Purulent | Bacterial infection | Rare |
History and physical examination
It is essential to obtain a thorough patient medical history, including a family history and information on menstrual status, pregnancies and lactation, hormone use, and prior breast surgeries and trauma. Additionally, it is important to ascertain whether the patient has felt any breast masses and determine the presence and characterization of any nipple discharge.
Bilateral breast examination is best performed following menstruation and before ovulation. At that time, breast engorgement and tenderness are less likely to be present. A multi-positional breast and axillary examination should be performed, including examination in the upright and supine positions. Breast retraction and subtle changes in the skin and nipple may be missed if the patient is examined in only one position.
The examination should begin with the patient in the seated position as asymmetry, skin or nipple retraction, and nipple ulceration are most exaggerated ( Fig. 11.3A ). When the patient’s arms are raised ( Fig. 11.3B ), skin changes in the lower half of the breast or in the inframammary fold become accentuated. Contraction of the pectoralis major muscle, affected by the patient pushing her hands against her hips ( Fig. 11.3C ), may demonstrate an otherwise undetected skin retraction. Next, palpation of the breast with the patient still upright may allow detection of subtle lesions that would be more difficult to palpate if she were supine ( Fig. 11.3 D). Examination of the supraclavicular areas and both sides of the neck for the purpose of detecting suspicious lymphadenopathy is also best done when the patient is in the upright position.

The axilla should be examined with the patient in the upright position. The patient’s right arm should be fixed at the elbow and held there by the physician’s right hand. This allows relaxation of the chest wall musculature ( Fig. 11.3 E). Palpation with the left-hand permits assessment of the lower axilla, and with extension higher toward the clavicle, the middle and upper portions of the axilla can be assessed. The left axilla is examined with the right hand after relaxation of the patient’s left arm in the physician’s left hand. If lymph nodes are palpable, the clinician must assess their level and size, and whether they are suspicious, single or multiple, and mobile or fixed to underlying structures.
The second phase of the breast examination is conducted with the patient in the supine position and with the patient’s arm raised above the head ( Fig. 11.3 F). Digital palpation is carried out using the index and middle fingers, and by applying varying amounts of pressure with the flats or pads of the fingers. A thorough examination systemically covers the entire breast and chest wall. The examination can be done in a clockwise direction or by rows (stripwise). It is important to carefully examine beneath the nipple-areolar complex and within the axilla.
An inflammatory appearance of the breast should raise suspicion of an inflammatory carcinoma. The classic appearance of inflammatory breast cancer includes a red, swollen breast with skin edema (“peau d’orange”). The breast is generally not tender. If the inflammation persists following a short course of antibiotics to rule out cellulitis, biopsy of the breast and skin is warranted. Inflammatory breast cancer is often a clinical diagnosis, and a benign skin biopsy should not dissuade the clinician from undertaking further evaluation and treatment. Any asymmetric skin changes or changes of the nipple-areolar complex should arouse suspicion. Paget’s disease of the nipple suggests the presence of intraductal or invasive cancer involving the nipple, and cancer should be excluded by a nipple biopsy of the abnormal area following a mammogram.
If no abnormal findings are noted on clinical breast examination (CBE), it is critical to document negative findings. The date of the last mammogram, discussion of cancer screening, and plans for follow-up should also be recorded. Hormones (HRT or oral contraceptive pills [OCPs]) should not be renewed without a documented annual breast examination or mammography, if indicated. A great deal of litigation results from failure to diagnose breast cancer. Indeed, errors in diagnosis are the most common reason for malpractice claims against radiologists, and breast cancer is the most commonly missed diagnosis (3.57 claims per 1000 person-years). Additionally, in a recent study querying the LexisNexis database for cases related to breast cancer from 2010 to 2017, delay in breast cancer diagnosis was the most common reason for litigation (59%). Radiologists were the most commonly named defendants, making up 29% of defendants, followed by surgeons, who made up 28% of defendants. The verdict favored the defendant in 55% of cases.
CBE and breast self-examination (BSE), as methods for screening for breast cancer, mainly aim to detect palpable breast lesions. However, there are no published reports demonstrating these methods as being effective in breast cancer mortality risk reduction. A Cochrane systematic review published in 2003 included two large population-based studies from Russia and Shanghai, China that compared BSE without an intervention and did not find mortality benefit with BSE. In Russia, more breast cancers were diagnosed in the BSE group (RR 1.24, 95% confidence interval [CI] 1.09), but this increased detection was not observed in Shanghai. In both studies, almost twice as many biopsies with benign results were performed in the screening group compared to the control group (RR 1.89; 95% CI 1.79 to 2.00). A population-based trial of CBE was also included in the review; however, poor compliance with follow-up in the study precluded definitive conclusions. Since then, no studies have demonstrated improved patient outcomes, but both the CBE and BSE have been associated with high rates of false-positive findings, which can lead to invasive follow-up testing and are associated with potential psychological harms.
The breast cancer screening recommendations given by the 2016 United States Preventive Services Task Force (USPSTF) include encouraging patients to be aware of changes in their breasts, but they did not update their 2009 recommendations, which did not include a requirement for physicians to instruct women in BSE and cited insufficient evidence to recommend CBE. Similarly, the American Cancer Society (ACS) does not recommend CBE for breast cancer screening at any age among average-risk women; its recommendations stipulate that women should be familiar with their breasts and report changes to their providers. They also indicate that CBE may be offered to women with elevated breast cancer risk.
Breast imaging
Mammography
The primary goal of mammography is to screen asymptomatic women to help detect breast cancer at an early stage. The goal of screening mammography is to identify early-stage non-palpable cancers. Most studies of mammography use mortality as the endpoint, and the value of screening is often limited to mortality rates. Few studies factor the effects of early detection on quality of life or the fact that diagnosis and treatment at earlier stages are associated with less morbidity and more treatment options.
In general, a routine screening mammogram consists of a mediolateral oblique (MLO) view and a craniocaudal (CC) view of each breast. With modern low-dose screening, the dose is less than 0.1 rad per study (for comparison, a chest radiograph delivers 0.025 rad per study). The effectiveness of screening depends on the density of the breast: the greater the breast density, the lower the sensitivity of the mammogram.
Breast composition may be one of four patterns of increasing density:
- 1.
Almost entirely fat
- 2.
Scattered fibroglandular densities
- 3.
Heterogeneously dense
- 4.
Extremely dense
It is important to note that the false-negative rate for mammograms is 10% to 15%, and that a normal mammogram does not eliminate the need for further evaluation of a dominant mass in the breast. If the clinical examination is suspicious, a negative mammogram result should not delay further investigation. Breast density should be documented in the mammogram report, and many states require notification of the patient of their breast density in their results letter.
Mammographic screening in women 40 years of age or older has reduced mortality by 20% to 30%. The efficacy of screening mammography in decreasing breast cancer mortality has been demonstrated in numerous studies. In the 1960s the Health Insurance Plan of Greater New York performed a study of physical examination and mammography in a study group of 30,756 women and a control group of 30,239 women between the ages of 40 and 64 years. At 10 years follow-up, the study group had a 30% decrease in breast cancer mortality compared with the control group.
An updated Cochrane systematic review of screening mammography was published in 2013. The authors identified eight randomized trials, but only reported on seven due to inadequate randomization in one of the trials. A total of 600,000 women who were 39 to 74 years of age were included in the analysis. There were neither signs of statistically significant reduction in total cancer morality at 10 years, nor signs of reduction in breast cancer mortality or all-cause mortality at 13 years, in the three trials with adequate randomization; however, a significant reduction in breast cancer mortality (RR 0.75, 95% CI 0.67 to 0.83) was observed in the four trials with suboptimal randomization. The authors concluded that the effect of screening on breast cancer-specific and overall survival is uncertain in the setting of improved breast cancer awareness and more efficacious breast cancer treatments.
In the meta-analysis that informed the updated USPSTF breast cancer screening recommendations, there was evidence of RRs of breast cancer mortality based on age groups. While there was no mortality benefit for women age 39 to 49 years (RR 0.92, 95% CI 0.75 to 1.02) and 70 to 74 years (RR 0.67, 95% CI 0.51 to 1.28), a mortality benefit was observed for women age 50 to 59 years (0.86, 95% CI 0.68 to 0.97) and 60 to 69 years (RR 0.67, 95% CI 0.54 to 0.83). Observational studies demonstrated a risk reduction of 25% to 31% in breast cancer mortality with mammographic screening, but did not demonstrate a difference in all-cause mortality. Additionally, while women age 50 years or older had lower rates of advanced breast cancer (RR 0.62, 95% CI 0.46 to 0.83), women age 39 to 49 years did not (RR 0.98, 95% CI 0.74 to 1.37). Combined data from five Swedish trials yielded a statistically significant mortality decrease of 29% ( Table 11.4 ).
| Trial | Relative Risk (95% Confidence Interval) |
|---|---|
| Malmö | 0.96 (0.68–1.35) |
| Canada | 1.08 (0.84–1.40) |
| Göteborg | 0.55 (0.31–0.95) |
| Stockholm | 0.73 (0.50–1.06) |
| Kopperberg | 0.58 (0.45–0.76) |
| Östergötland | 0.76 (0.61–0.95) |
| New York | 0.79 (0.64–0.98) |
| Edinburgh | 0.87 (0.70–1.08) |
Screening interval
The appropriate age at which to initiate screening for average-risk women is controversial, with significant differences in recommendations from government-sponsored groups and medical societies. The American College of Radiology (ACR) and the National Comprehensive Cancer Network (NCCN) recommend annual mammogram starting at age 40 years. The American College of Obstetricians and Gynecologists (ACOG) currently recommends an individualized approach to screening prior to age 50 years, and mammography every 1 to 2 years starting at age 50 years until at least age 75 years. In October 2015 the ACS revised their guidelines with regards to mammography screening in average-risk women. The guidelines now advocate the commencement of annual mammography at age 45 years until age 54 years. Thereafter, from age 55 years onwards, biennial mammograms are recommended as long as one’s overall health is good, or when one’s life expectancy is 10 years or longer. While the ACS does not recommend routine mammography screening between the ages of 40 to 44 years, they state that women should have the opportunity to initiate screening and that these decisions should be individualized. Table 11.5 lists recommendations by society.
| Age (Years) | |||||
|---|---|---|---|---|---|
| 40–45 | Individualize | Individualize | 1 year | 1 year | Individualize |
| 45–50 | 1–2 years | 1 year | 1 year | 1 year | 2 years |
| 50–55 | 1–2 years | 1 year | 1 year | 1 year | 2 years |
| 55–74 | 1–2 years | 1–2 years | 1 year | 1 year | 2 years |
In 2015 the Breast Cancer Surveillance Consortium reviewed mammography screening from 15,440 patients age 40 to 85 years who had either annual or biennial imaging. Premenopausal women diagnosed at the longer interval screening time point had more advanced tumors, stage IIB or higher, than those screened annually. Among women who were postmenopausal and not taking HRT, there was no difference in the proportions of stage IIB or higher tumors regardless of whether they were screened at 1 or 2 years. This suggests that menopausal status rather than age may be a better factor to consider when determining mammographic screening intervals.
The USPSTF published its updated screening recommendations in 2016. After reviewing the evidence regarding the efficacy of BSE, CBE, and mammography in reducing breast cancer mortality, it recommends starting mammography at age 50 years and performing biennial screening. They advise that although there may be an improvement in breast cancer mortality with screening under age 50 years, the mortality benefit is smaller compared to older women. Additionally, the task force cites the risk of false-positive mammography results (up to 56%) as well as pain, anxiety, distress, and other psychologic responses as potential harms. Therefore, they recommend individualized, shared decision making that weighs personal acceptability of the risk/benefit ratio to inform the decision of whether to initiate screening prior to age 50 years. For some women with an elevated lifetime risk of breast cancer, annual screening mammography may commence earlier than age 40 years ( Table 11.6 ).
| Condition | Timing of Annual Mammography |
|---|---|
| Lobular cancer in situ or breast cancer diagnosis | At time of diagnosis |
| First-degree relative with premenopausal breast cancer | 10 years earlier than relative’s age at diagnosis, but not younger than 30 years |
| Mantle irradiation for Hodgkin’s disease | Eight years after completion of radiation therapy |
| BRCA1 or BRCA2 mutation | Age 30 years or individualized if earliest onset in the family is <25 years. Annual MRI is recommended beginning at age 25 years |
Breast imaging reporting and data system
A lack of uniformity in mammography terminology and reporting often led to confusion regarding the malignant nature of a lesion and prompted the implementation of the Mammography Quality Standards Act in 1994. This act requires that mammography facilities monitor the results of their breast cancer detection programs, including the number of recommended biopsies as well as the size, number, and stage of cancers detected. The ACR developed a standardized classification system, called the Breast Imaging-Reporting and Data System (BI-RADS), to report mammographic findings in 1995 ( Table 11.7 ). Each of its categories leads to a fixed assessment and specific management recommendations. Additionally, associated findings, such as skin or nipple retraction, skin thickening, skin lesions, axillary adenopathy, and the presence of architectural distortion are also reported.
| BI-RADS Category | Assessment |
|---|---|
| Assessment is Incomplete | |
| 0 | Need additional imaging evaluation and/or prior mammograms for comparison |
| Assessment is Complete: Final Categories | |
| 1 | Negative |
| 2 | Benign finding(s) |
| 3 | Probably benign finding; initial short-interval follow-up suggested |
| 4 a | Suspicious abnormality; biopsy should be considered |
| |
| |
| |
| |
| 5 | Highly suggestive of malignancy; appropriate action should be taken |
| 6 | Known biopsy; proven malignancy; appropriate action should be taken |
a By subdividing Category 4 into 4A, 4B, and 4C, it is encouraged that relevant probabilities for malignancy be indicated within this category so the patient and her physician can make an informed decision on the ultimate course of action.
While the estimated rate of malignancy for the BI-RADS categories are between 0% and 2% for category 3, and 98% or greater for category 5, category 4 includes more variability. Liberman and colleagues and Orel and colleagues have reported category 4 malignancy rates as approximately 30%.
The ACR Task Force has published a fifth edition of the BI-RADS classification system, in the form of a BI-RADS Atlas, that includes BI-RADS classifications for ultrasonography and MRI. The new edition of BI-RADS includes the previously mentioned classification of patterns of breast density for uniform reporting in mammogram reports. Category 4 is divided into three parts based on the pre-biopsy risk for malignancy of the lesion—4A (low suspicion), 4B (moderate), and 4C (high)—to better estimate pre-biopsy risk of malignancy. Using this new classification, retrospective studies have estimated the risk of malignancy to be 6% (4A), 15% (4B), and 53% (4C).
Diagnostic mammography
Abnormalities found on mammographic screening may need further evaluation with additional mammography views or other imaging modalities, such as ultrasound or MRI. In some screening programs, the mammograms are reviewed by the radiologist as they are performed, and if additional views are needed, they are performed on the same day. In other programs, if additional studies are required, the patient is called back for them later. Studies have reported “call-back” frequencies ranging from 5% to 11%.
Mammographic lesions
A “mass” is defined as a space-occupying lesion seen in two different projections. If a potential mass is seen on only one view, it is called a “density” until its three-dimensionality is confirmed. A description of the shape and the margins of the lesion are also necessary. Irregular shape and spiculated borders are often associated with pleomorphic calcifications and higher rates of malignancy. This fine, discontinuous linear pattern of calcifications suggests irregular filling of a lactiferous duct with abnormal cells.
Microcalcifications
The BI-RADS lexicon describes calcification morphology (shape) and distribution. Calcifications may be scattered or clustered, coarse or fine, and old or new. Comparison with prior mammograms is often necessary ( Table 11.8 ).
| Morphology | Description/Associated Lesion |
|---|---|
| Typically Benign | Includes skin (lucent-centered) Vascular (parallel tracks) Coarse “popcorn-like” (fibroadenomas) Large rod-like (secretory disease) Eggshell or rim (fat necrosis) Milk of calcium (within tiny cysts) Dystrophic (after trauma or irradiation) |
| Suspicious | Amorphous Coarse heterogenous Fine pleomorphic Fine linear or fine linear branching |
| Distribution | Diffuse Regional Grouped Linear Segmental |
Digital mammography
Digital image processing permits manipulation of image contrast and may enhance subtle contrast differences compared to standard films. In January 2000 the General Electric Senographe 2000D was approved by the FDA. Since its introduction, population-based screening trials that compare screen-film and full-field digital mammography have been conducted. In the Oslo I Study conducted in Norway, full-field digital and screen-film mammography were performed in 3683 women age 50 to 69 years. The investigators did not find a statistically significant difference in cancer detection rates between the two modalities. The Oslo II Study yielded similar results in cancer detection rates.
In Canada and the United States, 49,528 women were enrolled in the Digital Mammographic Imaging Screening Trial (DMIST). All participants underwent both digital and film mammography in random order. The investigators noted that, although the diagnostic accuracy of digital and film mammography was similar, the accuracy of digital mammography was better in women under age 50 years, women with radiographically dense breasts, and premenopausal or perimenopausal women. Thus, in women who met those criteria, the investigators recommended digital mammography.
Digital mammography is now standard of care. It can be also combined with computer-aided detection (CAD), which is a process designed to analyze mammographic images for suspicious areas, and aid in the early detection of breast cancer. Digital breast tomosynthesis (DBT), or 3-D mammography, is thought to improve cancer detection rates and the positive predictive value of mammography. Rather than taking two standard views, DBT comprises multiple reduced-dose mammograms at various angles, which are then digitally reconstructed to create 3-D images. Cancer detection rates were almost twice as high with the addition of DBT to standard mammography compared to standard mammography alone in a randomized control trial (8.6 vs. 4.5 per 1000; RR 1.9, [95% CI 1.3 to 2.7]). While the addition of DBT to standard mammography is associated with increased radiation doses, the radiation dose is still within the FDA limits for mammography. DBT techniques that can also create 2-D images are associated with more comparable rates of radiation relative to standard mammography.
Contrast-enhanced digital mammography is an emerging technique that can identify morphologic abnormalities as well as tumor vascularity because of the addition of iodinated contrast. Its high sensitivity, relative low cost, and shorter protocol times have made it an attractive alternative to MRI for some indications. As a result, it has been used for problem solving in cases of inconclusive screening mammograms, determining disease extent, monitoring response to neoadjuvant systemic therapy, and intermediate- and high-risk screening.
Breast ultrasound and magnetic resonance imaging
Breast ultrasonography can distinguish between solid and cystic masses in the breast, and may help with the evaluation of a focal mass identified on a mammogram or clinical breast examination. It can also help facilitate breast biopsies. Because of its low specificity and inability to detect microcalcifications, it is not thought to be a good single-agent modality for screening; however, it can complement mammography in young women with dense breasts, as increased density limits the sensitivity of the mammogram.
MRI has a high sensitivity for the detection of breast cancer, even among women with dense breasts. Screening studies to date have been done in high-risk patients, primarily those at risk because of known or suspected BRCA mutations or a family history of breast cancer. In the FaMRIsc multicenter, randomized, controlled trial, 1355 women with familial risk of breast cancer were allocated to MRI or mammography. Not only were more breast cancers detected in the MRI group than in the mammography group (40 vs. 15, P = .0017), but MRI screening was found to detect cancers at an earlier stage than mammography (median size 9 mm vs. 17 mm, P = .010; node-positivity P = .023).
While MRI is more sensitive, it is less specific in the detection of invasive carcinoma among high-risk women in both prospective and retrospective studies. In a systematic review of 11 prospective studies comparing MRI and mammographic screening, MRI sensitivity was 75% (95% CI 62 to 99). The addition of mammography increased the sensitivity to 84% (95% CI 70 to 97). The specificity of mammography (98.5%, 95% CI 97.8 to 99.2) exceeded specificity of MRI (96.1%, 95% CI 94.8 to 97.4). Two prospective observational studies have suggested a breast cancer mortality benefit for breast MRI screening for high-risk women, but small numbers, selection bias, and treatment differences limit the ability to draw definitive conclusions.
The ACS has issued guidelines on the use of annual MRI for screening in high-risk women. A guideline panel was assembled to review evidence and develop new recommendations for women at different levels of risk. Based on this panel screening, annual MRI and mammogram are recommended for women with an approximately 20% to 25% or greater lifetime risk of developing breast cancer. This includes women with strong family history of breast and/or ovarian cancer, untested first-degree relatives of BRCA mutation carriers, and women with a history of mantle radiation between the ages of 10 and 30 years. Screening MRI is also recommended for patients with Li-Fraumeni or Cowden syndrome and their first-degree relatives.
MRI can be useful in treatment selection for newly diagnosed breast cancers, particularly in evaluating tumor multifocality and disease span in women wanting to pursue breast conservation. However, it has not been shown to increase the likelihood of negative margins or the need to convert from lumpectomy to mastectomy. MRI can also be used to measure response to neoadjuvant chemotherapy and assess for an occult primary breast tumor in patients who present with axillary nodal metastases. There is insufficient evidence to recommend for or against screening with MRI for women with a personal history of breast cancer, atypical hyperplasia, or extremely dense breasts on mammography.
Diagnostic evaluation
Palpable mass
The work-up of a patient with a dominant mass should include a bilateral mammogram and ultrasound. In addition to gaining valuable information about the characteristics of the mass, a secondary purpose in this setting is to screen the normal surrounding breast and the contralateral breast for non-palpable mammographic abnormalities (densities or calcifications). Evaluation of a palpable mass is necessary even in the absence of mammographic findings, as imaging occult masses can be malignant.
Fine-needle aspiration or biopsy
FNA can be extremely useful in providing a cytologic analysis of a palpable breast mass. Many palpable thickenings and all dominant masses should be considered for FNA because, in addition to providing tissue for histologic evaluation of solid masses, it can diagnose and treat simple cysts and provide cellular material for cytologic analysis. The FNA should be performed after radiologic examination because the resultant hematoma could mask an underlying abnormality.
The breast is prepped with alcohol; with the physician facing the patient, the lesion is stabilized with the physician’s opposite hand. Usually, a 21-gauge or 25-gauge needle on a 10 cc syringe is used. Approximately 3 cc of air is aspirated into the syringe to facilitate expulsion of the contents onto the slide following the procedure. The needle is introduced into the lesion, and suction is applied on the syringe. If the mass is cystic, the fluid is completely evacuated, and the lesion should completely disappear. The syringe is withdrawn, and the fluid is discarded if it is serous and non-bloody. The patient should return in 4 to 6 weeks for re-examination.
If the lesion encountered is not cystic or suspected to be solid, an FNA biopsy can be performed in the same manner. After insertion into the lesion, multiple passes (10 to 15) through the lesion with changes in direction allow extensive sampling and create a “feel” for the mass (carcinomas are usually hard and gritty). The goal of sampling is to obtain material in the hub of the needle, not to fill the syringe. Care should be taken to release the suction before withdrawing the needle to prevent aspiration into the syringe. The sample is then ejected onto a glass slide, gently smeared with another slide, and placed in sterile jars containing 95% ethanol for transport to the cytology laboratory. Alternatively, it can be placed in a specimen jar containing cytofixative. The needle should be removed from the syringe, the medium aspirated into the syringe, the needle replaced, and the medium then ejected into the jar.
An FNA requires a cytopathologist experienced in breast pathology. While the false-positive rate of FNA is 1% or less, in contrast, the false-negative rate can range from 3% to 35% depending on the expertise of the aspirator and cytopathologist, the size of the lesion, the location within the breast, and the cellular composition of the lesion. Negative findings in the presence of a suspicious mass should not preclude further diagnostic evaluation. Lesions that have discordant imaging-pathologic correlation require surgical biopsy. Any mass remaining after aspiration of a cyst should be excised. Similarly, a cyst that recurs in the same location after one or two aspirations should be excised.
Patients with palpable solid masses can have a core needle biopsy of the mass in the office with use of a spring-loaded Tru-cut 14-gauge or similar biopsy device. The breast is prepped sterilely, and a local anesthetic is used to infiltrate the skin. A small nick is made in the skin with a scalpel to accommodate the biopsy instrument. A core biopsy of the solid mass is obtained. The instrument has a “firing range” and, therefore, should be kept parallel to the chest wall to avoid penetrating trauma and pneumothorax. The specimen is placed in formalin and sent for pathologic examination.
Image-guided percutaneous breast biopsy
Percutaneous biopsy methods differ with respect to the method of imaging guidance and the tissue-acquisition device used. The use of image-guided percutaneous biopsy has advantages over surgical excision for the diagnosis of breast lesions. It is less invasive, and because less tissue is removed, it will result in less scarring on subsequent mammograms. Regardless of whether the diagnosis is benign or malignant, the patients who have percutaneous biopsies will undergo fewer operations. The choice of which image-guided modality to use depends on the lesion characteristics. Ultrasound is the preferred modality given its ease of use and cost-effectiveness. The use of ultrasound imaging for percutaneous biopsy of lesions seen on ultrasound has several advantages: it requires no specialized equipment, generates no radiation exposure, and has the ability to sample areas that may be inaccessible with stereotactic biopsy (such as the axilla).
Stereotactic core needle biopsy uses specialized mammography equipment to calculate the location of a lesion in three dimensions and remains the best option for patients with calcifications. Biopsy may be performed with the patient in a prone position on a dedicated table or with the patient sitting in an upright unit. Some patient and lesion characteristics may pose technical challenges, including morbid obesity, difficulty remaining prone for the 20- to 40-minute procedure duration, and superficial lesion location. A vague asymmetric density or diffuse group of widely separated calcifications may also present difficulties.
An automated core needle or directional vacuum-assisted biopsy (VAB) probe is used to obtain multiple tissue specimens for pathologic analysis. Sensitivity of core needle biopsy is estimated to be 70% to 100% with a specificity of 85% to 100%. Studies have shown 99% accuracy with a 14-gauge needle obtaining five specimens. Specimen radiography of biopsies targeting breast microcalcifications should be routinely performed to determine whether the calcifications were obtained. Clips are routinely placed in the breast to mark the biopsied area.
While VAB probes are typically more expensive than spring-loaded devices, they have been associated with false-negative rates as low as 0.45% with 11-gauge needles. They are able to collect multiple samples with a single pass; however, their heavier weight makes them more practical for use with stereotactic and MRI-guided biopsies than for palpation or ultrasound-guided biopsies that require more manipulation of the biopsy device. For ultrasound- and palpation-guided biopsies, spring-loaded biopsy devices provide a low cost and light-weight alternative with real-time visualization of the lesion.
Ultrasound-guided biopsy
The use of ultrasound imaging for percutaneous biopsy of lesions seen on ultrasound has several advantages: it requires no specialized equipment, generates no radiation exposure, and can sample areas that may be inaccessible with stereotactic biopsy (such as the axilla).
Tissue-acquisition devices
Available tissue-acquisition devices include fine needles, automated core needles, directional vacuum-assisted probes, and biopsy cannulas. Excellent results have been obtained using the 14-gauge automated needle for biopsy of masses under ultrasound or stereotactic guidance. Most centers use larger tissue-acquisition devices instead of fine needles because of accuracy of tissue diagnosis when a larger volume of tissue is obtained. Compared with the automated needle, the vacuum device acquires larger samples of tissue, has a higher frequency of retrieval of calcifications, and may provide more accurate lesion characterization. Accurate placement of a localizing clip through the biopsy probe is necessary to facilitate subsequent localization if needed.
Excisional biopsy
Needle localization is a technique that allows surgical excision of a lesion that is non-palpable, which is the historic standard of care. The technique uses a hook-wire system to target the lesion, and image guidance can be provided by mammogram, ultrasound, or MRI. In mammography-guided needle localization, coordinates of the lesion are obtained by placing the breast in an alphanumeric grid. The needle is inserted and, when adequate placement is noted, the hook wire is deployed, and the needle removed. Two mammographic views are then obtained.
The mammography films are available intraoperatively and show the relationship between the lesion and localizing hook. Excision with needle localization allows the surgeon to minimize the amount of breast tissue removed by following the needle to the targeted lesion. For patients with wide disease spans, placement of multiple wires can be considered to “bracket” the entire disease span and delineate the area that requires resection. After removal, a specimen radiograph is obtained to ensure that successful removal of the lesion has been performed. Radiologists and surgeons experienced in needle localization and excisions report only 0.2% to 0.3% of lesions missed with this approach.
While some studies report margin clearance rates approaching 90%, localization techniques that do not require wire traversing the breast have been explored. A variation of this approach to excising non-palpable lesions is the use of radioactive seed localization (RSL), which involves placement of a small titanium Iodine-125-containing seed (4 by 0.8 mm) in a similar fashion to the needle localization. The use of an intraoperative handheld gamma probe allows for accurate excision on the lesion, which is similarly x-rayed to ensure retrieval of the lesion and the radioactive seed. It has advantages over needle localization as the placement of the radioactive seed can be performed several days before the procedure, which can improve flexibility with regards to scheduling; however, stringent radiation safety protocols must be implemented.
Neither randomized controlled trials nor observational studies have demonstrated a significant difference in inadequate margin rates, re-excision rates, or specimen size between traditional needle localization and RSL; however, shorter operative times and increased ease of technique have been reported for RSL compared to needle localization. Recent studies have also indicated potential benefits of RSL, including lower costs, more efficient scheduling, and shorter wait times for patients, leading to increased adoption of RSL. A variety of non-wire localization devices have become available in recent years, and appear to have similar rates of margin clearance and do not require the same radiation safety oversight. Because they are relatively new, outcomes data for these devices are limited.
Surgical excision/breast biopsy
Excisional biopsy can be performed on an outpatient basis under local anesthesia with monitored sedation in most patients. It is important to choose the appropriate incision to access the target lesion. The specimen should be adequately oriented for margin analysis by the pathologist and sent for the appropriate markers, such as ER and progesterone receptor (PR) status, and human epidermal growth factor receptor 2 (HER2) status. Orientation of the specimen is important because the biopsy is both diagnostic and therapeutic, and a re-excision of an involved margin may be required.
The incision should be closed with fine suture material and with a subcuticular closure. Hemostasis needs to be confirmed before closing. No particular immobilization is required, but a brassiere with good support is recommended to minimize hematoma, induration, and discomfort.
Epidemiology of breast cancer
Table 11.9 lists major risk factors for breast cancer.
|
Risk factors and assessment
Age
The most significant risk factor for breast cancer is increased age.
Family history and hereditary risk
Approximately 10% of newly diagnosed breast cancers are thought to be hereditary. BRCA1 and BRCA2 gene mutations are the most commonly identified mutations, but mutations in additional genes, including PALB2, ATM, CHEK2, as well as others, have also been associated with increased risks of breast cancer. One of the most characteristic features of hereditary breast cancer is its tendency to manifest at a young age.
Even outside of a pathogenic mutation, family history informs personal risk of breast cancer. Prospective data from the Nurses’ Health Study noted that the risk of breast cancer doubled among women with a mother diagnosed with breast cancer before age 40 years or with a sister diagnosed with breast cancer. The risk decreased with advanced maternal age; however, it remained elevated even with maternal diagnosis at age 70 years (RR 1.5; 95% CI 1.1 to 2.2). In the population of middle-aged women, the authors found that only 2.5% of breast cancer cases were attributed to a positive family history.
In the large ACS Cancer Prevention Study II, the authors evaluated the association between fatal breast cancer and family history. They found that a family history of breast cancer in a mother or sister was significantly related to fatal breast cancer risk after multivariate analysis. Association was significantly modified by age with a risk ratio of almost 5 in women younger than age 40 years at enrollment compared with 1.28 in women at age 70 years or older.
Personal history
A patient’s history of prior breast biopsy is important. Although the number of breast biopsies does not increase a woman’s lifetime risk of breast cancer, certain pathologic entities do increase risk. Atypical ductal or lobular hyperplasia and LCIS are considered markers of increased risk of developing invasive breast cancer. A personal history of breast cancer increases the risk for development of ipsilateral recurrence or a contralateral breast cancer, which can approach as high as 10% at 10 to 15 years posttreatment.
Reproductive history
Early menarche, late menopause, and nulliparity are thought to be risk factors for breast cancer. The age of a woman’s first live birth is also thought to be an RR factor for breast cancer.
An early age at a woman’s first live birth is often associated with a lower risk for breast cancer. For a woman younger than 19 years of age at first full-term birth, it is predicted that there is a 50% reduction in the risk of breast cancer compared with that of a nulliparous woman. In contrast, if the first full-term pregnancy occurs at age 30 to 34 years, the risk of breast cancer is approximately the same as that noted in nulliparous women. After age 35 years, pregnancy in women is associated with an increased risk compared with that of a nulliparous woman. Pregnancies that are not full term do not show this protection.
Some studies have suggested that the time interval between the onset of menarche and the first full-term pregnancy is an important factor informing the endocrine milieu; however, data regarding this “estrogen open window hypothesis” have been conflicting. Breast feeding has also been associated with a reduced risk of breast cancer. Studies in the United States, however, have suggested that breast feeding has only a weak protective effect, and these studies do not demonstrate an association between reduction in risk and duration of breast feeding.
Early menarche and late menopause have also been investigated as potential risk factors for breast cancer, given the longer exposure to the menstrual hormonal milieu. In one large international case-control study, it was noted that for each 2-year delay in the onset of menstruation, the risk for breast cancer was reduced by approximately 10%. Additionally, for every 5-year increase in age at menopause, the risk for breast cancer increased by approximately 17% and was seen 10 to 20 years after menopause. Data from observational studies also demonstrate that bilateral oophorectomy preceding natural menopause is associated with a reduced risk of breast cancer compared to later natural or artificial menopause.
Exogenous hormone use
The role of exogenous estrogens in the promotion of breast cancer remains controversial. Studies of OCPs and HRT have yielded conflicting results. Studies of HRT and breast cancer risk indicate that women who are currently using HRT are at increased risk for breast cancer development. A large 2019 meta-analysis of 108,647 postmenopausal women performed by the Collaborative Group on Hormonal Factors in Breast Cancer demonstrated that every HRT type, except vaginal estrogens, was associated with an increased risk of breast cancer. This risk increased proportionally with the duration of use and was higher for women taking combination HRT compared to those taking estrogen alone. After stopping HRT, the risk of developing breast cancer remained elevated for over 10 years. Of note, multiple studies have shown that patients who develop breast cancer while taking HRT have smaller, less-aggressive cancers, and a lower risk of death from breast cancer.
The WHI performed two randomized control trials of HRT. One study randomized 16,609 women with an intact uterus to receive either combination HRT (0.625 mg/day conjugated equine estrogens and 2.5 mg/day medroxyprogesterone acetate) or a placebo between 1993 and 1998. The planned duration of the trial was 8.5 years; however, after a mean follow-up of 5.2 years, the data and safety monitoring board of the committee recommended halting the trial because the incidence of invasive breast cancer had exceeded the stopping boundary that had been set at the initiation of the trial. The reported increased risk of breast cancer hazard ratio was 1.25 (95% CI 1.00 to 1.59). HRT was also associated with an increased risk of coronary heart disease (hazard ratio 1.29; 95% CI 1.02 to 1.63) and stroke (hazard ratio 1.41; 95% CI 1.39 to 3.25). The beneficial effects of HRT included a decreased risk in colorectal cancer (hazard ratio 0.63; 95% CI 0.43 to 0.92) and hip fracture (hazard ratio 0.66; 95% CI 0.45 to 0.98).
The other WHI trial randomized 10,739 postmenopausal women with prior hysterectomy to receive 0.625 mg/day of conjugated equine estrogen or placebo between 1993 and 1998. In February 2004 the National Institutes of Health decided to terminate the intervention phase of the estrogen-only study, which had been scheduled for a close-out interval of October 2004 to March 2005. With an average follow-up of 6.8 years, there was an increased risk of stroke (hazard ratio 1.39; 95% CI 1.10 to 1.77), a decreased risk of hip fracture (hazard ratio 0.61; 95% CI 0.41 to 0.91), and no effect on coronary heart disease incidence (hazard ratio 0.91; 95% CI 0.75 to 1.12). The investigators noted a possible reduction in breast cancer risk (hazard ratio 0.77; 95% CI 0.59 to 1.01) that warrants further investigation.
The updated analysis published in 2013 found that many of these risks dissipated post-intervention. Notably, neither combination HRT nor conjugated estrogens alone affected all-cause mortality, and the quality-of-life outcomes were mixed with both regimens. Notably, elevated breast cancer risk persisted in the estrogen with progestin group (hazard ratio 1.28; 95% CI 1.11 to 1.48), whereas conjugated estrogen alone was not associated with an increased breast cancer risk (hazard ratio 0.79; 95% CI 0.61 to 1.02). This suggested that while HRT should not be prescribed for perceived health benefits, a short course of estrogen alone to improve menopause symptoms is unlikely to increase the risk of breast cancer.
ACOG stresses the importance of shared decision making and addressing the reasons for initiating or continuing HRT. HRT is no longer recommended to prevent heart disease in healthy women (primary prevention) or to protect women with preexisting heart disease (secondary prevention). Additionally, it is no longer recommended solely for prevention of osteoporosis. HRT is highly effective in treating vasomotor symptoms with limited effective alternative therapies. In this setting, short-term use (less than 5 years) can be considered because data on short-term use do not show an increased association with breast cancer.
In the past, most studies addressing OCP-use and breast cancer risk concluded that there was a significant increase in risk associated with OCP-use. However, most studies regarding OCP-use and breast cancer risk have demonstrated little association with breast cancer incidence rates. In women who have used OCPs for extended periods (greater than 10 years), a minimal, non-significant increase in breast cancer cases has been reported, seen most commonly in the group of women who began using OCPs at a young age (younger than 20 years). Past or present use of OCPs at the time of diagnosis of breast cancer does not affect mortality from breast cancer. The presence of a family history of breast cancer does not appear to further increase the risk of breast cancer associated with either the use of OCPs or HRT.
Prior exposure to radiation therapy
Exposure to ionizing radiation, such as mantle radiation for Hodgkin’s disease, poses a risk for breast cancer. This is noted 7 to 10 years after completion of radiation therapy, and the risk of breast cancer associated with radiation exposure decreases with increasing age at the time of exposure. In these women, the cumulative probability of developing breast cancer at age 40 years approaches 35%. The NCCN guidelines recommend the initiation of breast cancer screening within 8 years of radiation therapy.
Other factors
Breast cancer is more frequent in Jewish women than in non-Jewish women. Women of Ashkenazi Jewish descent have a 1 in 40 (2.5%) risk of carrying a mutation in BRCA1 and BRCA2 genes, thus accounting for the increased risk in these women. While Black women have a lower incidence of breast cancer compared to Caucasian women, breast cancer mortality is higher among Black women. Many studies have endeavored to elucidate the reasons, both socioeconomic and biologic, that underpin these disparities.
Asian women have a lower incidence of breast cancer. Japanese women show lower rates of breast cancer than Caucasian women. Although postmenopausal breast cancer is less common in Japanese women who have migrated to Western countries than among the general populations of these countries, the incidences of breast cancer in these women approach that of White women after two or three generations. The Western diet, with its increased intake of animal fat, has been implicated in these studies.
Alcohol consumption has been reported to increase breast cancer risk in a dose-related manner. Women who drink approximately one drink per day have a slightly elevated risk of breast cancer compared to those who do not consume alcohol, and this risk further increases with moderate-to-high alcohol consumption (two to five drinks per day).
Relative risk and risk-assessment models
RR is a ratio that depicts the likelihood over time of an event’s occurrence in a study population relative to that in a reference population. It is often used to quantify risk factors for breast cancer. Absolute risk is a percentage that depicts the likelihood over time of the occurrence of an event. While these values are often similar when quantifying rare events, they can be quite different when assessing common events. When discussing risks with individual patients, it is best to discuss absolute risk rather than RR.
Several models exist to estimate a woman’s lifetime risk of breast cancer. The Gail model, developed for use in the National Surgical Adjuvant Breast and Bowel Project (NSABP P-1) Breast Cancer Prevention Trial, is available from the National Cancer Institute and provides a measurement of absolute risk over time for breast cancer. In familial-type hereditary cases, however, it underestimates the risk of breast cancer by overlooking age at onset, bilaterality of disease among affected family members, and breast cancer in non–first-degree relatives ( Table 11.10 ).