Gestational trophoblastic disease (GTD) comprises a spectrum of neoplastic conditions derived from the placenta. Hydatidiform moles, gestational choriocarcinoma, placental site trophoblastic tumor (PSTT), and epithelioid trophoblastic tumor (ETT) are histologic diagnoses, whereas postmolar gestational trophoblastic neoplasia (GTN) is defined by clinical and laboratory criteria. The disease entities included in GTD have a wide variation in behavior, whereas GTN specifically refers to those with the potential for tissue invasion and metastases.
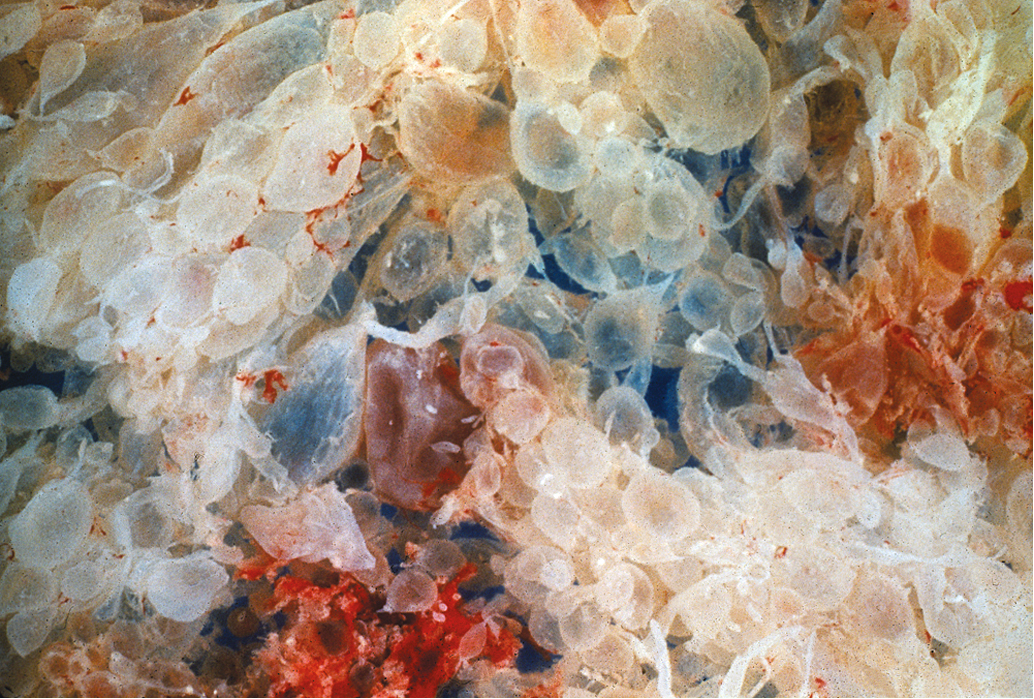
GTD has been recognized since antiquity. Individual hydropic molar villi ( Fig. 7.1 ) were sometimes identified as separate fetuses. In the early 19th century, Velpeau and Boivin recognized hydatidiform mole as a cystic dilation of the chorionic villi. In 1895, Marchand demonstrated that hydatidiform mole and, less frequently, normal pregnancy preceded the development of choriocarcinoma. In the early 20th century, Fels identified elevated chorionic gonadotropic hormones in the urine of women with hydatidiform moles.
Before the mid-1950s the prognosis for women with metastatic GTN was dismal and there were no effective treatments. In the 1940s, Hertz demonstrated that fetal tissues required large amounts of folic acid and that their growth could be inhibited by methotrexate. In 1956 Li reported the first sustained remission in a patient with choriocarcinoma treated with methotrexate.
Since that report, GTN has been recognized as the most curable gynecologic malignancy. Reasons for this include: (1) identification and development of quantitative assays for human chorionic gonadotropin (hCG) in serum and urine allowed hCG to become the prototype for tumor markers; (2) sensitivity of GTN to various chemotherapeutic agents; and (3) identification of risk factors allowing individualization of treatment, using simple chemotherapy regimens for low-risk patients and multiagent chemotherapy combined with surgery and/or radiation therapy in selected high-risk cases.
In general, GTD represents a derangement in the development of the conceptus, associated with unregulated trophoblastic proliferation and invasion, with the propensity for hematogenous metastasis in GTN. The diseases are characterized by paraneoplastic disorders from secretion of gestational hormones, most notably hCG. GTD is the only group of female reproductive neoplasms with a component of paternal genetic material (androgenic origin).
The etiology of GTD likely involves genetic abnormalities involved in fertilization. Abnormalities in differentiation and pronuclear cleavage, decidual implantation, myometrial invasion, and host immunologic tolerance are also involved. Several candidate tumor suppressor genes (e.g., DOC-2/hDab2), chromosomal loci (e.g., 7qp12-7q11,23 and 9q13,3-13,4), and oncogenes (e.g., CD9) have been implicated in the pathogenesis of GTD. Rare maternal genetic mutations have been implicated in biparental complete moles (CMs).
Epidemiology
The incidence of molar pregnancies in the United States is approximately 1 in 1500 pregnancies. Race or ethnicity, age, socioeconomic status, diet, and prior reproductive history all influence the risk. The most reliable studies suggest that the incidence of hydatidiform mole is slightly less than 1 in 1000 pregnancies in most of the world, 1.2 in 1000 in South Africa, as high as 2 in 1000 in Japan, 7 in 1000 deliveries in Malaysia and the Philippines, and possibly higher in Saudi Arabia. The reported incidence of molar pregnancies among various ethnicities and races may be biased as a result of dependence on hospital records, particularly from tertiary referral centers, rather than population-based studies. Historically, GTD was thought to occur more frequently in the Asian population; however, recent population-based studies in Japan have shown a decreasing incidence of GTD in Japan, even with a relatively homogeneous population.
Extremes of reproductive age are associated with increased risk of molar pregnancy. Based on an analysis of 2202 patients with hydatidiform moles compared with a contemporary control group including all types of pregnancy events, a Duke study found a significantly higher incidence of molar pregnancy in women younger than 15 years and older than 40 years of age. Other studies have confirmed the peak incidence of molar pregnancies at the extremes of maternal reproductive age. Some of these studies also noted an increased risk associated with increasing paternal reproductive age for CM but not for partial mole (PM).
Dietary factors associated with an increased risk of hydatidiform mole may include a low-protein diet and vitamin A deficiency, although these factors are difficult to separate from effects of lower socioeconomic class alone. Berkowitz and colleagues have suggested that a deficiency of animal fat and fat-soluble vitamin carotene is associated with increased risk of molar pregnancies. Populations with a high prevalence of vitamin A deficiency have been noted to have higher incidences of hydatidiform mole. Although carotene-rich vegetables are available in these countries, the lack of dietary fat needed for carotene absorption may result in an overall carotene-deficient condition.
Prior molar pregnancy also increases the risk of recurrent molar pregnancy. Women with a single previous molar pregnancy have more than 10 times the risk of having another molar pregnancy compared with those who have never had one. Based on data by Bagshawe and colleagues, after having had one molar pregnancy, the risk for a second was 1 in 76; after two molar pregnancies, the risk for a third was 1 in 6.5. Sand reported that the risk of a third molar pregnancy was 28%. Goldstein and colleagues noted that 9 of 1339 patients (1 in 150) had at least two consecutive molar pregnancies. Other centers have reported that the incidence of a second molar pregnancy is as high as 1 in 50 women with one prior molar pregnancy. Up to half of all patients with more than two repetitive moles may be associated with maternal germline mutations in NLRP7 or less frequently KHDC3L genes.
With recurrent molar pregnancies, there is also an apparent increased risk of developing malignant GTN. Patients with consecutive molar pregnancies may have subsequent normal pregnancies. Berkowitz and colleagues noted that four of their patients with consecutive molar pregnancies later had full-term pregnancies. Lurain noted that five of eight patients with consecutive molar pregnancies later had normal full-term pregnancies.
In a case-controlled study from Baltimore, factors found to be associated with GTN included professional occupation, history of prior spontaneous abortions, and the mean number of months from antecedent pregnancy to the index pregnancy. Contraceptive history, irradiation, ABO blood groups, and smoking factors of the male partner were not associated with development of GTN. Among PMs, no clinical factors such as gravidity, parity, age, uterine size, gestational age at diagnosis, or hCG levels at presentation were associated with development of GTN.
Hydatidiform mole
Two distinct forms of molar pregnancies, CM and PM, are currently recognized ( Table 7.1 ). Cytogenetic studies have conclusively demonstrated that they are completely separate entities. Despite the cytogenetic, pathologic, and clinical differences outlined in Table 7.1 , the clinical management of patients with CMs and PMs is similar.
| Complete Mole | Partial Mole | |
|---|---|---|
| Karyotype | 46, XX or 46, XY—all paternal | 69, XXX or 69, XXY—2⁄3 paternal and 1⁄3 maternal |
| Pathology | ||
| Fetus | Absent | Sometimes present |
| Amnion, fetal RBCs | Absent | Usually present |
| Villous edema | Diffuse, marked | Focal, variable |
| Trophoblastic proliferation | Diffuse, slight to severe | Focal, slight to moderate |
| p57 staining | Negative | Positive |
| Clinical Presentation | ||
| Clinical diagnosis | Molar gestation | Missed abortion |
| Uterine size | Large for dates (30%–50%) | Small for dates |
| Serum hCG >100,000 mIU/mL | >60% | Rare |
| Theca-lutein cysts | 6%–9% | Rare |
| Medical complications | 1%–20% | Rare |
| Postmolar GTN | 6.8%–20% | <5% |
Cytogenetics and pathology
Molar pregnancies consist of CMs and PMs. In most series, CMs and PMs comprise approximately 70% and 30% of molar pregnancies, respectively.
Complete hydatidiform moles are completely derived from the paternal genome (see Table 7.1 ). Most frequently, CMs have a diploid 46, XX genome with exclusively androgenetic markers on the chromosomes, implying duplication of a haploid sperm chromosomal complement in the fertilized ovum. However, approximately 5% to 10% of CMs have a 46, XY androgenetic genome, indicating that dispermic fertilization occurs in approximately 10% to 20% of CMs. The mechanism for exclusion of the maternal polar body from the nucleus of the fertilized ovum is unknown. No 46, YY moles have been reported, but aneuploid karyotypes, such as tetraploidy, have been associated with CM. The fetus fails to develop a circulatory system in CM, because fetal red blood cells are not observed in sections of the villi.
In contrast, PMs are composed of both paternal and maternal genomes (see Table 7.1 ). Usually, two paternal haploid sets of chromosomes are combined with one maternal set, resulting in complete triploidy (see Fig. 7.1 ). The 69, XXX karyotype is most common , but some PMs have a 69, XXY karyotype, implying dispermic fertilization. Sometimes other aneuploid karyotypes are observed in PM, but none with duplication of the Y chromosome. All karyotypes reported for PM feature a maternal haploid set and multiples of the paternal chromosomal complement. Gross or histologic evidence of fetal development such as fetal red blood cells is a feature of PM that differentiates it from CM.
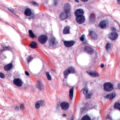
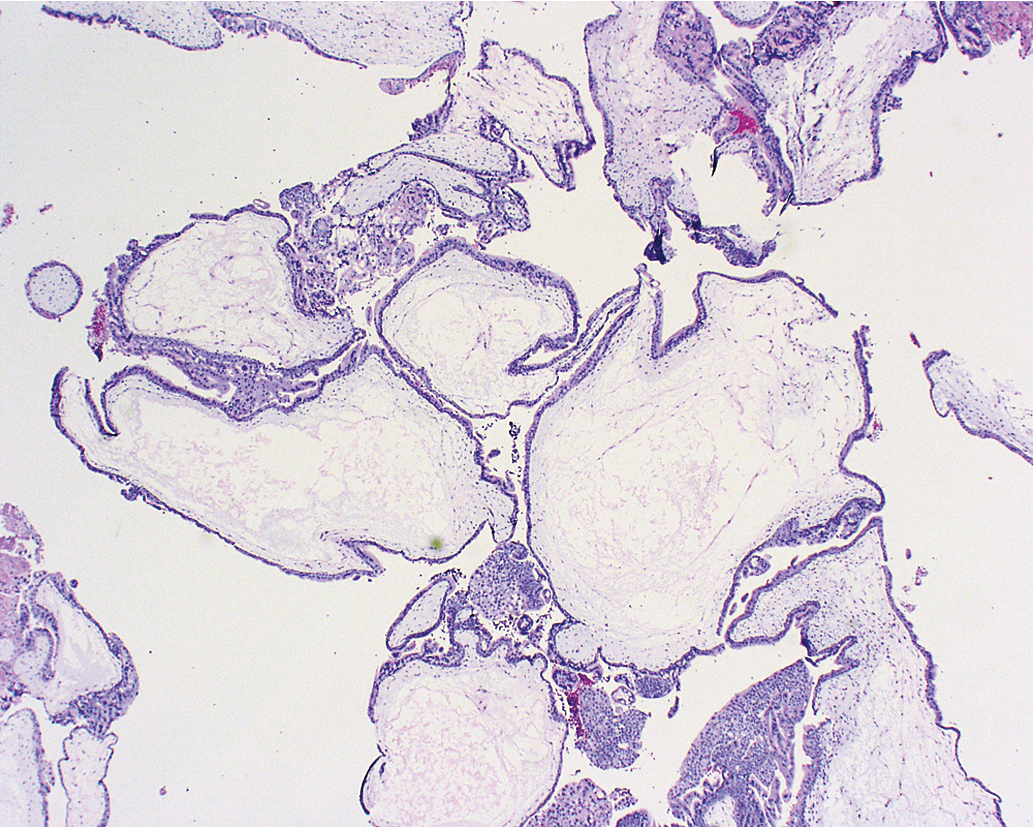
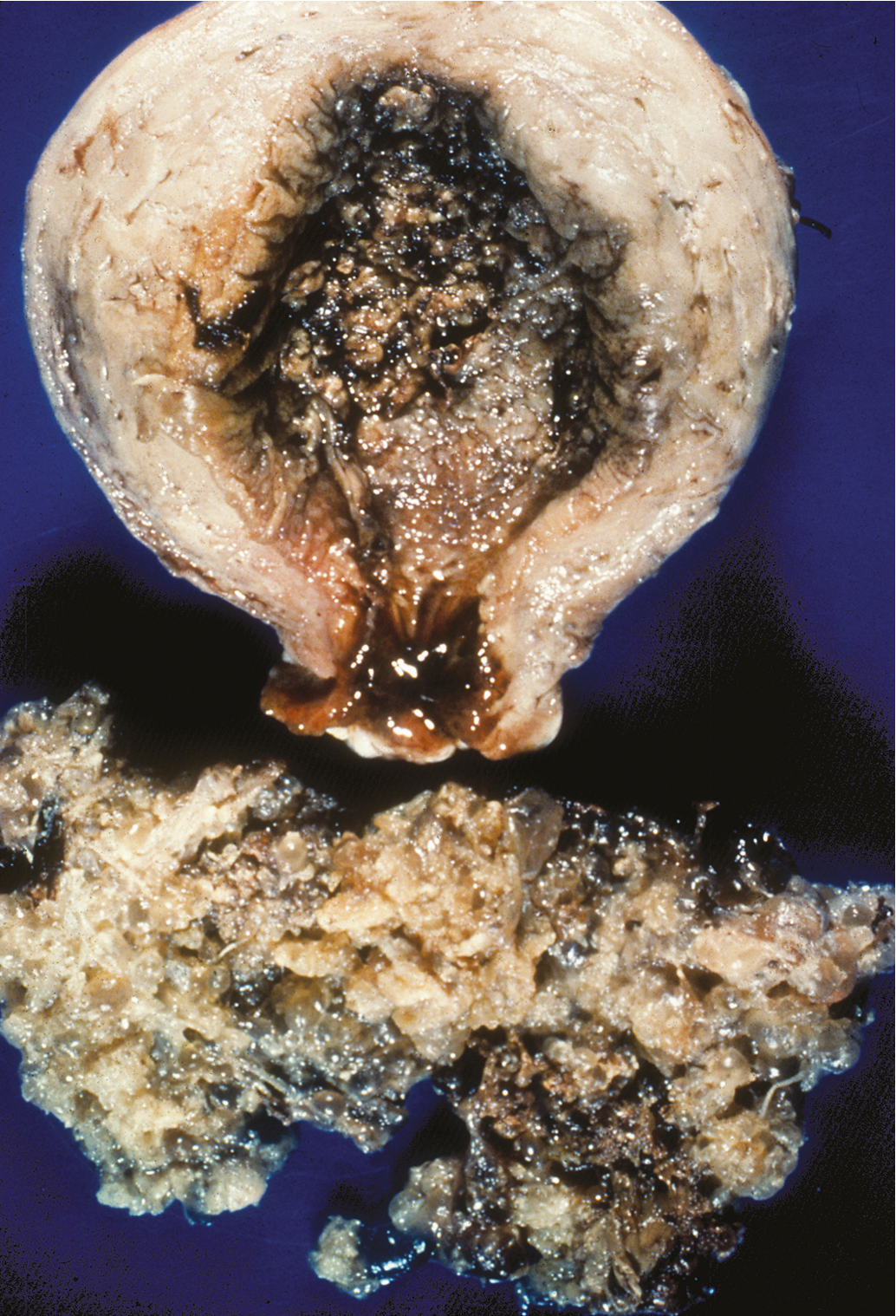
Other histopathologic features of CM and PM differ. CMs have diffuse villous edema ( Fig. 7.2 ), often with central cisterna formation ( Fig. 7.3 ). Trophoblastic proliferation is usually diffuse ( Fig. 7.4 ) but may vary in extent. Histologic evidence of a fetus is lacking. Histologic features of PM can be subtle, and many PMs are probably under diagnosed erroneously as spontaneous abortions. A PM exhibits focal, varying degrees of hydropic villi with scalloping and trophoblastic inclusions within the villi ( Fig. 7.5 ). Focal trophoblastic proliferation is usually subtle compared with CM. Fetal red blood cells are observed within vessels of the villi ( Fig. 7.6 ), and sometimes a fetus can be grossly identified. The fetus is usually nonviable and rarely survives beyond 20 weeks of gestation. The difference in the amount of trophoblastic proliferation between CM and PM is related to their different clinical presentations. Malignant GTN occurs in 2.5% to 5% of patients with PM compared with approximately 6.8% to more than 20% after evacuation of a CM mole (see Table 7.1 ).
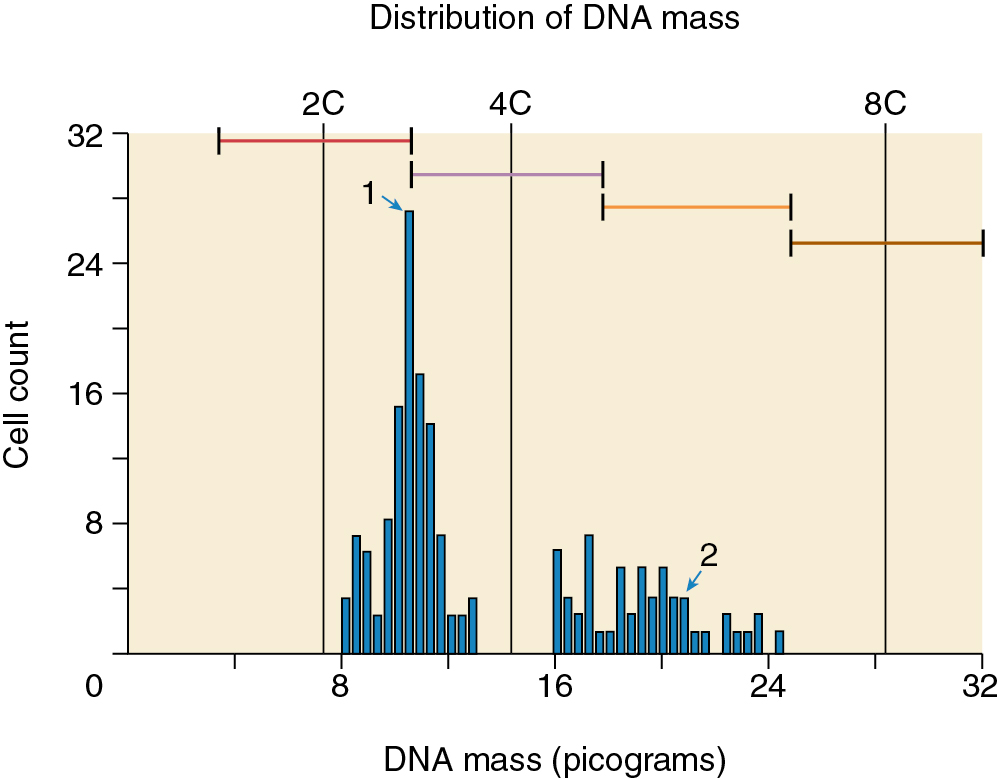
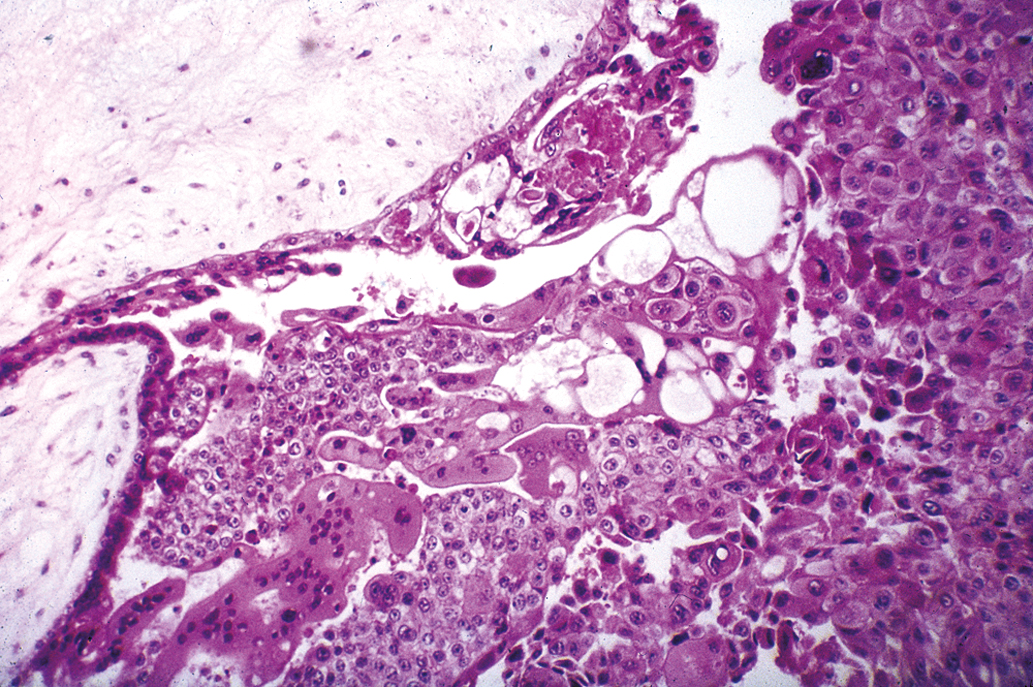
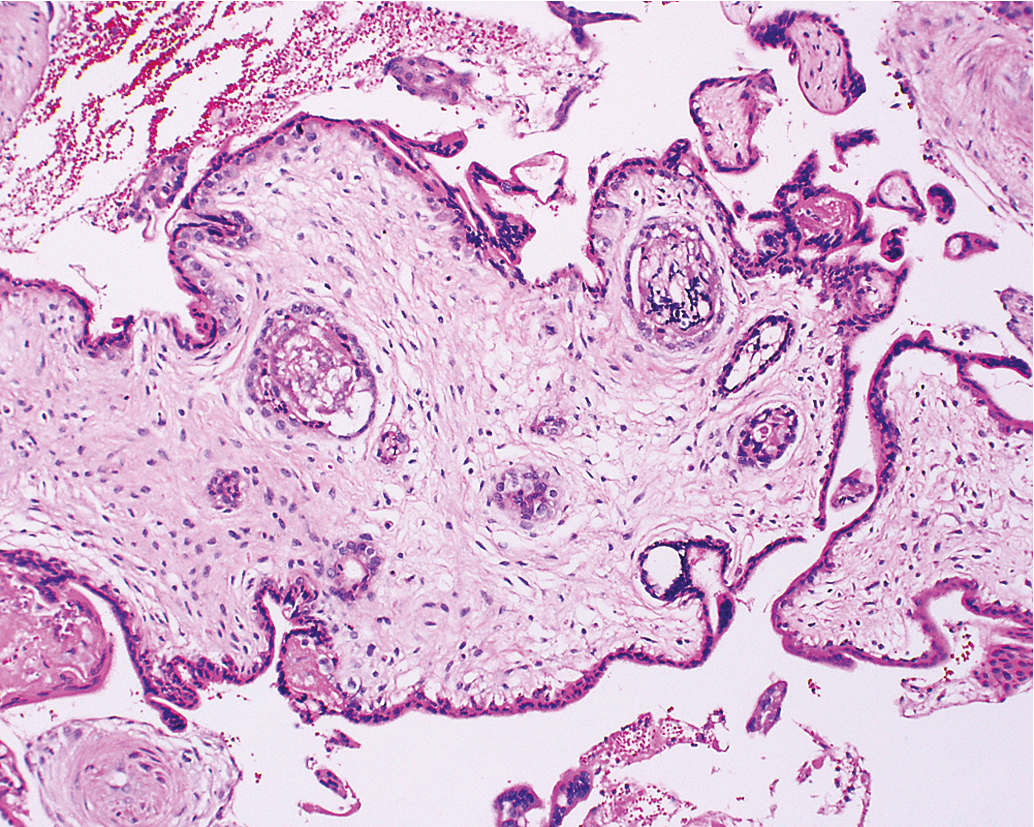
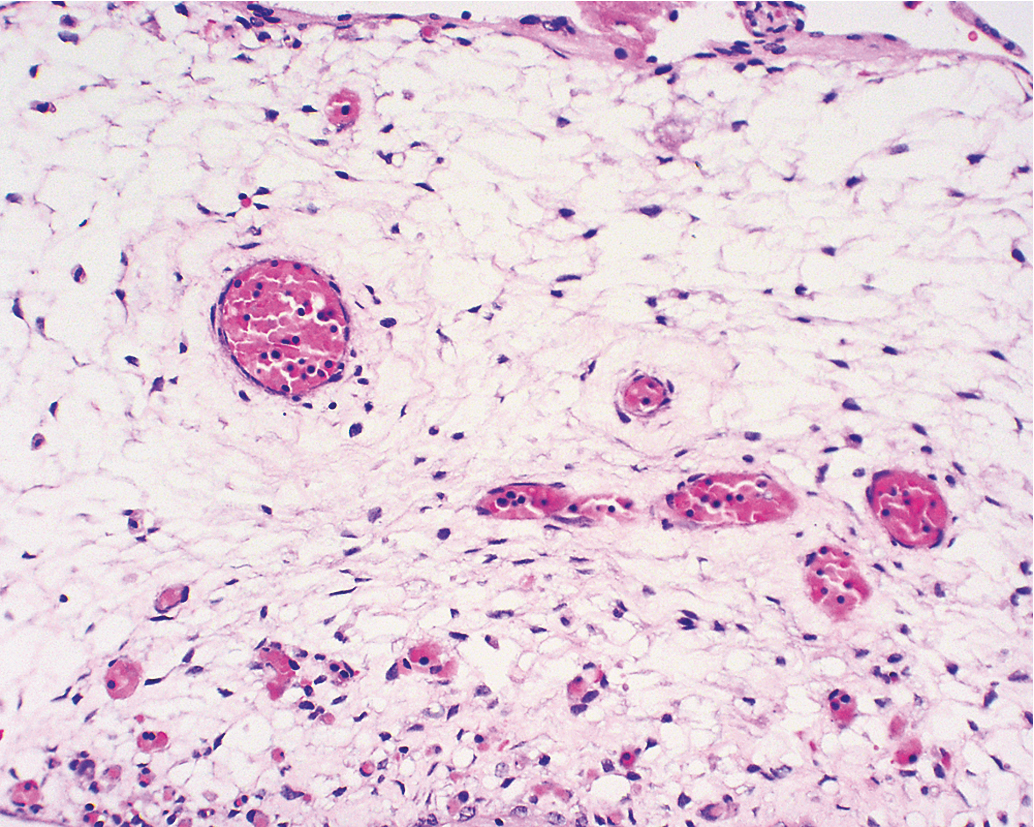
Other techniques have been developed to differentiate CM from PM. These include karyotyping, flow cytometry, fluorescence in situ hybridization, and amplification of short tandem repeat loci. In addition, p57 immunohistochemistry (IHC) staining is absent in CM but present in PM because it is paternally imprinted, that is, maternally expressed (see Table 7.1 ). Unfortunately, IHC staining for p57 does not distinguish PM from nonmolar abortuses, which also uniformly express p57. Although most cases of molar pregnancy can be classified as CM or PM using histopathology alone, histologic differences may not be apparent if diagnosis is made early in gestation. In this scenario, molecular genetic techniques may be critical for accurate diagnosis.
Presentation and symptoms
Irregular bleeding is the hallmark symptom for hydatidiform moles. Nearly all patients with moles have delayed or irregular menses for varying time periods. Vaginal bleeding usually occurs during the first trimester. The bleeding may vary from spotting to hemorrhage requiring blood transfusion. In earlier series, bleeding occurred in 73% to 100% of molar pregnancies including CM and PM. Expulsion of recognizable molar vesicles may accompany vaginal bleeding when the gestation approaches the second trimester.
There is increasing information that the presentation of moles may be changing as a result of earlier and improved diagnostic tests, including early ultrasound and hCG serum assays. The New England Trophoblastic Disease Center (NETDC) group compared symptoms in CM treated at their institution from 1994 to 2013 with those treated from 1988 to 1993. Patients in the more recent cohort were less likely to present with vaginal bleeding (46% vs. 84% in the earlier cohort). Patients in the recent cohort were also more likely to be diagnosed with a molar pregnancy at less than 11 weeks (56% vs. 41%). However, earlier diagnosis did not result in decreased rates of GTN.
Other symptoms include nausea and vomiting caused by the high levels of hCG. Nausea and vomiting were reported in almost one-third of patients with hydatidiform mole in older studies. However, in the most recent series from the NETDC, 14% of 180 patients presented with hyperemesis from 1994 to 2013. Curry and colleagues also reported hyperemesis in 14% of patients.
Preeclampsia-like symptoms in the first trimester of pregnancy are nearly pathognomonic of a hydatidiform mole. Symptoms of headache with hypertension, hyperreflexia, and proteinuria define preeclampsia. Preeclampsia was present in 1% of patients in the NETDC cohort and in 12% of the patients in the earlier study by Curry and colleagues. Fortunately, eclampsia in the setting of hydatidiform mole is rare.
Molar pregnancy is also associated with other medical comorbidities, such as tachycardia and hypertension from hyperthyroidism or shortness of breath and chest pain from acute respiratory distress syndrome. Hyperthyroidism occurs rarely but can be very severe. Laboratory evidence of hyperthyroidism can occur in as many as 10% of patients; however, clinical manifestations are less frequent. Hyperthyroidism is caused by the production of elevated levels of normal hCG by molar tissue. Therefore patients present with elevated T3 and T4, but thyroid stimulating hormone (TSH) levels are usually normal. hCG has structural homology with the TSH molecule and binds to TSH receptors at elevated levels, resulting in secondary hyperthyroidism. Furthermore, Yoshimura and colleagues demonstrated that the isoforms of hCG produced by molar pregnancies have higher thyrotrophic activity compared with the hCG produced by normal pregnancies. Clinical manifestations of hyperthyroidism disappear once the molar pregnancy is evacuated. Antithyroid therapy may be indicated for a short period to control hyperthyroidism during molar evacuation. Specifically, the use of beta-blockers to control tachycardia and hypertension may be indicated.
Acute respiratory distress can also occur . It is very rare, occurring in only 2% of patients in the NETDC cohort but can be a complication in approximately 20% of patients with uterine enlargement more than 16 weeks’ gestational size. These patients often have coexisting pathology that may alter cardiac or pulmonary function such as preeclampsia, hyperthyroidism, and anemia. This combination can contribute to acute cardiopulmonary decompensation because of flash pulmonary edema. Trophoblastic embolization is a rare cause of respiratory distress in patients with hydatidiform moles. Respiratory distress is most often associated with a large volume of molar tissue and uterine enlargement greater than 16 weeks’ gestational size.
Theca-lutein cysts are caused by hyperstimulation of the ovaries by excessive hCG production. In older series, approximately 15% to 25% of patients with unevacuated molar pregnancies presented with theca-lutein cysts larger than 6 cm. More recent series have shown rates of theca-lutein cysts at presentation of 6% to 13%. Although theca-lutein cysts will resolve after molar evacuation, there may be considerable lag behind the decline of hCG levels. Surgical intervention is rarely required for acute torsion or bleeding. Patients who develop theca-lutein cysts have a higher incidence of postmolar GTN. Furthermore, patients with the combination of enlarged ovaries with a large for gestational age uterus are at a high risk for malignant GTN. Up to 57% of patients with this combination require subsequent therapy for postmolar GTN.
Classically, a patient with a hydatidiform mole was said to have a uterine size excessive for gestational age. Historically this was found in more than 50% of patients with CM; however, approximately one-third of patients have uteri smaller than expected for gestational age. In the NETDC cohort from 1994 to 2013, uterine size was excessive for dates in only 24%, reflecting earlier diagnosis.
Similar to the comparative study performed by NETDC, Mangili and colleagues from Italy compared clinical features of molar pregnancies between two historical cohorts, 1970 to 1982 versus 1992 to 2004. The later cohort had significantly less vaginal bleeding on presentation (51% vs. 74%, P < .0001), fewer with increased uterine volume (29% vs. 51%, P < .0001), and fewer with theca-lutein cysts (13% vs. 21%, P = .03). There was no difference in rates of hyperemesis or preeclampsia. Because molar pregnancies are diagnosed at an increasingly early gestational age, clinical symptoms are less frequent. As a result, there is increasing reliance on histopathologic analysis to identify cases of molar pregnancies.
The previously outlined complications are seen mainly in patients with CMs. Patients with PMs usually do not exhibit excessive uterine size, theca-lutein cysts, preeclampsia, hyperthyroidism, or respiratory problems. In most patients with PMs, the clinical and ultrasound diagnosis is usually a missed or incomplete abortion. PMs are often underdiagnosed due to the lack of clinical suspicion and the often subtle or focal nature of the pathologic changes in the placental tissues. Therefore products of conception from missed or incomplete abortions should be thoroughly examined by pathologists to prevent a missed diagnosis of PM. Cytogenetic testing may be required to accurately diagnose GTD.
Diagnosis
Patients with CMs are often clinically diagnosed by characteristic ultrasound features and a markedly elevated hCG. In contrast, the majority of patients with PM and an increasing proportion of patients with CM are clinically misdiagnosed as missed abortion. A quantitative hCG of greater than 100,000 IU/L, an enlarged uterus with absent fetal heart sounds, and vaginal bleeding suggest a diagnosis of hydatidiform mole. However, a single hCG determination is not diagnostic. A single high hCG value may be seen with a normal single or multiple pregnancy, especially if there has been bleeding or disruption of the placenta. Therefore an isolated hCG value alone should not be used as the sole determining factor in diagnosing hydatidiform mole. Conversely, a “normal” hCG level for an anticipated gestational age does not exclude the possibility of a molar pregnancy.
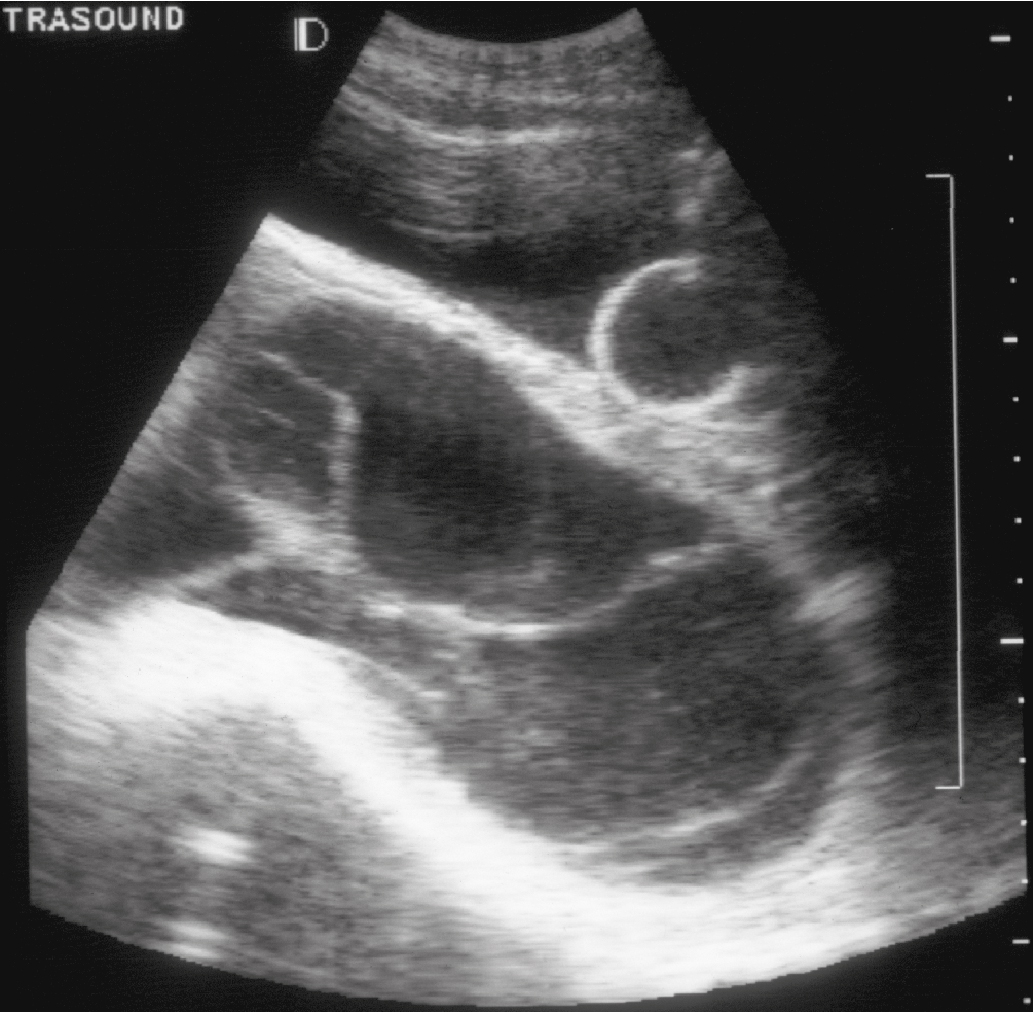
Ultrasound has replaced all other radiographic means (e.g., amniography or uterine angiography) for diagnosis of hydatidiform mole. Molar tissue typically is identified as a diffuse mixed echogenic and vesicular pattern replacing the placenta ( Fig. 7.7 ), traditionally labeled a “snowstorm appearance.” Recently, with increasing use of early ultrasonography, classic features may be subtle or lacking in cases of early CM or PM. In a recent series based on histopathologic identification of molar pregnancies, 52% of patients were diagnosed based on ultrasound examination before evacuation of the uterus. In another series, the abundant diffuse vesicular pattern was absent in 10% of CM pregnancies and only subtly present in 63% of CMs, based on ultrasounds performed at mean gestational age of 7 weeks and 6 days.
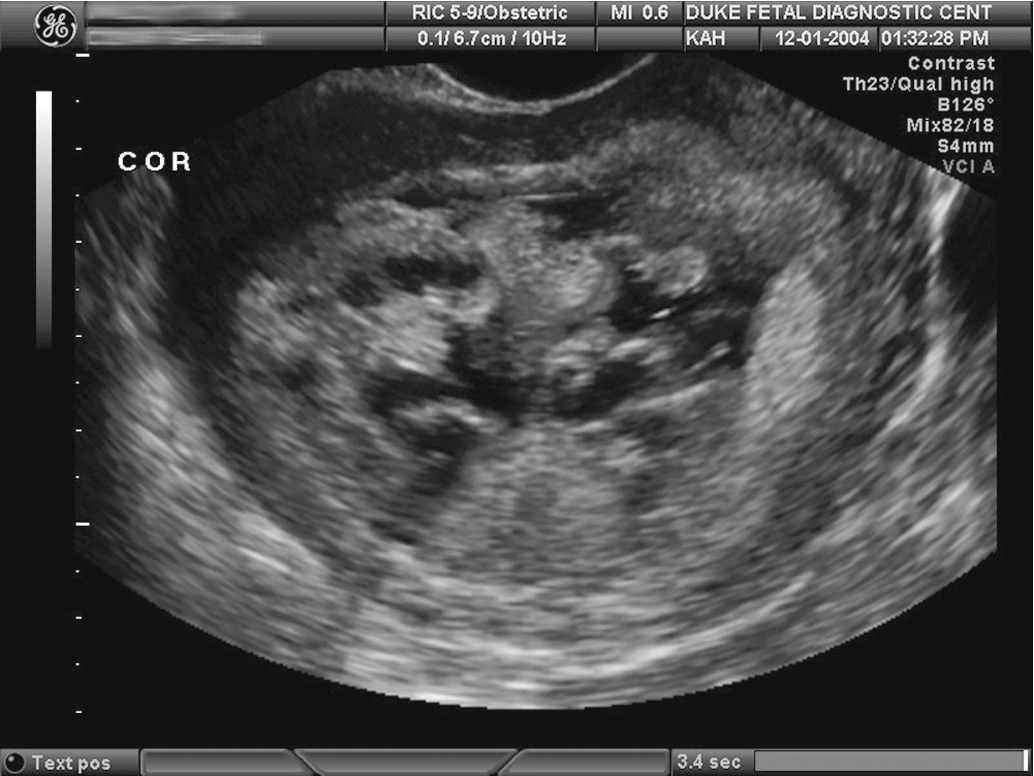
A large study from Charing Cross Hospital in the United Kingdom assessed more than 1000 cases of GTD from 2002 to 2005. They found an overall 44% detection rate by ultrasound of molar pregnancies before evacuation. The overall detection rate was 35% to 40% before 14 weeks of gestation and 60% after 14 weeks. The sensitivity was 44% and specificity was 74%. The positive predictive value was 88%, and the negative predictive value was 23% for detecting molar pregnancy of any type. Detection rates were higher for CM versus PM and for later gestations. Nonetheless, fewer than 50% of molar pregnancies were diagnosed by ultrasound in first-trimester scans.
In a smaller series from St. George’s Hospital in London, the sensitivity of ultrasound for diagnosis of a CM was 95% and 20% for a PM. Another study evaluated the use of both ultrasound and hCG values to diagnose hydatidiform mole. When ultrasound was used alone, 42% of patients with molar pregnancies did not have a definite diagnosis on first examination. When the hCG value was above a threshold of 82,350 mIU/mL and was used with the initial ultrasound findings, 89% of hydatidiform moles were correctly identified. A combined diagnostic approach using hCG, clinical history, examination, and imaging is often required to make the clinical diagnosis of hydatidiform mole.
Several reports have been made of a hydatidiform mole arising in ectopic sites, such as the fallopian tube. These patients present with classic symptoms and signs of ectopic pregnancy, including possible tubal rupture and hemorrhagic shock. Tubal GTD was diagnosed in 16 (0.8%) of 2100 women with GTD who were managed at the NETDC. Various authors have warned that the current trend of treating ectopic pregnancies with conservative surgery or single-dose methotrexate necessitates close monitoring of serum hCG levels to avoid missing the diagnosis of ectopic GTD.
Evacuation
Given the subtle presentation of GTD in the modern era, the diagnosis of molar pregnancy will increasingly be made only after histologic evaluation of uterine curettings. Curettage is often performed for a suspected incomplete spontaneous abortion. When hydatidiform mole is diagnosed in this setting, patients should be monitored with serial hCG measurements. A baseline postevacuation chest radiograph should be considered. For patients in whom hydatidiform mole is suspected prior to evacuation, the following laboratory evaluation is recommended ( Box 7.1 ): complete blood count with platelet determination, clotting function studies, renal and liver function studies, blood type with antibody screen, and hCG level. A preevacuation chest radiograph should also be obtained.
Stabilize medically (e.g., treat clinical hyperthyroidism, transfusion if needed)
Evacuation: Suction D&E (or hysterectomy in selected patients)
Post evacuation quantitative hCG and chest x-ray
Effective contraception
Monitor quantitative serum hCG levels every 1–2 weeks until 3 normal values or criteria for GTN
Partial mole—no further surveillance required after normal hCG
Complete mole—further surveillance after normal hCG may be indicated (see text)
Initiate treatment for GTN using indications listed in Box 7.2 .
Evacuation of molar pregnancies should be performed expeditiously. Medical complications are observed in approximately 25% of patients with molar pregnancy and uterine enlargement of greater than 14 to 16 weeks gestational size. The molar pregnancy should be evacuated as soon as possible after stabilization of any medical complications. The choice of facilities for molar evacuation should be based on the expertise of the physician, uterine size, and ability of the facility to manage existing medical complications. In most patients, the preferred method of evacuation is suction dilation and evacuation (D&E) (see Box 7.1 ).
Medical induction of labor with oxytocin or prostaglandin is not recommended for evacuation because of increased blood loss. Induction may increase the risk for malignant sequelae compared with suction D&E. The Charing Cross group reported a significant trend toward more frequent evacuation by suction curettage compared with sharp curettage or medical induction for molar evacuation over their study interval. Postmolar GTN developed in 5.9%, 3.8%, and 9.1% of their patients evacuated with suction curettage, sharp curettage, and medical induction, respectively ( P < .05, for increased postmolar GTN in the medical induction group). Furthermore, many patients require D&E to complete the evacuation of the mole after medical induction of labor.
Evacuation is usually performed under general anesthesia, with ultrasound guidance, but local or regional anesthesia may be used for a cooperative patient with a small uterus. After serial dilatation of the cervix, uterine evacuation is accomplished with the largest cannula that can be introduced through the cervix. Most providers will begin suction curettage in the lower uterine segment allowing the uterus to contract progressively as the products of conception are evacuated. Evacuation may be accompanied by significant blood loss, particularly with increasing gestational age. Intravenous (IV) oxytocin is begun after the cervix is dilated and continued for several hours postoperatively to assist with uterine contractility. Other uterotonics such as methylergonovine or misoprostol may be used if needed. After completion of suction D&E, gentle sharp curettage may be performed. Repeat uterine evacuation by D&E has been performed for persistent GTD, defined in the United Kingdom as failure of hCG to normalize within 4 to 6 weeks or a rise of hCG any time after primary dilation and curettage (D&C). The utility of repeat D&C is discussed subsequently.
Hysterectomy is an alternative to suction D&E for molar evacuation in selected patients who do not wish to preserve childbearing ( Fig. 7.8 ). The adnexa are routinely preserved and theca-lutein cysts should be left in situ unless they are torsed or ruptured and actively bleeding. A meta-analysis of the effect of hysterectomy in the management of hydatidiform mole in patients 40 years old or older found a significant advantage to hysterectomy. Compared with uterine evacuation in these patients, the overall risk (OR) for developing postmolar GTN was 0.19 (95% confidence interval [CI] 0.08 to 0.48, P = .0004). The risk of postmolar GTN after hysterectomy remains approximately 3% to 5%; therefore these patients should be monitored postoperatively with serial hCG levels.
Risk factors for postmolar gestational trophoblastic neoplasia
Many of the risk factors for development of postmolar GTN indirectly reflect the amount of trophoblastic proliferation at the time of evacuation. Age older than 40 years, preevacuation hCG levels greater than 100,000 mIU/mL, uterine size larger than expected by dates, and theca-lutein cysts ( Fig. 7.9 ) increase the risk. Many of these factors interact. The combination of theca-lutein cysts and uterus larger than expected for dates increases the risk of postmolar GTN to 57%. Other series have used multivariate analysis to identify risk factors for postmolar GTN and have reported that age, parity, initial uterine size, presence of theca-lutein cysts, and initial hCG concentration were not independent prognostic factors.
Multiple risk factors incorporated into scoring systems seek to categorize patients and predict which patients will develop postmolar GTN. However, many of these efforts have not been successful. Parazzini and colleagues found that, although 15% of their patients had risk factors that classified them as “high risk,” these patients accounted for a minority of the cases of postmolar GTN. Of note, despite the earlier diagnosis and decreasing symptom burden among patients diagnosed in the modern era with molar pregnancies, there has not been a decrease in the incidence of postmolar GTN. This implies that other molecular biologic mechanisms may be responsible for the development of postmolar GTN, rather than the amount of trophoblastic proliferation at the time of evacuation. Most centers do not treat “high-risk” molar pregnancies with postevacuation chemotherapy to prevent the development of postmolar GTN; rather they rely on surveillance with serial hCG levels and treat when postmolar GTN is diagnosed.
Postmolar surveillance
Serial quantitative serum hCG determinations should be performed after molar evacuation using one of several commercially available assays capable of detecting beta-hCG to baseline values (<2 to 5 mIU/mL). For monitoring patients with GTD, an hCG assay that can detect all forms of hCG including free beta, nicked-free beta, C-terminal hCG, beta core, and hyperglycosylated hCG should be used. Ideally, serum hCG levels should be obtained within 48 hours of evacuation and followed every 1 to 2 weeks while elevated (see Box 7.1 ). Reliable contraception during monitoring is recommended. After hCG values normalize, additional monitoring is usually recommended. Many sources recommend following patients with hCG values for 3 months after a PM and at least 6 months after a CM. Patients with a histologic diagnosis of malignant GTN (choriocarcinoma, invasive mole, PSTT, or ETT), the development of metastatic disease, a rising hCG value, or a plateauing of the hCG values over several weeks’ time are diagnosed with postmolar GTN, as discussed later. Approximately 6.8% to 20% of patients develop postmolar GTN after evacuation of a CM, whereas 1% to 5% of patients develop postmolar GTN after a PM.
The rationale for recommending an interval of monitoring after hCG value remission is to allow identification of patients who develop postmolar malignant GTN after achieving normal hCG values. Although rare instances of long latent periods preceding postmolar GTN have been reported, most cases of GTN occur within 6 to 12 months of evacuation. After normalization of hCG has been confirmed with a second hCG value, the risk of developing postmolar GTN is extremely small. In the largest study reported from the United Kingdom, Coyle and associates followed more than 20,000 women with hydatidiform moles after evacuation. For PM the risk of postmolar GTN was 1:3195 after normalization of hCG, whereas it was 1:406 for CM. The risk of postmolar GTN dropped rapidly during the first 6 months of monitoring. Women with CM where hCG normalized more than 56 days after evacuation had a 3.8-fold increased higher risk of GTN after normalization of hCG. They recommended that hCG monitoring could be stopped for PMs after a confirmatory hCG but recommended continuing to monitor CMs for 6 months. The International Federation of Gynecology and Obstetrics (FIGO) and National Comprehensive Cancer Network (NCCN) support this recommendation.
A meta-analysis of 19 studies performed by Albright and associates reported a similar low risk of GTN after normalization of hCG levels following molar evacuation. In 14,864 patients with PMs, the risk was 0.35% compared with 0.03% in 18,357 patients with CMs ( P < .002). In CMs that developed GTN after hCG normalization, 89.6% occurred when the time from evacuation to normal hCG exceeded 56 days. The risks for late GTN continued past 6 months after normalization of hCG in 60.7% of CM cases and 40% of the PM cases. Based on these data, the authors recommended discontinuing monitoring of hCG levels after a single confirmatory value in patients with a PM or a CM with hCG normalization within 56 days. For patients with CM who had a longer interval for hCG normalization, they recommended hCG monitoring at 3-month intervals for 12 months, which would require fewer hCG determinations than monthly values over a 6-month surveillance.
Although pregnancies that occur within 12 months after molar evacuation are usually normal gestations, an early pregnancy obscures the value for monitoring hCG values during this interval. For this reason, reliable contraception is recommended during hCG surveillance. Several studies, including a randomized study performed by the Gynecologic Oncology Group GOG), established that moderate-low dose oral contraceptives do not increase the incidence of postmolar GTN and significantly decrease intercurrent pregnancies during postmolar surveillance.
After completion of surveillance documenting remission, pregnancy can be encouraged. Patients with prior PM or CM have a 1% to 2% incidence of a second mole in subsequent pregnancies. All future pregnancies should be evaluated by an early obstetric ultrasound examination. The risk of repetitive molar pregnancies increases substantially if a woman has had two or more prior moles. Women with consecutive molar pregnancies should undergo genetic testing for mutations in NLRP7 and KHDC3L, which produce biparental moles. These mutations are identified in up to half of women with consecutive moles. Assisted reproduction techniques with the use of donor eggs are recommended for future pregnancies in patients with these mutations.
Prophylactic chemotherapy after molar evacuation
Randomized studies have evaluated prophylactic chemotherapy after molar evacuation. Kim and colleagues reported that a single course of alternating methotrexate/folinic acid reduced the incidence of postmolar GTN from 47.4% to 14.3% ( P < .05) in patients with high-risk molar pregnancies, but the incidence was not reduced in patients with low-risk molar pregnancies. Patients who received prophylactic chemotherapy but developed GTN required more chemotherapy than those who had not been exposed to prophylactic chemotherapy. Kashimura and associates evaluated oral or IV methotrexate 10 mg daily for 7 days within 3 weeks of evacuation in patients with both low-risk and high-risk moles. They reported that the risk of postmolar GTN was reduced from 18% to 7.5% in control versus treated groups, respectively ( P < .01). In the study by Limpongsanurak, a single course of actinomycin D was compared with observation in patients following evacuation of high-risk molar pregnancies. Postmolar GTN was 50% in the control group, compared with 13.8% in the treatment group ( P < .05). In these studies, there were no deaths in the treatment or control groups caused by GTN or treatment toxicity.
However, there are anecdotal cases of fatalities caused by prophylactic chemotherapy, and prophylactic chemotherapy does not eliminate the need for postevacuation follow-up. Furthermore, patients are exposed to drugs most frequently used to treat postmolar GTN, which could lead to relative chemoresistance, as reported by Kim et al. Receipt of chemotherapy is also associated with decreased quality of life, cost, and morbidity. In compliant patients, the low morbidity and mortality achieved by monitoring patients and instituting chemotherapy only in patients with postmolar GTN appear to outweigh the potential risk and small benefit of routine prophylactic chemotherapy. Exceptions to this are high-risk patients in whom hCG follow-up is not possible due to patient or system factors. Prophylactic chemotherapy might be considered in these cases.
Coexistent molar pregnancy with a normal fetus
Coexistence of a genetically normal fetus with molar change of the placenta is relatively rare ( Fig. 7.10 ), occurring in 1 in 22,000 to 1 in 100,000 pregnancies. The majority of the literature covering this relatively rare entity consists of case reports and case series. Both CM and PM with a coexistent normal fetus have been reported. A variety of criteria have been used to evaluate these pregnancies. Many of the reports that antedated the histologic and cytogenetic distinction between CM and PM likely included twin gestations with coexistent fetus and molar gestation in addition to singleton PM. Although there might be an increased incidence of coexisting molar pregnancy and fetus related to an increase in multifetal pregnancies caused by ovulation induction for infertility, this may reflect only reporting bias.
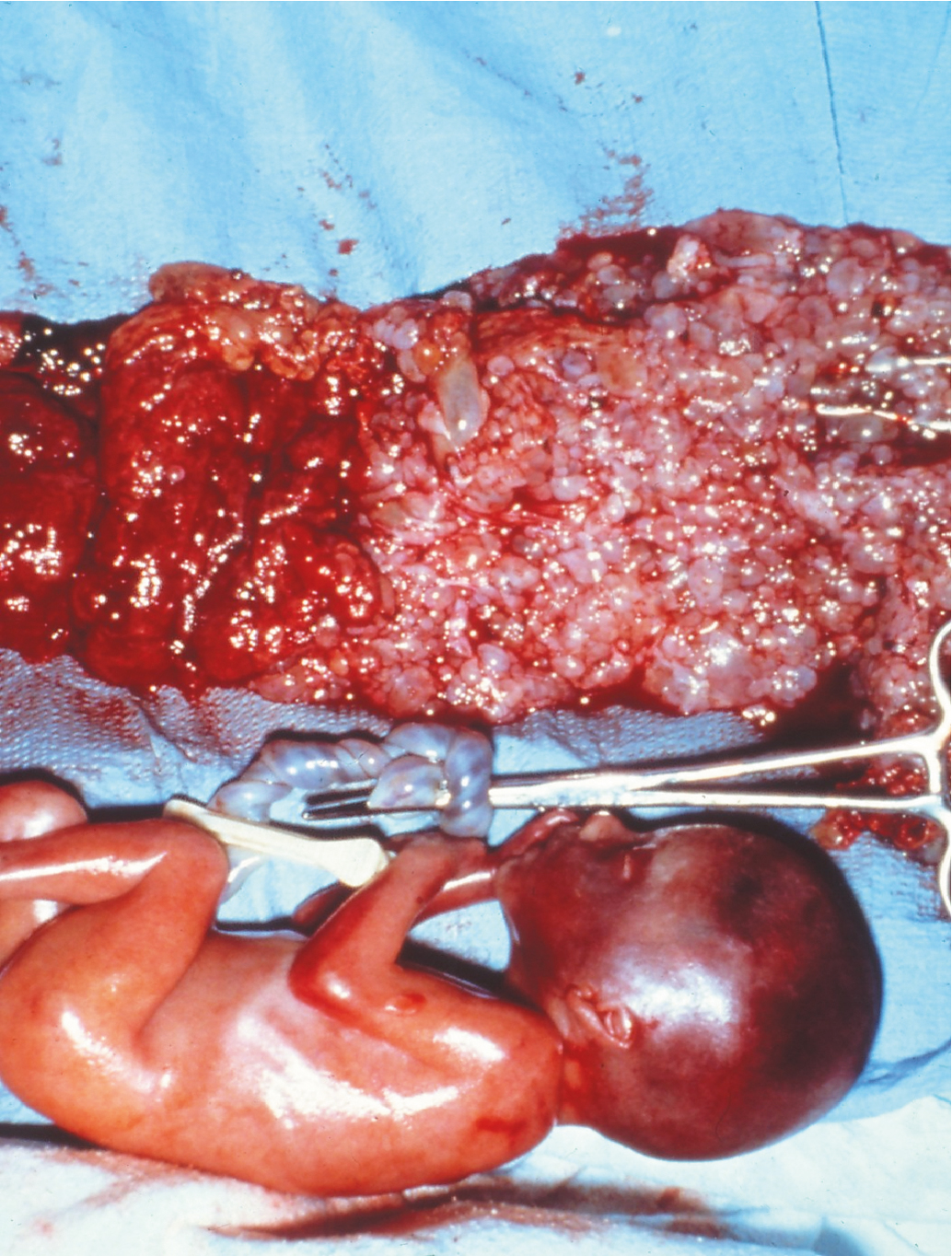
Most of these are diagnosed antepartum by ultrasound findings of a complex, cystic placental component distinct from the fetoplacental unit. However, in a few cases the diagnosis is not suspected until examination of the placenta following delivery. Medical complications of hydatidiform mole appear to be increased, including hyperthyroidism, hemorrhage, and pregnancy-induced hypertension.
Compared with singleton hydatidiform moles, twin pregnancy with fetus and mole has an increased risk for postmolar GTN and a higher proportion of patients develop high-risk GTN requiring multiagent chemotherapy. In patients with a coexistent mole and fetus who continue pregnancy beyond 12 weeks, the subset who develop early complications leading to termination of the pregnancy before fetal viability may have increased risk of postmolar GTN compared with pregnancies continuing into the third trimester.
Among 72 patients collected by a national survey of physicians in Japan, 24 patients underwent first-trimester evacuation, with 20.8% subsequently developing postmolar GTN. In comparison, 45.2% of 31 patients who required evacuation during the second trimester for medical complications or hemorrhage and 17.6% of the 17 who delivered in the third trimester developed postmolar GTN ( P < .05). Nine (50%) of the 18 patients with proven androgenic mole in association with a fetus subsequently were treated for postmolar GTN, but it is not certain that this apparent increased risk was a result of selection bias. Major congenital abnormalities were not reported in surviving infants.
Niemann and associates reported 90 cases from the United Kingdom, and Lin and colleagues collected 72 cases from Boston and Brazil. In both series, approximately 60% of pregnancies that were not terminated for early complications resulted in a viable pregnancy. Termination of pregnancies for maternal complications occurred in 19% of the patients from the United Kingdom and 10% of the patients in the combined series. Miscarriage or intrauterine death occurred in 19% and 25% of pregnancies for each of the series, respectively. Postmolar GTN developed in 27% and 46% of patients, respectively, in these reports. In both reports, elective termination of pregnancy did not decrease the risk of postmolar GTN, but the patients who had pregnancy termination for medical complications or hemorrhage were at higher risk for postmolar GTN.
For patients with coexistent hydatidiform mole and fetus suspected by ultrasound, there are no clear guidelines for management. The ultrasound should be repeated to exclude retroplacental hematoma, other placental abnormalities, or degenerating myoma. The fetoplacental unit should be fully evaluated for evidence of gross fetal malformations. If the diagnosis is still suspected and continuation of pregnancy is desired, fetal karyotype should be obtained to exclude the diagnosis of PM, and serial serum hCG values should be followed.
Patients should be counseled that they are at increased risk for medical complications of pregnancy requiring evacuation, including bleeding, premature labor, and pregnancy-induced hypertension. They should be counseled about these risks and the increased risk of postmolar GTN after evacuation or second-trimester delivery. If fetal karyotype is normal, major fetal malformations are excluded by ultrasound, and there is no evidence of metastatic disease, it is reasonable to allow the pregnancy to continue unless pregnancy-related complications force delivery. After delivery, the placenta should be histologically evaluated and the patient should be followed closely with serial hCG values, as one would with a singleton hydatidiform mole.
Gestational trophoblastic neoplasia
GTN is defined by a plateaued, rising, or prolonged persistence of elevated hCG values after molar evacuation, histologic diagnosis of choriocarcinoma, invasive mole, PSTT, ETT, or identification of metastatic disease (see Table 7.3 ). Prompt diagnosis of malignant GTN is important because delay in diagnosis may increase the patient’s risk and decrease response to treatment. All patients with GTN should undergo a complete evaluation aimed at identifying metastatic sites ( Box 7.2 ) and other clinically important prognostic factors. Several classification systems have been used to determine prognostic groups and assist in the triage of management for individual patients. FIGO revised its staging system for patients with malignant GTN in 2000 to reflect prognostic factors other than simple anatomic distribution of disease and is currently the standard nomenclature for staging GTN.
Diagnosis of GTN
After molar evacuation a sustained plateau of four hCG values (±10%) over at least 3 weeks: days 1, 7, 14, and 21.
After molar evacuation a sustained rise of three hCG values (>10%) over at least 2 weeks: days 1, 7, and 14.
After molar evacuation persistence of detectable hCG for more than 6 months.
Histologic diagnosis of invasive mole, choriocarcinoma, PSTT, or ETT.
Metastatic disease with elevated hCG level after molar evacuation or without an established primary site in the absence of pregnancy.
Evaluation of GTN
Complete physical and pelvic examination
Baseline hematologic, renal, and liver function tests
Pretherapy quantitative hCG value used to compute risk score
Chest computed tomography scan
CT or MRI scan of abdomen and pelvis
Contrasted MRI or CT scan of brain
Diagnosis
Between one-half and two-thirds of cases of GTN that require treatment follow evacuation of CM or PM. Because many of these patients are treated on the basis of hCG levels rather than hysterectomy, the distribution of histologic lesions is not known. In older studies, approximately 50% to 70% of patients with postmolar GTN had persistent or invasive moles, whereas 30% to 50% had postmolar gestational choriocarcinoma.
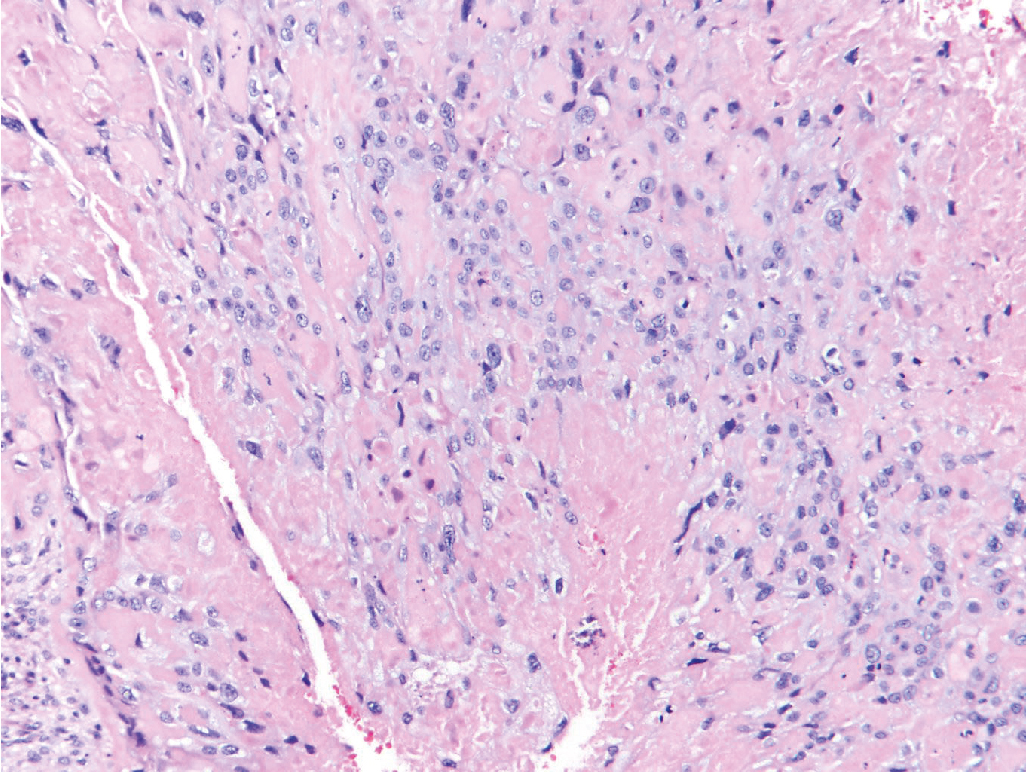
Gestational choriocarcinoma ( Fig. 7.11 ) derived from term pregnancies, spontaneous abortions, and ectopic pregnancies account for the vast majority of the other cases of malignant GTN. PSTT ( Fig. 7.12 ) and ETT ( Fig. 7.13 ) are rare forms of GTN that can follow any pregnancy event.
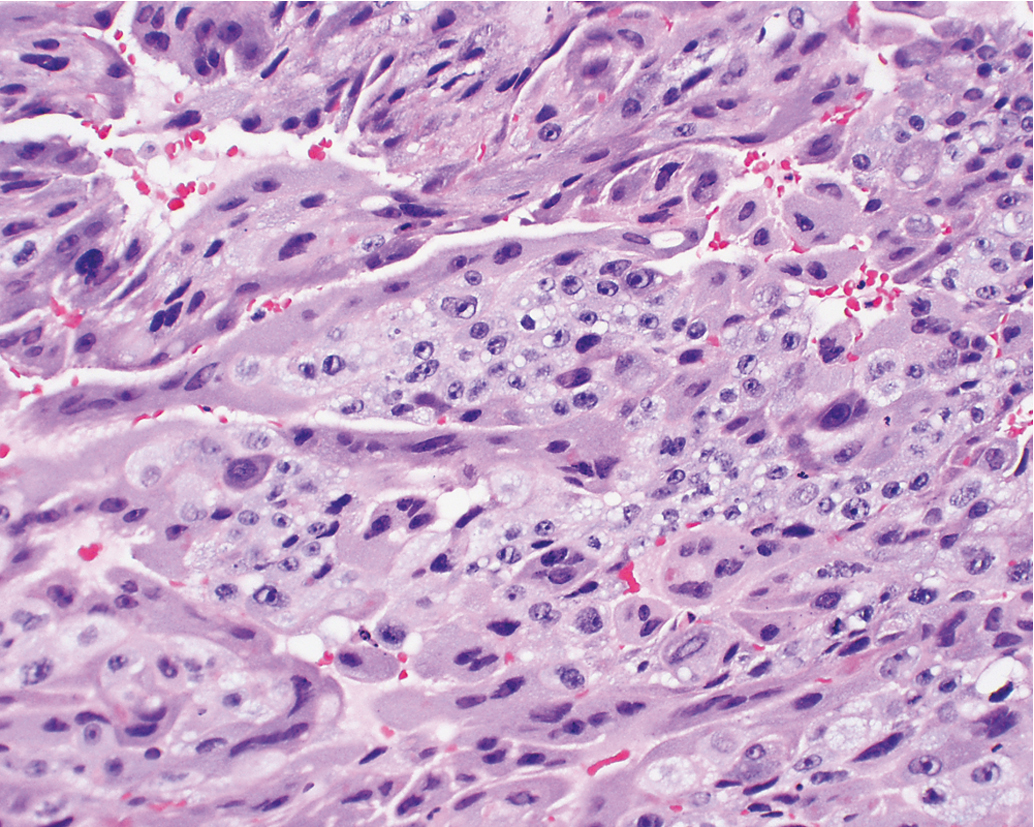
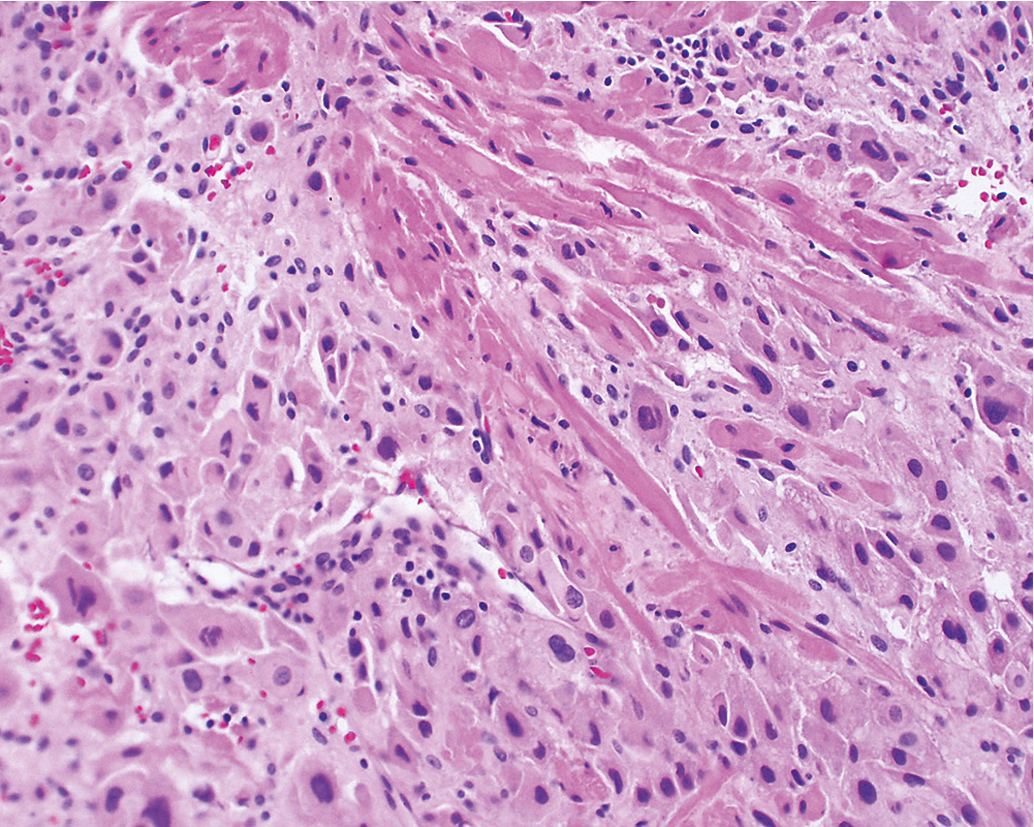
Invasive moles are characterized by the presence of edematous chorionic villi with trophoblastic proliferation that invade directly into the myometrium . Metastasis of molar vesicles may occur. Most often invasive moles will undergo spontaneous resolution after many months, but they are treated with chemotherapy to prevent morbidity and mortality caused by uterine perforation, hemorrhage, or infection. Gestational choriocarcinoma is a pure epithelial malignancy, comprising both neoplastic syncytiotrophoblast and cytotrophoblast elements without chorionic villi (see Fig. 7.11 ). Gestational choriocarcinomas are highly malignant, and chemotherapy is usually indicated.
PSTTs and ETTs are rare. These tumors are characterized by absence of villi with proliferation of intermediate trophoblast cells (see Figs. 712 and 7.13 ). The syncytiotrophoblast population observed in choriocarcinoma is lacking, with relatively lower levels of intact hCG secreted by these tumors. In general, PSTT and ETT are not as sensitive to chemotherapy as other forms of GTN . Fortunately, most patients with PSTT and ETT present with disease confined to the uterus and can be treated with hysterectomy.
Women with postmolar GTN are usually diagnosed early in the course of disease by serial hCG monitoring. In contrast, patients with malignant GTN after nonmolar gestations often present with predominantly nongynecologic symptoms including hemoptysis or pulmonary embolism, cerebral hemorrhage, gastrointestinal or urologic hemorrhage, or metastatic malignancy from an unknown primary site. The index pregnancy event may have occurred years before presentation or may have been a subclinical spontaneous abortion. In a series from the NETDC, the presentation of choriocarcinoma after term gestation in the modern era (1996–2011) was compared with an earlier period (1964–1996). They found 30% of women in the modern era were asymptomatic and diagnosed by an incidental positive pregnancy test or placental pathology compared with 2% in the previous time period ( P = .001). The possibility of malignant GTN should be suspected in any woman of reproductive age who presents with metastatic disease from an unknown primary site or cerebral hemorrhage. Under these circumstances the diagnosis is facilitated by a high index of suspicion coupled with serum hCG testing and exclusion of a concurrent pregnancy. A high level of hCG confirms the diagnosis of GTN without need for tissue biopsy.
Before the development of effective chemotherapy, 10% of patients required hysterectomy after molar evacuation for the treatment of invasive molar pregnancy or choriocarcinoma. Other prechemotherapy reports indicated a mortality rate of more than 20% among patients with invasive mole. Since the development of chemotherapy, between 15% and 36% of patients in the United States are treated after molar evacuation based on inclusive hCG level criteria for initiating chemotherapy in an attempt to limit the morbidity and mortality caused by postmolar GTN. In contrast, Bagshawe and colleagues used very conservative hCG criteria to initiate chemotherapy and reported treating only approximately 7.8% of their patients after molar evacuation.
The pattern of hCG regression after evacuation of hydatidiform mole is most often used to make a diagnosis of postmolar GTN. After evacuation, most patients will have an initial fall in hCG levels and should be followed with serial hCG values every 1 to 2 weeks. Almost every series has treated patients on the basis of a confirmed hCG rise. However, in many studies, chemotherapy was initiated on the basis of an hCG level plateau of relatively short (<3 weeks) duration. Kohorn noted that 15% of his patients had hCG plateaus after molar evacuation that lasted at least 2 weeks, followed by spontaneous regression without intervention. In a trial of actinomycin D, Schlaerth and colleagues observed that 25% of their patients had a substantial spontaneous fall in hCG levels on the day that chemotherapy was initiated. Based on the correlation between hCG level and tumor burden, Kohorn suggested an approach using shorter observation periods for patients with high hCG level plateaus but allowing longer periods of observation for those who have hCG plateaus at levels less than 1000 mIU/mL.
In some studies, an additional criterion for initiating chemotherapy after molar evacuation has been persistence of hCG for an arbitrary length of time, ranging between 6 weeks and 6 months after evacuation. The initial reports of chemotherapy from the National Institutes of Health (NIH) used persistence of hCG for more than 60 days after molar evacuation as an indication for chemotherapy treatment. Many series have documented that persistence of hCG for more than 60 days after evacuation increases the risk for developing postmolar GTN. However, 60% to 64% of patients with delayed regression of hCG beyond 60 days will ultimately undergo spontaneous hCG regression, with no deaths in those who began therapy more than 60 days from molar evacuation. Furthermore, groups reporting from the United Kingdom, where care for GTD patients is centralized, follow patients with elevated hCG levels for up to 6 months without an increase in morbidity or mortality among those treated after this prolonged follow-up.
Lurain and colleagues reported their experience with 738 patients with complete hydatidiform moles. In their study, 80.7% of patients had spontaneous regression of hCG values to normal, although only 65% had done so by 60 days. An additional 27.9% reached normal values during the next 110 days. Overall, 142 (19%) were treated with chemotherapy because of rising or plateauing hCG levels. Of these, 125 had a clinical diagnosis of invasive mole and 17 had choriocarcinomas. Only 15% of the 142 treated patients, or 3% of the total, developed metastases outside of the uterus. Morrow and Kohorn treated a higher proportion of their patients (36% and 27%, respectively) using hCG criteria derived from regression curves, compared with studies that use the individual patient’s hCG regression pattern. It appears that use of hCG regression curves may result in treatment of more patients than necessary, and it did not prevent metastatic disease.
In an effort to provide uniformity to the diagnosis of postmolar GTN, FIGO conducted workshops among members of the Society of Gynecologic Oncologists, International Society for the Study of Trophoblastic Disease, and International Gynecologic Cancer Society to formulate its current recommendations for evaluation and staging this disease. The current FIGO requirements for making a diagnosis of postmolar GTN are as follows (see Box 7.2 ):
- 1.
Four values or more of plateaued hCG (±10%) over at least 3 weeks: days 1, 7, 14, and 21.
- 2.
A rise of hCG of 10% or greater for 3 values or more over at least 2 weeks: days 1, 7, and 14.
- 3.
The histologic diagnosis of choriocarcinoma.
- 4.
Persistence of hCG beyond 6 months after mole evacuation.
The level and duration for observation of the hCG plateau beyond 3 weeks would be determined at the discretion of the treating physician. Observation of hCG level plateau for longer than 3 weeks was permitted because this does not appear to have an adverse effect on survival.
Recently, laboratory assays for hyperglycosylated hCG have been found to be highly sensitive for differentiating active GTN or choriocarcinoma from inactive or “quiescent” GTD in patients with hCG level plateaus. These newer assays, developed by Cole and associates at the US hCG Reference Service at the University of New Mexico, may allow earlier diagnosis of postmolar GTN in the future.
In 2016 investigators from Brazil questioned the need for treatment in women with elevated hCG values 6 months after molar evacuation. They reported that 16 (19.8%) of 81 patients followed without immediate treatment required chemotherapy for GTN based on subsequent hCG rise or development of metastatic disease. Only three patients required multiagent therapy for high-risk disease or failure of primary chemotherapy. Their current policy is to continue hCG monitoring without intervention in these patients as long as hCG values continue to fall.
The majority of centers in the United States will treat patients if metastases are identified. In contrast, Bagshawe and colleagues in the United Kingdom followed patients with pulmonary nodules conservatively and based treatment decisions on hCG regression patterns. Although the overall results from this policy were excellent, outcome for patients with pulmonary metastases was not detailed. Li and colleagues reported a retrospective study of patients with radiographic evidence of pulmonary metastases after molar evacuation. Seventeen patients received immediate chemotherapy, and 36 patients were monitored with serial hCG values and therapy was not given if the hCG values continued to decrease. Eighteen (50%) of the monitored patients required chemotherapy but required fewer cycles of chemotherapy than the group that was treated immediately, with only two patients needing multiagent chemotherapy for first line chemotherapy failure. Patients with a single pulmonary lesion were more likely to have spontaneous remission than those with greater than 1 lesion. In the United States, where care is decentralized, identification of metastases is included as an indication for initiation of therapy.
Abnormal human chorionic gonadotropin assays
“phantom” human chorionic gonadotropin
Rarely, women present with persistently elevated hCG levels but are subsequently found to have a false-positive hCG assay result, sometimes after receiving chemotherapy or surgery for presumed malignant GTN. Most patients with “phantom” hCG present with low-level hCG elevations, but occasionally values as high as 200 to 300 mIU/mL have been recorded. The false-positive hCG values result from nonspecific heterophile antibodies that interfere with the hCG immunometric sandwich assays. Many of these patients have an undefined previous pregnancy event and do not have radiographic evidence of metastatic disease. Serial hCG values usually do not substantially vary, despite prolonged observation, and do not substantially change with therapeutic interventions such as surgery or chemotherapy.
“Phantom” hCG may also present after evacuation of a hydatidiform mole or following a clearly defined pregnancy event, such as an ectopic pregnancy. It should be suspected if hCG values plateau at low levels and do not respond to therapeutic maneuvers such as methotrexate. Evaluation should include study of serum hCG using a variety of assay techniques at different dilutions of patient serum combined with a urinary hCG level if the serum level is greater than the threshold for the urinary assay. Heterophile antibodies are not excreted in the urine. Therefore, if they are the cause of serum hCG level elevation, urinary hCG values will not be detectable. False-positive hCG assays will also not be affected by serial dilution of patient sera. Markedly different values will be obtained using different assay techniques, with the majority of assays reflecting undetectable hCG. Other techniques are available to inactivate or strip the patient’s serum of heterophile antibodies and many current assays now incorporate these techniques to reduce the incidence of false-positive results. It is important to exclude the possibility of “phantom” hCG before subjecting these patients to hysterectomy or chemotherapy for GTN.
The “hook effect”
Instead of being a false-positive test such as a phantom hCG due to heterophile antibodies, the “hook effect” causes a false-negative or low hCG value in the presence of extremely elevated hCG levels. hCG immunoassays rely on a sandwich assay approach. A sandwich assay uses antibodies attached to a matrix to bind hCG and additional free-floating labeling antibodies bind to a different site on the hCG attached to the matrix. The labeling antibodies have a fluorescent marker, and the hCG level is determined by the intensity of fluorescence after the serum has been flushed out of the system. In the “hook effect,” the assay is overwhelmed by very high levels of hCG, generally greater than 1,000,000 mIU/mL. The large number of hCG molecules bind not only to the antibodies on the matrix but also to the labeling antibodies. Many fewer or no “sandwiches” are created, and the hCG result will either be falsely low or falsely negative. The “hook effect” can be overcome by serial dilution of the serum prior to using an hCG immunoassay, and this is standard practice in many laboratories; however, if a false-negative hCG is suspected, serial dilution of the patient’s serum should be performed.
Pretherapy evaluation
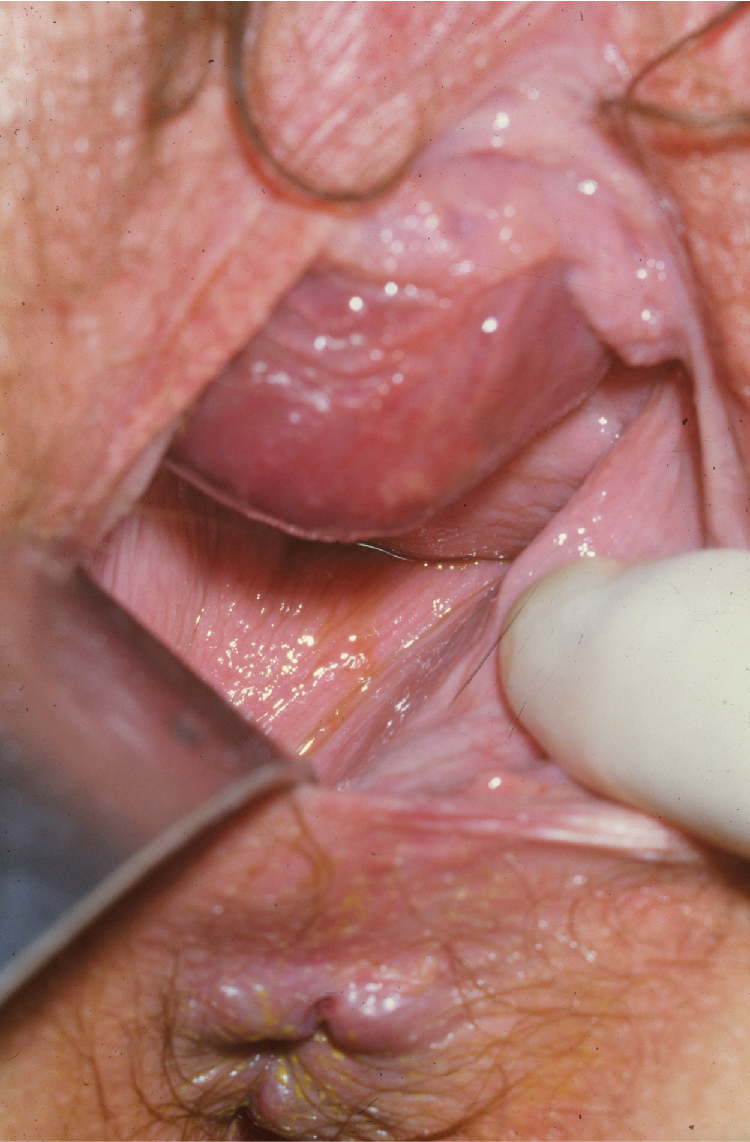
In contrast to epithelial endometrial cancer, GTN usually spreads by hematogenous dissemination. Venous metastasis can occur in the subvaginal venous plexus, resulting in vaginal metastases ( Fig. 7.14 ), or in the main uterine venous system with metastases to the parametrium and lungs. Although direct shunting into the systemic circulation may occur, the majority of disseminated metastases develop only after pulmonary metastases have become established. The brain, liver, gastrointestinal tract, and kidneys are the distant organs most often affected, but metastases to virtually every organ have been reported ( Table 7.2 ). Although lymphatic spread can occur, it is relatively uncommon except in PSTT and ETT. The hematogenous pattern of metastatic involvement is important when considering the radiographic evaluation of patients with GTN.
| Metastatic Site | Number (%) | (% Metastatic) |
|---|---|---|
| Nonmetastatic | 195 (54) | |
| Metastatic | 136 (46) | |
| Lung only | 110 | 81 |
| Vagina only | 7 | 5 |
| Central nervous system a | 9 | 7 |
| Gastrointestinal a | 5 | 4 |
| Liver a | 2 | 1.5 |
| Kidney a | 1 | 0.7 |
| Unknown b | 4 | 3 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree