Benign and Malignant Mesothelioma
Harvey I. Pass
Michele Carbone
Lee M. Krug
Kenneth E. Rosenzweig
INTRODUCTION
Malignant mesotheliomas (MM) are highly aggressive neoplasms that arise primarily from the surface serosal cells of the pleural, peritoneal, and pericardial cavities. The disease is characterized by a long latency from the time of exposure to asbestos to the onset of disease, suggesting that multiple somatic genetic events are required for tumorigenic conversion of a normal mesothelial cell.
MECHANISM OF ASBESTOS CARCINOGENESIS
There are six types of asbestos: amosite, crocidolite, anthophyllite, actinolite, tremolite, and chrysotile.1 The five types other than chrysotile are termed amphibole asbestos. Several nonasbestos minerals (e.g., erionite, wincherite) are very similar to asbestos structurally and chemically and are also capable of causing mesothelioma in humans. The mechanisms responsible for asbestos carcinogenicity are linked to the secretion of tumor necrosis factor (TNF)-alpha by mesothelial cells and macrophages exposed to asbestos that in turn leads to NFκB activation.2 The activation of the NFκB pathway in mesothelial cells allows them to survive the toxic insult and the genetic damage caused by asbestos, and these damaged but viable cells may proliferate into a mesothelioma.2
Animal Models of Asbestos Carcinogenicity
Mesotheliomas and sarcomas are induced in a dose-dependent fashion after intrapleural and intraperitoneal injection of asbestos into animals.3,4,5 Chronic inhalational studies are thought to be the best representation of asbestos carcinogenicity. Because of the cost of these experiments, however, very few have been performed.
Asbestos Carcinogenicity in Humans
Although injection and inhalation studies in animals suggest that all forms of asbestos are equally carcinogenic and that there is a clear dose-response relationship between asbestos exposure and mesothelioma risk, epidemiologic studies in humans do not support these observations. The proportion of mesotheliomas that are thought to be associated with asbestos varies in the literature from 16% to 90%.1 Although this wide range may reflect the distinct populations studied, it is more likely a result of the different methodologies used to determine exposure rates.1 Most human studies have indicated that amosite and crocidolite exposure carry a much greater risk than exposure to chrysotile.6,7 Chrysotile, however, is frequently combined with tremolite, an amphibole, that may instead be responsible for mesothelioma development.8
In humans, there is no clear dose-response curve to asbestos, and a threshold level below which mesotheliomas do not develop has not been determined, although it is generally accepted in the scientific community that background levels of exposure, such as those found in the lungs of almost all individuals, do not increase the risk of mesothelioma. It is estimated that <5% of asbestos miners exposed to high levels of asbestos develop mesotheliomas1; however, wives of some of these workers have developed mesotheliomas after they were presumably exposed to lower levels of asbestos compared to their husbands while washing their clothes.9,10 Similarly, mesothelioma development commonly occurs in workers in occupations in which asbestos exposure is higher than in the general population, but much lower than in asbestos miners. The carpenters, electricians, and construction workers who have developed mesotheliomas are a reflection of this.11 These data suggest that high levels of exposure are not necessarily correlated with increased risk of malignancy compared to moderate asbestos exposure, arguing against a classic doseresponse relationship.1
Mechanism of Asbestos Pathogenicity
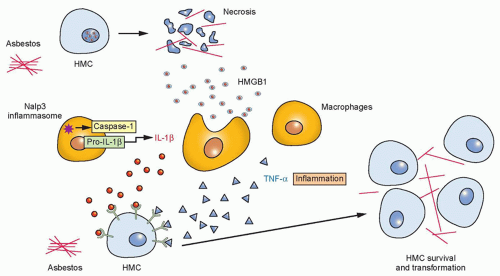
It is thought that when fibers reach the alveoli after inhalation, smaller fibers are phagocytized and efficiently removed from the lung. Larger fibers are not easily engulfed and can usually only be removed if solubilized. However, amphiboles are not soluble and thus remain in the lung. These fibers may eventually reach the pleura via the lymphatics or direct extension, where they may lead to pleural plaques, fibrosis, and mesothelioma. In the pleura, asbestos fibers may cause mutagenic changes through hydroxy radical and superoxide anion production during phagocytosis, leading to DNA strand breaks and deletions. Most recently, data implicating necrosis in the genesis of mesothelioma have been reported in which the release of HMGB1 is necessary as a critical initial step in the pathogenesis of asbestos-related disease and provides a mechanistic link between asbestos-induced cell death, chronic inflammation, and mesothelial carcinogenesis.12 Inflammation due to asbestos deposition contributes to asbestos carcinogenesis.2,13,14 Phagocytic macrophages at these sites of inflammation internalize asbestos and release mutagenic reactive oxygen species and numerous cytokines including TNF-α and interleukin (IL)-1, which have been linked to asbestos-related carcinogenesis.2,15 By inducing cell necrosis, asbestos causes the release of HMGB1,12 a typical damage-associated molecular pattern16,17 and a key mediator of inflammation.18 Activation of Nalp3 inflammasome ensues with subsequent IL-1β secretion, a process that is enhanced by asbestos-induced production of reactive oxygen species.19,20 Additionally, HMGB1 is released by reactive macrophages, other inflammatory cells, and human mesothelial cells (HMC12) upon exposure to asbestos. HMGB1 is a critical regulator in the initiation of asbestos-mediated inflammation leading to the release of TNF-α and subsequent NF-κB signaling12 (Fig. 40.1). During programmed necrosis, HMGB1 translocates from the nucleus to the cytosol and the extracellular space, where it binds several proinflammatory molecules and triggers the inflammatory responses that distinguish this type of cell death from apoptosis. Secreted
HMGB1 stimulates RAGE, TLR2, and TLR4 (the three main HMGB1 receptors) expressed on neighboring inflammatory cells such as macrophages and induces the release of several inflammatory cytokines, including TNF-α and IL-1β. TNF-α-induced NF-κB signaling has been shown to be a critical link between inflammation and carcinogenesis in multiple cancer models, including mesothelioma.2 Specifically, asbestos-mediated TNF-α signaling induces the activation of NF-κB-dependent mechanisms, promoting the survival of HMCs after asbestos exposure,2 and thus allowing HMCs with accumulated asbestos-induced genetic damage to survive, divide, and propagate genetic aberrations in premalignant cells that can give rise to a malignant clone. In addition, HMGB1 enhances the activity of NF-κB, which promotes tumor formation, progression, and metastasis.21 An investigation into the targeting of extracellular HMGB1 as a novel strategy for mesothelioma prevention and/or therapy is currently under way.22
HMGB1 stimulates RAGE, TLR2, and TLR4 (the three main HMGB1 receptors) expressed on neighboring inflammatory cells such as macrophages and induces the release of several inflammatory cytokines, including TNF-α and IL-1β. TNF-α-induced NF-κB signaling has been shown to be a critical link between inflammation and carcinogenesis in multiple cancer models, including mesothelioma.2 Specifically, asbestos-mediated TNF-α signaling induces the activation of NF-κB-dependent mechanisms, promoting the survival of HMCs after asbestos exposure,2 and thus allowing HMCs with accumulated asbestos-induced genetic damage to survive, divide, and propagate genetic aberrations in premalignant cells that can give rise to a malignant clone. In addition, HMGB1 enhances the activity of NF-κB, which promotes tumor formation, progression, and metastasis.21 An investigation into the targeting of extracellular HMGB1 as a novel strategy for mesothelioma prevention and/or therapy is currently under way.22
It is difficult to reconcile what is known about the carcinogenic actions of asbestos and the long latency period between asbestos exposure and mesothelioma development. Lanphear and Buncher23 reviewed 21 studies of 1,690 mesotheliomas and found that 96% occurred at least 20 years after exposure; the mean latency was 32 years. Several theories have been postulated to describe what happens during this latency period.24 It is widely believed that asbestos induces genetic alterations in mesothelial cells over a long period that eventually lead to malignant cells. This may occur if a key regulatory gene, such as the INK4a/ARF locus, is deleted or silenced. With loss of key regulatory genes, additional mutations could accumulate rapidly. INK4a/ARF codes for p16 (a cyclin-dependent kinase inhibitor) and for p14ARF, which promotes murine double minute-2 degradation, preventing murine double minute-2 from neutralizing p53. Both p16 and p14ARF are often deleted in MM (see “Alterations of Specific Tumor Suppressor Genes in Mesothelioma”). This process may allow enough mutations to accumulate to lead to malignancy. Cell growth from this point would be rapid, leading to a clinically detectable tumor.1
OVERVIEW OF MOLECULAR MECHANISMS IN MESOTHELIOMA
Cytogenetic Assessment of Malignant Mesotheliomas
Deletions of specific chromosomal sites in the short (p) arms of chromosomes 1, 3, and 9, and long (q) arm of 6 are repeatedly observed in these tumors. Using newer molecular platforms including array comparative genomic hybridization and representational oligonucleotide microarray analysis, deletions in chromosomes 22q12.2, 19q13.32, and 17p13.1 appear to be the most frequent events (55% to 74%), followed by deletions in 1p, 9p, 9q, 4p, and 3p, and gains in 5p, 18q, 8q, and 17q (23% to 55%). Increasing numbers of copy number abnormalities in mesothelioma are associated with poor prognosis, especially when associated with deletions in 9p21.3 encompassing CDKN2A/ARF and CDKN2B, as detailed later.24 Analysis of the minimal common areas of frequent gains and losses has pointed to novel potential tumor suppressor genes (TSG) including OSM (22q12.2), FUS1, PL6 (3p21.3), DNAJA1 (9p21.1), and CDH2 (18q11.2-q12.3).25
Alterations of Specific Tumor Suppressor Genes in Mesothelioma
p16
Cheng et al.26 reported homozygous deletions of one or more of the three p16 exons in 34 of 40 (85%) malignant mesothelioma cell lines and a point mutation in one cell line. Downregulation of p16 was observed in four of the remaining cell lines. Homozygous deletions of p16 were identified in 5 of 23 (22%) malignant
mesothelioma tumor samples. Downregulation of p16 in malignant mesothelioma cells may result from 5’CpG island hypermethylation, as has been demonstrated in other types of cancer. P16 inactivation by homozygous deletions or methylation is a frequent event in Japanese patients with malignant pleural mesotheliomas (MPM).27 Homozygous deletion is the major cause of the P16 inactivation,28 but methylation also leads to the inactivation of P16 when the P16 alleles are retained.27 Homozygous deletions at this locus can lead to the inactivation of another putative TSG, p14ARF, because p16 and p14ARF share exons 2 and 3 although their reading frames differ.29,30 The prognostic significance of p16/CDKN2A loss in MM has been defined both by immunohistochemistry and fluorescence in situ hybridization (FISH). With the FISH analysis of primary MM tissue samples or MM cells from the pleural effusion, over 70% of cases showed homozygous deletions of the CDKN2A/ARF locus. Both loss of p16 protein expression by immunohistochemistry and homozygous deletion of p16 by FISH are associated with an adverse prognosis.31 Loss of p15 is also associated with codeletion of microRNA (miR)-31.32 Protein phosphatase (PPP6C), upregulated in MPM, is a target of mir-31, and reintroduction of miR-31 in mesothelioma cells has a suppressive effect on MM cells.32
mesothelioma tumor samples. Downregulation of p16 in malignant mesothelioma cells may result from 5’CpG island hypermethylation, as has been demonstrated in other types of cancer. P16 inactivation by homozygous deletions or methylation is a frequent event in Japanese patients with malignant pleural mesotheliomas (MPM).27 Homozygous deletion is the major cause of the P16 inactivation,28 but methylation also leads to the inactivation of P16 when the P16 alleles are retained.27 Homozygous deletions at this locus can lead to the inactivation of another putative TSG, p14ARF, because p16 and p14ARF share exons 2 and 3 although their reading frames differ.29,30 The prognostic significance of p16/CDKN2A loss in MM has been defined both by immunohistochemistry and fluorescence in situ hybridization (FISH). With the FISH analysis of primary MM tissue samples or MM cells from the pleural effusion, over 70% of cases showed homozygous deletions of the CDKN2A/ARF locus. Both loss of p16 protein expression by immunohistochemistry and homozygous deletion of p16 by FISH are associated with an adverse prognosis.31 Loss of p15 is also associated with codeletion of microRNA (miR)-31.32 Protein phosphatase (PPP6C), upregulated in MPM, is a target of mir-31, and reintroduction of miR-31 in mesothelioma cells has a suppressive effect on MM cells.32
p53
Cell lines derived from murine models of asbestos-induced mesothelioma have reduced or absent expression of p53 messenger RNA (mRNA) compared to the RNA from nontumorigenic cell lines or reactive mesothelial cells.33 In human cell lines derived from patients with malignant mesothelioma, attention has turned to specific mutations or genetic abnormalities in p53. In general, mutational analyses of p53 in mesothelioma have not revealed reasons for its inactivation.
Wilms Tumor Gene
The detection of Wilms tumor gene (WT1) mRNA or protein provides a specific molecular or immunohistochemical marker for differentiation of mesothelioma from other pleural tumors, in particular adenocarcinoma.34 Vaccination protocols that exploit the selective presence of WT1 in mesothelioma are under way at selected institutions.35
Neurofibromatosis Gene
The tumor suppressor merlin is encoded by the neurofibromatosis type 2 gene (NF2), which is located on chromosome 22q12, and mutations in this gene have been found in 40% of mesothelioma and 50% of malignant mesothelioma cell lines.36,37 Mutations including deletions and insertions lead to truncated and inactivated merlin. Experimental animal models indicate that disruption of the NF2 signaling pathway, together with a deficiency in ink4a, is essential for mesothelioma development.38
ALTERATIONS OF ONCOGENES IN MESOTHELIOMA
C-sis (Platelet-Derived Growth Factor)
Malignant mesothelioma cell lines produce numerous growth factors and cytokines, possibly as a consequence of altered oncogene status. One of the more intriguing oncogenes in malignant mesothelioma is C-sis, which codes for one of the two chains (alpha and beta) of platelet-derived growth factor (PDGF). Gerwin et al.39 were the first to describe elevation of RNA levels for both chains of PDGF in mesothelioma cell lines and correlated the increase with PDGF-like activity secreted by the cells. It is of interest that overexpression of A chain transforms human mesothelial cells to the tumorigenic phenotype in vitro and that the use of antisense ODN to the A chain inhibits growth of mesothelioma cell lines. PDGF-D promotes mesothelioma cell proliferation by targeting ROCK or MAP kinase through autocrine activation of PDGF-beta receptor40; however, in general, therapies targeting PDGF in mesothelioma have been unsuccessful.41
Transforming Growth Factor
Transforming growth factor-B (TGF-B) is another growth-regulatory and immunomodulatory cytokine, and high levels of TGF-B have been described in several human and mouse malignant mesothelioma cell lines.42,43 TGF-B may have a potential role in regulation of the differential PDGF receptor expression in malignant mesothelioma and thus have an effect on proliferation as well as other events. TGF-B production by tumors plays a significant role in blocking immune response by preventing T-cell infiltration into tumors, inhibition of T-cell activation/function, and mediation of T regulatory cell-induced immunosuppression. TGF-blockers (soluble receptors/antibodies) and receptor inhibitors have antitumor effects that, in several models, are due primarily to immunologic mechanisms. TGF blockade had antitumor effects that were CD8+ T-cell dependent in a murine malignant mesothelioma model.44 Recently, it was reported that a combined triple treatment of anti-CD25 monoclonal antibody, anti-cytotoxic T-lymphocyte antigen-4 monoclonal antibody and TGF-beta soluble receptor resulted in long-term clearance of MPM tumors and memory against tumor rechallenge.45
Insulin Growth Factor
Insulin growth factor (IGF) is another of the important autocrine loops in mesothelioma.46 The IGF receptor-1 pathway is a significant regulator of mesothelioma growth acting through downstream kinases such as Akt. In humans, both normal mesothelium and mesothelioma cell lines express IGF-1 and IGF-1R RNAs (by Northern blot or reverse transcriptase polymerase chain reaction), and IGF-1 protein is detected in their conditioned media. These studies suggest that mesothelioma growth, both in human and animal models for the disease, may involve the IGFs, and trials using anti-IGF-1R antibodies or small-molecule IGF blockade are being formulated for mesothelioma.47 Cixutumumab, a humanized monoclonal antibody to IGF-IR, has been investigated using early passage tumor cells and an in vivo human mesothelioma tumor xenograft model. Cixutumumab induced antibody-dependent cell-mediated toxicity and in vivo, cixutumumab treatment delayed growth of H226 mesothelioma tumor xenografts in mice and improved the overall survival of these mice compared to controls.48 A phase 2 clinical trial of cixutumumab is currently ongoing for the treatment of patients with mesothelioma.
Angiogenic Mechanisms in Mesothelioma
The most relevant cytokines and growth factors in angiogenic pathways involve the ILs (specifically IL-6 and IL-8), fibroblast growth factors (FGF), vascular endothelial growth factors (VEGF), platelet-derived endothelial growth factors, and IGFs. Moreover, mutational or posttranslational silencing of TSGs or their products, particularly p53 and p16, has also been associated with increased angiogenic mechanisms.
IL-6 levels are elevated in the serum and pleural effusions of patients with mesothelioma, and there have been correlations seen between the IL-6 level and the degree of thrombocytosis in these patients.49 IL-6 expression is elevated in tissues undergoing angiogenesis, but by itself it has a limited role in proliferation of endothelial cells. IL-6, however, induces angiogenesis by elevating the expression of VEGF, a specific mitogen for vascular endothelial cells. Serum VEGF and IL-6 levels are elevated in
patients with mesothelioma, and their levels each correlate with platelet count.50 IL-8 is also an important angiogenic factor for the development of new capillaries in vivo and has direct growthpotentiating activity in mesothelioma. Pleural fluid from patients with mesothelioma has significantly higher IL-8 levels by enzyme-linked assay compared to patients with other malignant effusions.51 Mesotheliomas present with a high intratumoral vessel density that also have prognostic relevance,42,52,53 and immunohistochemically, VEGF, FGF-1 and -2, and TGF-B immunoreactivity are present in 81%, 67%, 92%, and 96%, respectively, of mesotheliomas, and in 20%, 50%, 40%, and 10%, respectively, of samples of the nonneoplastic mesothelium. Depending on the study, high immunohistochemical FGF-2 expression53 or VEGF expression by reverse transcriptase polymerase chain reaction correlate with more tumor aggressiveness and worse prognosis for mesothelioma.52,54
patients with mesothelioma, and their levels each correlate with platelet count.50 IL-8 is also an important angiogenic factor for the development of new capillaries in vivo and has direct growthpotentiating activity in mesothelioma. Pleural fluid from patients with mesothelioma has significantly higher IL-8 levels by enzyme-linked assay compared to patients with other malignant effusions.51 Mesotheliomas present with a high intratumoral vessel density that also have prognostic relevance,42,52,53 and immunohistochemically, VEGF, FGF-1 and -2, and TGF-B immunoreactivity are present in 81%, 67%, 92%, and 96%, respectively, of mesotheliomas, and in 20%, 50%, 40%, and 10%, respectively, of samples of the nonneoplastic mesothelium. Depending on the study, high immunohistochemical FGF-2 expression53 or VEGF expression by reverse transcriptase polymerase chain reaction correlate with more tumor aggressiveness and worse prognosis for mesothelioma.52,54
Simian Virus 40
SV40 is a DNA tumor virus that has been associated with the development of malignant mesothelioma.55 Although this virus is endogenous in rhesus monkeys, epidemiologic studies have shown that it is also widespread among the human population. The mode by which the virus was transferred from monkey to human is uncertain, but the bulk of this transfer may have occurred from 1954 to 1963 through SV40-contaminated polio vaccines administered worldwide. Moreover, polio vaccines produced in the former Union of Soviet Socialist Republics remained contaminated with infectious SV40 at least until 1978.56
The association of SV40 with mesothelioma started with the observation that when hamsters were injected with SV40 into the pleural space, all of the animals developed mesotheliomas within 6 months. This finding prompted investigations into the possibility that some mesotheliomas in humans could be attributed to SV40 infection directly or with SV40 acting as a cocarcinogen with asbestos. Mesothelioma samples studied in 1994 showed that 60% of the samples contained SV40 DNA and expressed the SV40 large T antigen. The results were confirmed by numerous laboratories using a variety of techniques such as PCR, FISH, Western blot, immunohistochemistry, and laser dissection/PCR, but the percentage of positive samples varied from 6% to 83%, and a few studies were completely negative. Technical and geographical differences may account for these variances.
Although the epidemiologic data are not available, mechanistic experiments in human mesothelial cells57,58 and animal experiments59 strongly support a pathogenic role of SV40 in mesothelioma. It is unlikely, however, that SV40 acts alone in mesothelioma development, as most cancers are multifactorial, and most mesotheliomas occur in asbestos-exposed individuals. SV40 has been shown to be a cocarcinogen in causing mesothelioma in animals and malignant transformation of mesothelial cells in tissue culture.60,61 It is safe to say that a definitive role for the virus in human mesothelioma has not been unequivocally demonstrated but ongoing debate regarding its role will continue.
Radiation and Mesothelioma
In the literature, there are a few studies reporting mesothelioma development in patients exposed to thorotrast (intravenously) or who had received radiation to the chest and abdomen.1 In some of these cases, asbestos exposure could not be ruled out, and SV40 status was not investigated. However, in a few cases in which mesotheliomas developed in young adults who received radiotherapy (RT) because of Wilms tumor, radiation was the only likely causative factor. Studies in rats support a role for radiation as a causative factor. Although it appears that radiation may cause mesotheliomas, the number of mesotheliomas for which it is responsible is probably small.
Carbon Nanoparticles
Engineered single-wall carbon nanotubes (SWCNT) are a class of nanoparticles being actively evaluated for myriad industrial and biomedical applications.62 Preliminary cellular and animal exposure investigations on the toxicity and pathogenicity of SWCNTs have demonstrated biologic interactions, including toxicity, inflammatory reactions, oxidative stress, and fibroproliferative response.63,64,65 SWCNTs are biopersistent and have the ability to distribute to subpleural areas after pharyngeal aspiration. Reports have described the induction of mesothelioma in p53+/- mice by multiwall carbon nanotubes.66,67 These investigations compelled the present studies of potential interactions of SWCNTs with mesothelial cells. Exposure to SWCNTs induced reactive oxygen species generation, increased cell death, enhanced DNA damage and H2AX phosphorylation, and activated poly(adenosine diphosphate-ribose) polymerase, AP-1, NFκB, p38, and Akt in a dose-dependent manner. These events recapitulate some of the key molecular events involved in mesothelioma development associated with asbestos exposure.68,69 In addition, nickel nanoparticles enhance the in vitro activity of PDGF in regulating chemokine production in rat mesothelial cells through a mechanism involving reactive oxygen species generation and prolonged activation of ERK-1.70
Genetic Predisposition to Mesothelioma: Erionite and BAP1
Recent evidence indicates that genetic predisposition plays an important role in determining individual susceptibility to mineral fiber carcinogenesis and to the development of mesothelioma. In the villages of Karain (population ˜600), Tuzkoy (population ˜1,400), and Sarihidir (this village was abandoned) in Cappadocia, a region in Central Anatolia, Turkey, ≥50% of deaths are caused by malignant mesothelioma.71 This epidemic has been linked to exposure to a mineral fiber called erionite, one of the most potent mineral fibers in causing mesothelioma. However, mesothelioma was more frequent in certain families than others, leading to observations that some families in Turkey were unusually susceptible to erionite carcinogenesis.71 Erionite is present in many parts of the world, including western American states such as North Dakota,72,73 but so far the contribution of erionite to the increased incidence of mesothelioma has not been studied except in Turkey.
Similar families have also been identified in other parts of the world74 and in the United States, where they appear to be unusually susceptible to asbestos carcinogenesis. Recently, BRCA-associated protein 1 (BAP1) has been identified as a novel MPM TSG. BAP1 is located at the 3p21, a region frequently deleted in MM, and encodes for a deubiquitinase enzyme known to target histones and other proteins. BAP1 appears to exert its antitumor activities mainly in a BRCA-independent manner,75 with regulation of the cell epigenome, via modulation of histone H2A ubiquitination and chromatin accessibility.76 However, the relevance of BAP1 to the biology of normal and cancer cells remains largely unexplained; in fact, manipulation of BAP1 in cancer cells has often yielded unexpected or even contradictory results. For example, silencing of BAP1 in MM and uveal melanoma cell lines resulted in reduced cell growth.77,78
Bott et al.77 originally described the association of somatic mutations of BAP1 in 23% of mesothelioma samples. In a larger series of patients, the same group confirmed that somatic BAP1 mutations occur in about 20% of pleural MM and reported that the only clinical variable significantly different among those with and without BAP1 mutations was smoking (former or current), with BAP1 mutations more prevalent among smokers (75% versus 42%).79 A Japanese study reported BAP1 gene alterations (either deletions or sequence-level mutations) in 61% of their 23 MM samples.80 Whether this discrepancy results from the different methodologies
in sample preparation and detection of BAP1 mutations or it is an intrinsic difference between the two populations (e.g., due to ethnicity) has still to be determined. A third recent study from Arzt et al.,81 with a separate cohort of 52 pleural MM, reported absence of BAP1 IHC staining in 60% of pleural MM, confirming previous results. Moreover, there was no correlation between BAP1 expression and asbestos exposure, and it was suggested that expression of BAP1 in tumor samples is inversely correlated to survival.
in sample preparation and detection of BAP1 mutations or it is an intrinsic difference between the two populations (e.g., due to ethnicity) has still to be determined. A third recent study from Arzt et al.,81 with a separate cohort of 52 pleural MM, reported absence of BAP1 IHC staining in 60% of pleural MM, confirming previous results. Moreover, there was no correlation between BAP1 expression and asbestos exposure, and it was suggested that expression of BAP1 in tumor samples is inversely correlated to survival.
Germline BAP1 mutations were reported by Testa and Carbone82 in two US families having a high frequency of MPMs. In the same study, 22% sporadic MM tumors harbored somatic BAP1 mutations. These findings have promoted speculation that BAP1 germline mutations can cause a novel cancer syndrome characterized by a significant excess of both pleural and peritoneal MM, uveal and cutaneous melanoma, and possibly other tumors.
PATHOLOGY OF MESOTHELIOMA
Benign Mesotheliomas
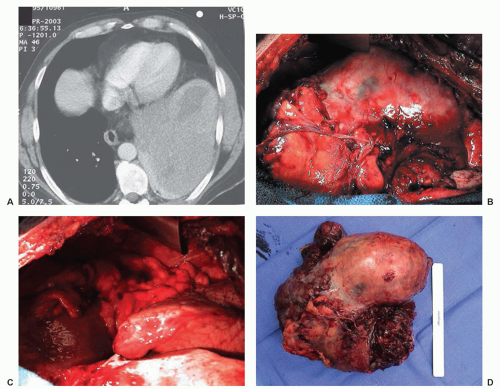
There are a number of benign mesothelial proliferations that must be distinguished from malignant mesothelioma including multicystic mesothelioma (multilocular peritoneal inclusion cyst), adenomatoid mesotheliomas, and well-differentiated papillary mesothelioma. Localized mesothelioma, better referred to as localized fibrous tumor of the pleura (FTP), is similar to other fibrous tumors found elsewhere in the body. The tumor cells have a benign appearance and are usually immersed in a fibrous and characteristically vascular stroma. FTPs are characteristically negative for cytokeratin (a marker of mesothelial cells) and positive for CD34, which suggests that these cells are not of mesothelial origin. The most important predictive factor in the prognosis of FTP is whether the tumor can be completely resected. Pedunculated tumors have a much better prognosis than tumors that grow over a broad pleural area.83,84,85 Surgical resection (Fig. 40.2) is the treatment of choice, with complete resection of the tumor and its pedunculated portion along with the site of origin. Five-year survival rates as high as 97% have been reported. En bloc resection of the lung, chest wall, or diaphragm may be necessary. For recurrences, re-excision is the treatment of choice. Long-term survival with re-excision is possible, but with incomplete resection or malignant transformation the median survival is 24 to 36 months.
Malignant Mesothelioma
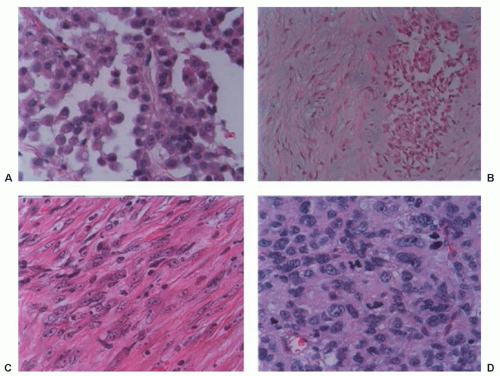
Histologically, malignant mesothelioma can show an epithelial morphology (malignant mesothelioma epithelial type), a fibrous morphology (malignant mesothelioma fibrous type, also called sarcomatoid type), or a combination of both (mixed type or biphasic malignant mesothelioma) (Fig. 40.3). Most MM (50% to 60%) are of the epithelial type, approximately 10% are sarcomatoid, and the remainder are biphasic MM.
The diagnosis of malignant mesothelioma is usually straightforward, in contrast to common belief, provided that the pathologist
has extensive experience with this malignancy. Some epithelial mesotheliomas grow forming sheets of epithelioid cells. To rule out mimics of mesothelioma, the diagnosis should be confirmed by immunohistochemistry, which shows that the tumor cells are diffusively positive for pankeratin, keratin 5/6, calretinin, and WT-1, and negative for the epithelial markers such as CEA, CD15, Ber-EP4, Moc-31, TTF-1, and B72.3. For epithelial mesotheliomas, positive stainings for pankeratin and calretinin and negative stainings for three epithelial markers are considered sufficient for diagnosis.
has extensive experience with this malignancy. Some epithelial mesotheliomas grow forming sheets of epithelioid cells. To rule out mimics of mesothelioma, the diagnosis should be confirmed by immunohistochemistry, which shows that the tumor cells are diffusively positive for pankeratin, keratin 5/6, calretinin, and WT-1, and negative for the epithelial markers such as CEA, CD15, Ber-EP4, Moc-31, TTF-1, and B72.3. For epithelial mesotheliomas, positive stainings for pankeratin and calretinin and negative stainings for three epithelial markers are considered sufficient for diagnosis.
Biphasic mesotheliomas can be distinguished from carcinosarcomas metastasizing from various organs and from biphasic synovial sarcomas by using either electron microscopy or molecular testing for the X;18 translocation that is diagnostic of synovial sarcoma.86
Newer methods are under development for distinguishing mesothelioma from adenocarcinoma and include microRNA profiling of the tumors. Gee et al.87 have described the use of seven miRNAs (miR-200c, miR-141, miR-200b, miR-200a, miR-429, miR-203, and miR-205) to distinguish lung adenocarcinoma from mesothelioma with a 10% misclassification error. Others have described a three microRNA profile (mir-205, mir-193a-3p, and mir-192) for the differential diagnosis of mesothelioma from colon, renal, lung, and breast cancer with a 95% specificity and 96% sensitivity.88
EPIDEMIOLOGY AND CLINICAL PRESENTATION
The death rates for mesothelioma parallel the consumption of asbestos by individual countries. When one considers mesothelioma death rates from 2000 to 2004 as a function of asbestos usage in the 1960s, Australia has the highest death rate for the disease in the world.89 It is predicted that between 2000 and 2049, over 400,000 lives will be lost in those countries from the disease.90 In the United States, there have been 2,500 deaths per year from the disease since 1999, with workers with industrial exposures in construction and shipbuilding being at highest risk, followed by plumbers, pipe fitters, and steamers. Mesothelioma affects men in their 50s, 60s, and 70s due to the aforementioned 25- to 40-year latency period between occupational asbestos exposure and the development of the tumor.91 Women and children can have the disease, but the male-to-female ratio is approximately 4.5:1.92
Symptoms
As many as 25% of patients with the disease have symptoms for ≥6 months before seeking medical attention. The right side is affected more than the left side (60% versus 40%), most likely because of the right side’s greater volume.
Approximately 60% of the patients present with nonpleuritic chest pain that classically is located posterolaterally and low in the thorax. Dyspnea is present in 50% to 70% of the cases, and, indeed, 80% of the patients present with dyspnea and effusion. The presence of a pleural effusion is documented at some time in the course of the disease in 95% of patients with MPM. With the increasing use of positron emission tomography for pretreatment planning, between 5% and 25% of patients are found to have metastatic disease at presentation.93
Physical Examination
Physical examination reveals decreased breath sounds, dullness to percussion, or decreased motion of the involved chest wall. Failure to significantly relieve the dyspnea after thoracentesis may indicate that the lung is “trapped.” In the late stages of the disease, there is often dramatic cachexia, marked contraction of the involved chest with narrowed interspaces, and hypertrophy of the contralateral hemithorax. A chest wall mass occurs in up to 25% of patients, often at the site(s) of prior thoracentesis, thoracotomy, or thoracoscopy wounds.
Laboratory Examination and Blood Biomarkers
Mesothelioma patients have nonspecific laboratory findings, including hypergammaglobulinemia, eosinophilia, and/or anemia of chronic disease. It has been recently noted that 14% to 15% of patients have elevated homocysteine levels, reflecting folic acid deficiency; 17% have biochemical evidence of vitamin B12 deficiency; and 32% have biochemical signs of vitamin B6 deficiency.94 The most striking laboratory abnormality is thrombocytosis (>400,000), which is seen in 60% to 90% of patients,95 and approximately 15% of patients have platelet counts >1,000,000.
Soluble mesothelin-related peptide (SMRP), osteopontin (OPN), and megakaryocyte potentiating factor are presently under investigation as early detection or therapy monitoring markers. Robinson et al.96 reported that determination of SMRP in serum is a marker of mesothelioma with a sensitivity of 83% and specificity of 95% in the first 48 patients with MPM tested. These data have been validated by other laboratories in the United States97,98 and internationally.99 Changes in serum SMRP levels parallel clinical course/tumor size, and SMRP was elevated in 75% of patients at diagnosis.100 Studies using SMRP in an attempt to diagnose early mesothelioma in high-risk cohorts have so far been disappointing, but follow-up is still short.101,102
OPN is a glycoprotein that mediates cell-matrix interactions and is regulated by proteins in cell-signaling pathways that have been associated with asbestos-induced carcinogenesis.103 In a study that compared serum OPN levels of patients with MPM to those of asbestos-exposed individuals without cancer, serum OPN levels rose with duration of asbestos exposure and degree of radiographic abnormality (plaques and fibrosis versus other lesser findings104), and using a cutpoint of 48.3 ng of OPN per milliliter resulted in 77.6% sensitivity and 85.5% specificity when comparing the group exposed to asbestos with the group with mesothelioma. Follow-up studies have demonstrated that serum OPN is not reliable because of the presence of a thrombin cleavage site, which causes degradation of the marker.105 Plasma OPN,106 although not specific to mesothelioma, is being evaluated in the disease for both prognosis and early detection utility. A blinded validation of plasma fibulin 3 confirmed excellent discrimination between MPM plasma and all non-MPM cohorts including those with benign or malignant effusions. Pleural effusion fibulin 3 was also discriminatory between MPM effusions and non-MPM effusions.107
Radiologic Examination
Malignant mesothelioma can have a diverse radiographic appearance. Many of the early changes are associated with a previous exposure to asbestos, consisting both of pleural and parenchymal changes, including pleural plaques or parenchymal pulmonary fibrosis.
Chest Radiography
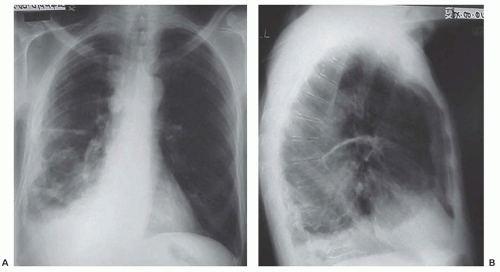
The most common chest radiography features associated with mesothelioma progression and symptoms include the presence of a pleural effusion, diffuse pleural thickening, and nodularity. The involved hemithorax can eventually have smooth, lobular pleural masses that infiltrate the pleural space and fissures108,109,110 in 45% to 60% of patients with contraction and fixation of the chest (Fig. 40.4). The lung becomes encased, and the mediastinum shifts because of volume loss.
Chest Tomography
Pleural changes on chest tomography (CT) include pleural plaques, diffuse pleural thickening, and pleural effusion. Additional CT features of mesothelioma are localized nodular or plaque-like pleural thickening, possibly associated with pleural effusion. The lobulated pleural encasement frequently causes lower lobe collapse (Fig. 40.5). Intrapulmonary nodules can occur in 60% of patients, and infiltration into fissures along with enlarged hilar and mediastinal lymph nodes may be seen. The CT allows a better view of the involved pericardium, which is irregularly thickened and associated with infiltration to the pericardial fat pad. A clear fat plane between the inferior diaphragmatic surface and the adjacent abdominal organs as well as a smooth inferior diaphragmatic contour may imply resectability.111 CT may reveal a hemidiaphragm encased by a mass or poor definition between the liver, stomach, and inferior diaphragmatic surface.
Volumetric measurements using CT have recently been studied with regard to their prognostic capability (see “Prognostic Indicators”).
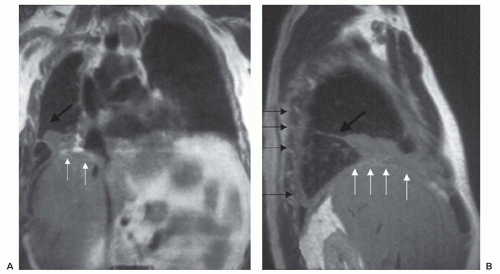
Magnetic Resonance Imaging
Positron Emission Tomography Imaging
There are a number of studies of positron emission tomography (PET) and the radionuclide imaging agent [18F]fluorodeoxyglucose (FDG) in mesothelioma (Fig. 40.7). Early studies reported that FDG-PET was accurate in the diagnosis of pleural malignancies, specifically mesothelioma.112,113,114,115,116 Recent studies have concentrated on the accuracy of PET in defining nodal and extrathoracic metastases prior to surgical therapy.93,112 Because of false-positive results in patients with FDG-avid pleural inflammatory/infectious etiologies, all FDG-avid extrathoracic sites should be histologically confirmed in patients with MPM being considered for extrapleural pneumonectomy (EPP).
DIAGNOSTIC APPROACH FOR PRESUMED MESOTHELIOMA
Thoracentesis and Closed Pleural Biopsy
Patients who present with a large, unexplained pleural effusion and minimal or moderate evidence of pleural thickening should have initial thoracentesis and pleural biopsy, especially if a history of asbestos exposure or mesothelioma in a family member is elicited. By preserving a cell block from the pleural fluid and using both histochemical
and immunohistochemical staining techniques along with electron microscopic analysis, the diagnosis of mesothelioma can be obtained from the pleural effusion in as high as 84% of suspected cases.
and immunohistochemical staining techniques along with electron microscopic analysis, the diagnosis of mesothelioma can be obtained from the pleural effusion in as high as 84% of suspected cases.
Thoracoscopy
A video-assisted thoracoscopy is indicated for patients at risk for mesothelioma who (1) develop a large effusion and who have negative studies on thoracentesis and pleural biopsy, or (2) recur with effusion after initial thoracentesis. Thoracoscopy can be invaluable for estimating extent of disease with regard to the lung, diaphragm, pericardium, chest wall, and nodes.117,118 Development of chest wall masses from seeding of the biopsy site or surgical scar is an uncommon complication (approximately 10%) of any diagnostic procedure, and the efficacy of radiotherapy to the scar to prevent recurrence is controversial.119,120,121
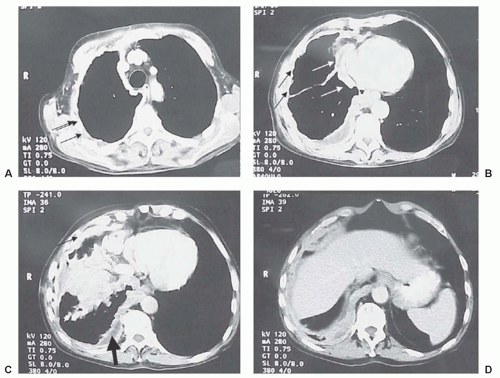
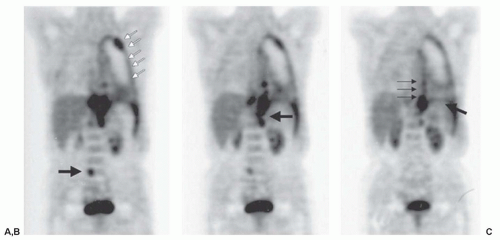 Figure 40.7 Positron emission tomography with [18F] fluorodeoxyglucose in a patient with an unresectable stage IV mesothelioma. (A) White arrows outline the involved pleura, with an area of bulky disease superiorly. Black arrow points out an occult metastasis in the abdomen. (B) Black arrow depicts posterior infracrural mediastinal lymph node disease. (C) Bold black arrow points out diaphragmatic involvement. Thin black arrows depict mesothelioma medially.
Stay updated, free articles. Join our Telegram channel
Full access? Get Clinical Tree
 Get Clinical Tree app for offline access
Get Clinical Tree app for offline access

|