Bartonella
Epidemiology and risk factors
Bartonella species are fastidious, slow-growing, gram-negative bacilli. There are 45 different species that can cause disease in zoonotic hosts, with Bartonella henselae and Bartonella quintana causing most Bartonella -related disease in human hosts in the United States. B. henselae has been reported to cause infection in pediatric and adult heart, liver, and renal transplant recipients and in adult lung transplant recipients. There have been rare reports of B. quintana causing disease in adult solid organ transplant (SOT) recipients. Both species have been reported to cause infection in pediatric patients undergoing chemotherapy for hematologic malignancies.
Cat-scratch disease (CSD), a syndrome of regional lymphadenopathy or granulomatous disease in the liver and spleen, is the most widely recognized illness attributed to B. henselae . The highest incidence of CSD in the United States is reported in people who live in the Southeast and among children 5 to 9 years of age. Granulomatous disease/CSD is the most common presentation of B. henselae in SOT recipients and is found in 19 of 32 (59%) reported cases. B henselae infection disproportionately affects children. Moulin and colleagues reported that 25% of the cases of Bartonella infection in transplant recipients occurred in children younger than 18 years, despite pediatric patients representing only 3% to 4% of the overall transplant population.
Bacillary angiomatosis (BA) and bacillary peliosis (BP) are vasoproliferative manifestations of both B. henselae and B. quintana in immunocompromised hosts. They are most commonly described in adults with very low CD4 + lymphocyte counts caused by human immunodeficiency virus (HIV). Cases have been described in pediatric and adult SOT recipients and those with hematologic malignancies. All patients reported with BA associated with cancer have had full resolution of their disease with treatment. Interestingly, in SOT recipients, BA tends to develop on average 1.9 years after transplantation, whereas granulomatous disease/CSD develops 5.2 years after transplantation. The reason for this is unknown but may be explained by greater immunosuppression close to the time of transplant, predisposing patients to vasoproliferative manifestations.
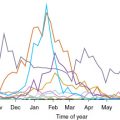
B. henselae causes bacteremia in cats, its natural reservoir. The seroprevalence of B. henselae is 13% to 90% in domestic and stray cats in the United States. Other animals, including dogs, can become infected and have been associated with human disease. Kittens and stray or sheltered cats are more likely to have bacteremia, which can last for weeks to months. Despite this, cats are usually asymptomatic. Transmission between cats occurs through the cat flea, whereas transmission to humans occurs through the lick, bite, or scratch of a bacteremic cat; the claws of the cat are thought to be contaminated by the feces of Bartonella -infected fleas. The incubation period from scratch to the appearance of a cutaneous lesion is 7 to 12 days. Lymphadenopathy usually occurs 5 to 50 days, with a median of 12 days, after the scratch.
There have been no cases of person-to-person B. henselae transmission. One case of possible donor-derived transmission has been reported in an asymptomatic liver transplant recipient with granulomatous hepatic lesions appearing 2 months after transplant. Subsequent 16S PCR of DNA from the lesion was positive for B. henselae . This patient had no reported contact with cats, raising the possibility of donor-derived infection.
B. quintana is closely related to B. henselae and is more commonly associated with louse-borne trench fever. Risk factors for infection with B. quintana include homelessness, chronic alcoholism, and body lice.
Clinical manifestations
In general, Bartonella -mediated disease in transplant recipients can be divided into two distinct groups: cat-scratch disease (typical and disseminated) and BA and BP.
Cat-scratch disease commonly occurs in immunocompetent children as a self-limited febrile illness characterized by a cutaneous papule at the site of a cat scratch and accompanied by regional lymphadenopathy. Cases of this “typical” regional presentation of CSD have also been reported in pediatric SOT recipients. , Lymph nodes are generally tender and can have central necrosis and suppuration. Nodes may regress spontaneously within 2 to 4 months in immunocompetent hosts, but usually respond promptly to antimicrobial therapy in SOT patients. Systemic symptoms are mild in immunocompetent hosts, whereas transplant recipients can present with fever, fatigue, myalgias, joint pain, and night sweats. Importantly, although fever is the most common symptom present in any manifestation of Bartonella disease in SOT recipients, the absence of fever does not rule out this infection. At least two cases of B. henselae infection without fever have been reported in children, , with another two cases reported in adults, one of whom died from infective endocarditis.
Lymphadenopathy can also be accompanied by hepatosplenic lesions, concerning for disseminated disease in SOT recipients. Children often present with prolonged fever, abdominal pain, joint pain, headache, weight loss, and chills. Immunocompromised patients may have splenic or hepatic enlargement or both, and abdominal imaging usually identifies hypodense lesions in either or both organs. Transaminase levels often remain normal. In almost all cases, cat, kitten, or flea exposure is given in the history. Recurrence has been reported, with one case of recurrent cat-scratch lymphadenitis described in a pediatric kidney transplant recipient. More unusual manifestations of CSD in immunocompetent hosts include endocarditis, osteomyelitis, encephalopathy, retinitis, optic neuritis, and Parinaud oculoglandular syndrome. A case of posterior uveitis and one case of pulmonary nodules caused by B. henselae have been described in pediatric kidney transplant recipients. Acute rejection of the renal allograft has been reported in conjunction with Bartonella infection in two children. Reports in adult renal transplant recipients have described a patient with sternal abscess and one with septic shock, encephalopathy and seizures, and bacteremia has been described in a hematopoietic stem cell transplant (HSCT) recipient.
BA and BP vasculoproliferative cutaneous or subcutaneous lesions are due to either B. henselae or B. quintana . Lesions of BA are most commonly seen on the skin but may involve subdermal structures, bones, and mucous membranes of the mouth, conjunctivae, and the gastrointestinal tract. BP lesions may appear as hemorrhagic parenchymous and cystic lesions usually seen in the liver or spleen. B. henselae is almost exclusively associated with lesions in the lymph nodes and the liver, whereas bone lesions are more associated with B. quintana . Children may be systemically asymptomatic, or more commonly, present similarly to those with CSD with systemic symptoms of fever, abdominal pain, anorexia, myalgias, nausea, vomiting, weight loss, night sweats, and weakness. Liver and spleen lesions, lymphadenopathy, and hepatomegaly have also been reported. , Vasculoproliferative cutaneous lesions can be papular, nodular, or vascular; are usually red or violaceous in color; and may be hemorrhagic, ulcerating, or have a collarette of scale. They can be solitary or multiple and may grossly resemble Kaposi sarcoma. Recurrence of BA is seen in HIV-positive patients and has been described in a pediatric renal transplant recipient despite prolonged therapy for the first episode. ,
Providers should also have a low index of suspicion for secondary hemophagocytic lymphohistiocytosis in transplant recipients with Bartonella infection, as it has been described in both children and adults. , Pancytopenias, elevated transaminase levels, low fibrinogen level, and a very elevated ferritin level should prompt the clinician to consider this diagnosis. Treatment of the underlying cause is prudent, but patients may also require corticosteroid therapy. Consultation with a hematologist may be warranted.
Disease prophylaxis/prevention
Because cats and kittens carry B. henselae and because cat fleas play a major role in cat-to-cat transmission, it is prudent to discuss pet ownership with transplant recipients and their families. They should be educated about obtaining and caring for pets, particularly cats and kittens. All pets should be seen regularly by a veterinarian and before introduction of the pet into the home. Flea control is essential. New pets should not be introduced during times of heightened immunosuppression (immediate posttransplant period or during treatment for rejection). Immunocompromised hosts should avoid all contact with cats younger than 1 year, stray cats, cats with fleas, or cats that bite or scratch. Although declawing of cats is not routinely recommended, patients should not engage in behavior that would cause a scratch or bite. If a scratch or bite should occur, it should be cleaned immediately and thoroughly. Testing cats for Bartonella infection is not recommended because cats can be transiently bacteremic. Good hand hygiene is always encouraged, especially after petting or caring for cats or kittens.
Diagnosis
The differential diagnosis of cat scratch disease includes other causes of lymphadenopathy and systemic symptoms in transplant recipients: cytomegalovirus, Epstein-Barr virus, and posttransplant lymphoproliferative disease, lymphoma, fungal or mycobacterial infections, and pyogenic abscesses. BA may mimic Kaposi sarcoma, pyogenic granuloma, or angiosarcoma. Bartonella infection should be considered in any transplant recipient with cat or cat flea exposure who presents with unexplained fever, culture-negative endocarditis, granulomatous or necrotic regional or disseminated lymphadenopathy, BA or BP, new hepatomegaly, new splenomegaly, or hepatosplenic lesions.
Confirmatory diagnosis can be challenging in immunosuppressed patients. The indirect immunofluorescent antibody assay for detection of serum antibodies to antigens of Bartonella species is available at many commercial laboratories and through the Centers for Disease Control and Prevention. Immunoglobulin (Ig) M production is brief and may easily be missed. Immunofluorescent antibody IgG titer greater than 1:256 is consistent with acute infection, although lower titers (>1:64) have been used for diagnosis. Documentation of a fourfold increase in IgG titers can also be suggestive of recent infection. In one review of the literature, of 23 SOT patients with Bartonella infection who had serologic testing performed, all were at least IgG or IgM positive initially or had evidence of a fourfold increase in titers on follow-up testing. The sensitivity of the indirect fluorescence antibody test in immunocompromised patients is lower than in immunocompetent hosts (75% vs. 82-95%). Furthermore, serologic test results can be negative early in the course and may take 2 to 4 weeks to develop. , In some cases, patients may never mount a response, even when serology is obtained later in the course. It is important to note that cross-reactions between the different Bartonella species exist, as well as with other zoonoses such as Rickettsiae.
Histologic examination of lymph nodes from affected patients can reveal lymphocytes with epithelioid granulomas, which may later in the course have central zones of necrosis or appear suppurative; some may contain stellate microabscesses. Warthin-Starry or Steiner silver stain demonstrates aggregates of small coccobacilli. This test, however, is not specific for B. henselae . Immunohistochemistry of lymph node tissue with B. henselae –specific antibodies may also reveal evidence of infection.
On biopsy, BA of lymph nodes or skin contains a dense vascular proliferation with plump endothelial cells that protrude into the vascular lumina. There is often a mixed inflammatory infiltrate of both lymphocytes and neutrophils. Biopsies of BP lesions differ from those in BA. Lesions often demonstrate dilated capillaries and cystic blood-filled spaces scattered throughout the hepatic or splenic parenchyma. As do biopsies of lymph nodes in typical CSD, both BA and BP demonstrate clusters of bacilli seen on Warthin-Starry or Steiner silver stain.
Identification of Bartonella in cultures is difficult due to its fastidious growth and culture techniques that are not sensitive. Culturing of Bartonella can require 1 to 4 weeks of incubation on blood agar plates under specific conditions. If culture is performed, specialized laboratories with experience in isolating Bartonella organisms are recommended for processing of cultures.
Polymerase chain reaction (PCR) of DNA extracted from tissue may confirm species identity. PCR assays are available commercially and in research settings for testing of tissue or body fluids, including blood. PCR of tissue has been used to determine the etiology from lymph nodes, hepatosplenic lesions, and lung nodules in immunocompromised hosts. This technique is both sensitive and specific.
Treatment
The need for treatment of CSD in the immuocompetent host is not well established. In the only prospective, randomized trial of treatment of typical CSD in immunocompetent children, there was no clinical difference except in lymph node size at 30 days between those treated with azithromycin versus placebo.
Because severe, progressive, disseminated disease can occur in immunocompromised patients, treatment for Bartonella -associated infections is always recommended in this population. There are no established treatment guidelines for Bartonella infection in transplant recipients. Unlike in immunocompetent hosts, response to treatment is usually prompt in the immunocompromised patient. Pediatric transplant recipients with granulomatous or suppurative disease, including hepatosplenic lesions, have been successfully treated with single agents or combinations of agents: aminoglycosides (gentamicin, amikacin), macrolides (azithromycin, erythromycin), tetracyclines (doxycycline), fluoroquinolones (ciprofloxacin), and trimethoprim-sulfamethoxazole. Durations of therapy have ranged from 2 weeks to 6 months. In most cases, therapy was discontinued when all lymphadenopathy and/or hepatosplenic lesions had resolved. Shorter therapy has been associated with at least one reported recurrence of lymphadenitis in a pediatric kidney transplant recipient.
Azithromycin, doxycycline, and ciprofloxacin have been used successfully in pediatric transplant recipients with BA treated with a 3- to 4- month duration of therapy. In adult SOT recipients, reported length of therapy has ranged from 4 weeks to 6 months, which is similar to reported guidelines for HIV-infected patients with BA. In presumed disseminated CSD and BA or BP, prolonged therapy is generally administered (for at least 3 months and until lesions resolve) with a macrolide antibiotic or tetracycline, either alone or in combination with another efficacious antimicrobial, such as rifampin, an aminoglycoside, or ciprofloxacin. It is important to note, however, that children younger than 8 years in whom a macrolide may be given, doxycycline is not recommended for more than 21 days. Clinicians should also be aware that a Jarisch-Herxheimer reaction may develop after the first few doses of treatment of BA.
Drug-drug interactions must be considered in SOT recipients. Macrolides can increase the serum concentration of some calcineurin inhibitors like tacrolimus, and rifampin is a potent hepatic enzyme inducer and interacts with many drugs.
In addition to antimicrobial therapy, a decrease in immunosuppression is recommended if possible.
Infection prevention and anticipatory guidance
Standard precautions are recommended for children with Bartonella infections.
Legionella
Epidemiology and risk factors
The term “Legionnaires’ disease” (LD) was first used to describe an outbreak of pneumonia that occurred among members of an American Legion convention in Philadelphia in 1976. Eventually, Legionella pneumophila was identified as the etiologic agent responsible for the outbreak, and LD became the name given to pneumonia acquired by susceptible people through inhalation of aerosols that contain Legionella species.
Legionella is a fastidious gram-negative bacillus, of which there are at least 60 different species. There were 6141 cases of legionellosis reported in the United States in 2016, more than a fourfold increase in reported cases since 2000. The most common species that cause disease in the United States is L. pneumophila serogroup 1. Other serogroups and species are also pathogenic and have been reported to cause disease in children, including L. micdadei, L. dumoffii , and L. bozemannii . L. longbeachae has been described in patients in the western United States who have had exposure to potting soil. It is the second most common cause of legionellosis in Australia and has been reported in adult transplant recipients.
Legionellosis is a rare cause of both community-acquired and nosocomial-acquired pneumonia in children. Between 2000 and 2009, 1% of cases reported in the United States were in children 19 years and younger. The incidence of legionellosis in immunocompromised children is unknown, but there have been numerous cases reported sporadically and in health care–associated outbreaks.
Risk factors for disease fall into two broad categories: those that increase exposure to contaminated water sources (well water and water in large buildings such as hotels or hospitals) and those with impaired pulmonary or immune defense mechanisms, especially transplant recipients, children with hematologic malignancies, and those who use glucocorticoids. Both are identified as risk factors in the cases reported in the pediatric population. There have been no reports of transmission between patients and health care providers or between patients and other patients, although there has been one report of possible transmission between a son and his mother who cared for him in a nonventilated residential room for several hours. Incubation periods have ranged from 2 to 19 days, most commonly 2 to 10 days.
Clinical manifestations
Legionellosis often manifests as a severe pneumonia in immunocompromised children. Symptom onset is often abrupt. Children tend to have fever, which may be the only initial symptom, but respiratory symptoms eventually become more prominent. Cough is the most frequent symptom and can be accompanied by dyspnea, tachypnea, and pleuritic chest pain. Hemoptysis is infrequently reported. Legionellosis in immunosuppressed patients may rapidly progress to respiratory failure. Hypoxia and abnormal finding on lung examination are the most common signs. Chills, abdominal pain, nausea and vomiting, headache, malaise, anorexia, and/or fatigue may occur. Diarrhea is a common extrapulmonary finding in adults but is infrequently reported in children. , There have also been case reports in adults of pericarditis, myocarditis, endocarditis, arthritis, and central nervous system manifestations.
Without appropriate antibiotic therapy, immunocompromised hosts often progress to severe respiratory compromise, multiorgan failure, and eventual death with a reported mortality rate in immunosuppressed children of 42%. In fact, delay in diagnosis has been considered a risk factor for increased morbidity and mortality in adults. Empyema or cavitation can occur. Extrapulmonary manifestations rarely occur in children, including Legionella -associated transverse myelitis and liver abscesses.
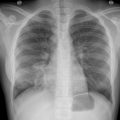
Unlike the interstitial pneumonias caused by other “atypical bacteria,” radiographic findings of LD are more often lobar consolidations with or without pleural effusion. Bilateral and/or nodular infiltrates may occur, mimicking invasive fungal infection, mycobacterial infection, or Nocardia . When nodules are present, patients may be asymptomatic. Cavitation can occur for up to 14 days after presentation, despite appropriate antibiotic therapy. Abscess can also occur. Chest computed tomography often reveals a mixture of ground-glass opacities and consolidation. Later in the course, an organizing pneumonia pattern may also be seen.
Nonspecific abnormal laboratory results are common and may not help in the diagnosis. Leukocytosis, leukopenia, lymphopenia, and thrombocytosis can be seen. Elevated transaminase levels and renal dysfunction are common. Recurrence can occur and has been noted in children after HSCT.
Disease prophylaxis/prevention
Minimizing Legionella growth in large buildings and hospital water systems, including potable water, showers, hot tubs, decorative fountains, and cooling towers, is key to prevention of this infection. A word of caution should be given to transplant recipients who travel or who will be around fountains or other manmade water sources that may aerosolize water droplets.
Diagnosis
Pneumonia is common in children and it may be difficult to distinguish between pneumonia caused by Legionella and pneumonia caused by other etiologies. Lobar consolidation may easily be confused with other bacterial causes of pneumonia, whereas those with nodular infiltrates or cavitation may be difficult to distinguish from fungal or mycobacterial infection. Mycoplasma , psittacosis, and Q fever may be suspected but tend to have an interstitial pattern on chest radiographs. If the respiratory symptoms are accompanied by systemic symptoms, viral causes such as influenza may also be considered.
Laboratory diagnosis can be challenging, and clinicians must have a high index of suspicion to ensure appropriate testing. Most assays and culture methods must be requested because they are not considered routine at many institutions. Culture of Legionella from sputum and other lower respiratory tract specimens remains the gold standard and allows for the diagnosis of all Legionella species, outbreak investigations, and antimicrobial susceptibility testing. However, culture requires special media (buffered charcoal yeast extract supplemented with α-ketoglutaric acid) and is not time efficient (it may take up to 7 days to grow). Sensitivity also varies widely (20% to 80%) and depends on the type of sample. Blood and pleural fluid cultures have very low yield. Urinary antigen detection allows for the most rapid method of diagnosis, but this test detects only L. pneumophila serogroup 1, and therefore results may be negative in disease caused by other species or other serogroups. It does have a high specificity and sensitivity for legionellosis caused by serogroup 1 and should be performed in any transplant recipient with pneumonia for which an underlying etiology is not readily apparent.
Detection of L. pneumophila antigen from lung tissue or sputum by immunofluorescence can be performed within 1 to 2 hours and is highly specific (99%) but may be insensitive (25% to 75%). Appropriate diagnosis requires expertise and training of laboratory personnel and many laboratoriess do not perform this test.
Detection of Legionella DNA in respiratory tract specimens and urine can be done by PCR and may detect other serogroups and species. Sensitivity and specificity are excellent, but different laboratory assays vary. Additionally, no commercial laboratories in the United States offer PCR testing for specific Legionella species.
Serologic diagnosis is not helpful in immunocompromised patients. The average time to seroconversion in immunocompetent patients is 2 weeks. Measuring convalescent serum is delayed and therefore is not efficient for acute case diagnosis and initiation of therapy. Finally, seroconversion may not occur at all in the immunosuppressed patient.
Treatment
Adequately sized prospective clinical studies of antimicrobial therapy for legionellosis are lacking. Newer macrolides such as azithromycin can be considered for therapy in immunocompromised children. A dosage of 10 mg/kg once daily (maximum dose 500 mg) for 5 to 10 days is recommended. Azithromycin is available in intravenous and oral forms and approved by the U.S. Food and Drug Administration for the treatment of legionellosis in adults. Macrolides often interact with immunosuppressive therapies such as tacrolimus, and their use may prove challenging in the transplant population. Fluoroquinolones also have efficacy in vitro and in vivo, and levofloxacin is the drug of choice for Legionella pneumonia in adult transplant recipients. It is approved by the Food and Drug Administration for the treatment of LD in adults. Length of therapy should be 14 to 21 days with a quinolone because of the shorter half-life compared with azithromycin. Improvement in most patients occurs within a few days to a week after starting therapy.
One observational study of non-immunocompromised adults with legionellosis compared those who received levofloxacin with those who received erythromycin or clarithromycin therapy. The levofloxacin group had a significantly shorter time to defervescence and a shorter length of hospital stay. Although combination antibiotic therapy has been reported, there is no clear benefit to this beyond what is achieved with use of a fluoroquinolone or a macrolide alone.
Infection prevention and anticipatory guidance
Standard precautions are recommended for children with legionellosis. Multiple nosocomial and community outbreaks of legionellosis have been reported. Reservoirs include potable water distribution systems, air conditioning cooling towers, hot tubs, decorative fountains, and others. Hospital water systems should maintain hot water at the highest temperature allowable, usually stored at a minimum of 60°C (140°F) and with a minimum returned temperature of 51°C (124°F). If even a single case of legionellosis is detected, an epidemiologic and environmental investigation is warranted.
Hospitals with transplantation programs should maintain a high index of suspicion for legionellosis. Using sterile water for nebulization equipment is recommended. Periodic culturing of the hospital’s potable water system should also be considered, particularly after construction has been done or hospital additions built. Outbreaks have been halted by emergency superheating of water to 70°C to 80°C. Measures for long-term prevention include the use of water management systems and decontamination using copper-silver ionization, hyperchlorination, and ultraviolet light.
Legionellosis is a nationally notifiable disease in the United States.
Mycoplasma and Ureaplasma
Mycoplasmas and ureaplasmas are small, fastidious organisms that lack a cell wall and cause an array of clinical symptoms in both immunocompetent and immunocompromised patients. , Species that cause human disease include Mycoplasma pneumoniae, M. hominis , M. genitalium , Ureaplasma urealyticum , and U. parvum . Disease processes caused by these pathogens are numerous and include lower respiratory tract infection, necrotizing pneumonia, meningoencephalitis, genitourinary infections, bone and joint infections, and intraabdominal infections as well as parainfectious and postinfectious syndromes, including erythema multiforme and Stevens-Johnson syndrome (SJS).
Epidemiology and risk factors
The epidemiology of Mycoplasma and Ureaplasma infections is highly species dependent. M. pneumoniae causes upper and lower respiratory tract infections in both endemic and epidemic patterns. Classically, M. pneumoniae causes a significant proportion of community-acquired pneumonia in otherwise healthy school-age and adolescent children. In a recent Centers for Disease Control and Prevention–sponsored multicenter pneumonia epidemiology study of pneumonia causing hospitalization in children younger than 18 years, Mycoplasma accounted for 8% of pneumonia overall and 19% in children 5 to 18 years. The epidemiology of M. pneumoniae is not well defined in pediatric SOT, HSCT, or oncology patients with only case reports and case series defining the occurrence of infection in these populations (see later text). In one prospective active surveillance study of upper or lower respiratory tract infection in children with cancer, M. pneumoniae respiratory infection occurred in 4 of 253 (1.6%) children over a one-year period.
Urogenital colonization by M. hominis and M. genitalium is well documented in sexually active adults. Despite this predilection, non-genitourinary infections are described, although they occur often in adolescents (see “Clinical Manifestations” later). Donor-derived acquisition of M. hominis is reported in an adult lung transplant recipient in whom bilateral pneumonia, pleural effusion, and multijoint arthritis developed early after transplant.
Ureasplasma species colonize the male and female genital tracts with infants becoming colonized during passage through the birth canal. Before the onset of sexual activity, detection of Ureaplasma species from the genital tract of prepubertal children is infrequent. Similar to M. hominis , donor-derived transmission of Ureaplasma is documented. Additionally, hypogammaglobulinemia may be a risk factor for severe or disseminated Ureaplasma infection.
Clinical manifestations
An array of clinical manifestations are associated with Mycoplasma and Ureaplasma spp. infections. It is important to note that disease manifestations are highly dependent on the species of infecting organism. Although literature reports of Mycoplasma and Ureaplasma infections in pediatric transplant or oncology patients are limited to case reports and case series, several patterns emerge from the literature, including occurrence of a greater variety of species causing disease, the occurrence of atypical locations and manifestations of infection, chronicity, and pyogenicity.
Pneumonia can occur as a consequence of infection with both Mycoplasma . and Ureaplasma spp. M. pneumoniae is a frequent cause of pneumonia in children; numerous studies have defined the typical age of occurrence ranging from young school-age (4 to 5 years) to the early teenage years (10 to 14 years). Of note, many of the early studies used culture and serologic assays, which likely underestimate the contribution of M. pneumoniae to pneumonia incidence compared with current molecular assays.
In immunocompetent children, respiratory Mycoplasma spp. infections are typically limited to M. pneumoniae and cause acute onset of pulmonary manifestations. A variety of radiographic patterns are reported in the literature, including scattered interstitial infiltrates, consolidative lobar pneumonia, pleural effusions, and occurrence of clinically severe and extensive pulmonary disease. In immunocompromised subjects, subacute to chronic presentations and purulent infections are also described. Chronic, recurrent focal consolidative pneumonia caused by to M. pneumoniae was diagnosed in a 4-year-old child who presented with persistent fevers and weight loss 3 years after kidney transplantation. Empyema caused by M. hominis was reported in an 18-year-old lung transplant recipient. In adult transplant patients, M. hominis pulmonary infection has been accompanied by surgical site infections. U. urealyticum was detected in the lower airways by culture in three pediatric cancer patients (2-year-old boy and 16-year-old girl each with HSCT, and a 16-year-old girl with rhabdomyosarcoma) with radiographic findings of lower respiratory tract infection; two of these three patients had diffuse bilateral pneumonitis. Thus lower respiratory tract infections caused by Mycoplasma and Ureaplasma spp. can present as either focal and purulent/pyogenic or diffuse, bilateral infection.
Nonrespiratory sites of infection are reported primarily as those caused by M. hominis and Ureaplasma spp ., with infection sites including urogenital, intraabdominal, bone/joint, and central nervous system. In a series of 10 adult kidney transplant patients with pyuria and detection of U. urealyticum and/or M. hominis , five patients were asymptomatic and three patients had pyelonephritis that responded to directed therapy. Intraabdominal abscesses caused by M. hominis and U. urealyticum have been reported in adult kidney transplant recipients. Septic arthritis of the hip caused by M. hominis was diagnosed in a 15-year-old woman in whom leukopenia and right hip weakness and pain developed 6 weeks after kidney transplantation. Septic polyarthritis caused by U. urealyticum was reported in an 18-year-old acute lymphocytic leukemia patient and a 28-year-old HSCT patient. , Meningitis caused by U. urealyticum has also been reported in SOT patients.
In addition to direct infections, parainfectious and postinfectious complications such as SJS and toxic epidermal necrolysis after Mycoplasma infection may occur. Attribution of causality of these events to Mycoplama is difficult given that immunocompromised pediatric patients are often receiving multiple medications that may cause SJS or toxic epidermal necrolysis.
The occurrence of severe and fatal hyperammonemia syndrome as the result of disseminated U. urealyticum infection has not been reported in children. However, potential exists for rare but severe occurrence in this population. Therefore understanding of the pathophysiology and features of this syndrome is critical for practitioners caring for immunocompromised pediatric patients. Primarily reported in lung transplant recipients, hyperammonemia syndrome occurs with a gradual elevation in serum ammonia concentration that ultimately results in severe neurologic dysfunction and progresses to cerebral edema and death. In 2015, the development of hyperammonemia syndrome in a cohort of adult lung transplant recipients was linked to disseminated Ureaplasma infection. The initial correlation with Ureaplasma infection was made in four patients who had died of neurologic sequelae of hyperammonemia syndrome. Identification of U. parvum and timely treatment in two subsequent patients resulted in resolution of hyperammonemia and excellent outcomes. Detection and treatment of U. parvum infection in a 21-year-old patient 2 weeks after matched unrelated HSCT led to resolution of hyperammonemia and associated neurologic abnormalities. Idiopathic hyperammonemia has been reported as a rare complication of HSCT and has not yet been associated with Ureaplasma infection. Practitioners caring for children undergoing transplantation should have a high index of suspicion for Ureaplasma infection in the setting of acute neurologic changes and/or altered mental status, especially when accompanied by hyperammonemia.
Disease prophylaxis/prevention
Prophylaxis against Mycoplasma and Ureaplasma spp. infections is not typically performed. Importantly, levofloxacin prophylaxis used in the setting of certain oncologic or HSCT therapeutic plans would theoretically provide prophylaxis against these infections (see “Treatment” section), although this has not been formally studied.
Diagnosis
Diagnosis of Mycoplasma and Ureaplasma spp. can be achieved by serologic, PCR, or culture-based methods. Serologic assays to detect both M. pneumoniae –specific IgG and IgM are readily available. As the currently available serologic assays have low specificity in otherwise healthy children, concern exists regarding both the sensitivity and specificity of these assays in immunocompromised children. Clinically available serologic assays for other Mycoplasma spp., such as M. hominis and M. genitalium and for Ureaplasma spp., are not available.
Detection of M. pneumoniae by PCR has been reported and is clinically available from a variety of specimens, including sputum throat, nasopharynx, cerebrospinal fluid, urine, synovial fluid, and tissue. Detection of M. pneumonia by PCR has greater sensitivity than serologic assays though may also detect asymptomatic colonization. More recently, M. pneumoniae detection has been included on several multiplex PCR platforms for evaluation of respiratory infections. PCR detection of other Mycoplasma and Ureaplasma spp. is also readily available through commercial laboratories.
Culture-based diagnosis of Mycoplasma and Ureaplasma spp. is challenging given the generally slow-growing and fastidious nature of these pathogens and may require up to 6 weeks of incubation to confirm negative results. Further, species-level identification requires either biochemical tests or PCR-based identification from subculture.
Treatment
Because all Mycoplasma s and Ureaplasma spp. lack a cell wall, these organisms are intrinsically resistant to β-lactams and glycopeptides. Treatment options for these infections include macrolides, such as azithromycin and clarithromycin, tetracyclines including doxycycline, and “respiratory” fluroquinolones such as levofloxacin. In the case of M. pneumoniae , macrolides and levofloxacin have the lowest MICs and are bactericidal. Importantly, macrolide resistance in M. pneumoniae develops after point mutations in the 23S rRNA target of these antibiotics. Because of their high mutation rate and small genomes, mutations can occur rapidly during therapy. The frequency of resistance varies geographically with resistance as high as 90% in areas of Japan and China, as low as 1% to 2% in some European countries, and approximately 10% to 13% in the United States. Although the clinical significance of macrolide resistance in M. pneumoniae is not well defined, macrolide resistance is of importance to immunocompromised pediatric patients given their increased reliance on antimicrobial activity in the clearance of infection. Resistance to tetracyclines and fluoroquinolones has not been detected in clinical isolates of M. pneumoniae .
Treatment options for other Mycoplasma and Ureaplasma spp. are highly dependent on the infecting species. M. hominis is always resistant to macrolides but is susceptible to clindamycin. Variable susceptibility to doxycycline and rare resistance to fluoroquinolones exist in M. hominis isolates. In contract, U. urealyticum remains susceptible to macrolides, tetracyclines, and fluoroquinolones but is not susceptible to clindamycin. M. genitalium is frequently resistant to macrolides and nonresponsive to tetracycline-based therapy, although fluroquinolones maintain susceptibility.
Infection prevention and anticipatory guidance
M. pneumoniae is transmitted from symptomatic persons via respiratory droplets, and transmission to household contacts as well as outbreaks has been reported. When hospitalized, patients with suspected respiratory illness caused M. pneumoniae should have droplet precautions. Immunocompromised children exposed to a household contact with atypical pneumonia or documented Mycoplasma infection should be counseled regarding the potential for transmission and advised to contact their primary and appropriate subspecialty providers if symptoms of fever or respiratory illness develop.
References
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree