Clostridioides difficile , formerly knowns as Clostridium difficile, is a Gram-positive, anaerobic bacillus, with a spore phase that prolongs survival in the environment. Ingestion of spores that are resistant to gastric acid results in maturation into the bacillus stage. C. difficile replicates disproportionally to other colonic flora when the balance is affected by another pathogen, absence of enteral feeds, exposure to antimicrobials, or chemotherapeutics. C. difficile produces two different toxins. Toxin A is an enterotoxin that attaches to the basal membrane damaging the villi. Toxin B is an extremely potent cytotoxin that induces apoptosis. Toxin-mediated damage, with progressive neutrophilic infiltration and fluid secretion by the intestine, results in many of the symptoms that are a sequelae C. difficile activity.
The presence of C. difficile in the gastrointestinal tract is the normal, baseline state. Newborns have sterile gastrointestinal tracts and C. difficile colonization is highest by 1 month of life, then declines in the following months until there are very low rates of colonization by 2 years of age. Most infants do not develop clinical disease, probably secondary to lack of toxin-binding receptors in their immature intestinal mucosa and/or by protection from secretory immunoglobulin A and oligosaccharides in human milk. Differentiating colonization from active infection in immunocompromised children is challenging, as dysbiosis is the common baseline state. Diarrhea, abdominal pain, and other nonspecific gastrointestinal symptoms are the result of numerous factors, including medications, chemotherapy, and frequent courses of antimicrobials.
Despite shared risk factors for C. difficile , there are differences in the epidemiology of C. difficile colonization and disease between children and adults that are essential to understand in order to interpret results and manage disease. Most of the limited evidence for the diagnosis and treatment of C. difficile is derived from studies in immunocompetent adults, with few studies in children. The focus of this chapter is to outline the differences between immunocompetent and immunocompromised C. difficile colonization and disease in children, focusing on pediatric solid organ (SOT), hematopoietic stem cell transplant (HSCT) recipients, and pediatric oncology patients.
Epidemiology and risk factors
Definitions of C. difficile disease and colonization
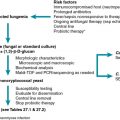
C. difficile disease is defined as new-onset of diarrhea (at least three unformed stools in less than a 24-hour period) and a positive diagnostic test result for C. difficile in stool, or colonoscopic or histopathologic evidence of pseudomembranous colitis. Because the probability of colonization is higher in younger children (usually those younger than 2 years), it is important to consider other common infectious and noninfectious etiologies of diarrhea even when C. difficile testing results are positive. There is no accepted definition for severe C. difficile disease. The presence of a complication of C. difficile disease or at least two abnormal laboratory findings ( Table 34.1 ) has been used in previous research studies, , but validation of this proposed scoring system is still required.
Clinical Criteria (At Least One)
Laboratory Criteria (2 or More)
|
Several studies noted an increase in the rates of C. difficile disease among children. , A retrospective study of more than 20 children’s hospitals in the United States reported that the rates of pediatric C. difficile disease doubled between 2001 and 2006. Another study analyzing secondary data from more than 30,000 pediatric patients noted an increase in the rates of C. difficile disease diagnosed by toxin assay between 1999 and 2006, and a decrease in rates of C. difficile disease between 2006 and 2010. Molecular diagnostics have altered surveillance. The increased sensitivity of molecular assays has limited the ability to make comparisons between rates of colonization and disease calculated using toxin assays with those calculated using molecular diagnostics.
A retrospective study of 200 children with C. difficile disease reported that 38 (19%) had underlying conditions that increased their risk of infection, and 149 (75%) had received antibiotics in the 2 months before the episode. Risk factors for the development of C. difficile disease in children include younger age (1 to 4 years), prolonged hospitalization, feeding via gastrostomy or jejunostomy, use of broad-spectrum antibiotics, HSCT, SOT, cancer, immunodeficiency, and human immunodeficiency virus (HIV) infection, fungal infections, viral gastroenteritis, cystic fibrosis, and inflammatory bowel disease. A nested case-control study in children identified the following risk factors for CDI: SOT, lack of hospitalization, presence of gastrostomy or jejunostomy, and receipt of fluoroquinolones or nonquinolone antibiotics in the 4 weeks before C. difficile disease.
C. difficile infection in pediatric SOT recipients
The estimated prevalence of C. difficile disease among pediatric SOT recipients is 12%, varying by transplanted organ. The rates of C. difficile disease among pediatric SOT recipients ranges between 5% and 16% for kidney recipients; between 11% and 12.9% for heart recipients; between 11% and 18% for liver recipients; between 11.5% and 9% for lung recipients ; between 3% and 7% for pancreas recipients , ; and 20% for small bowel transplant recipients. A study that evaluated secondary data from more than 50,000 hospitalized adult SOT recipients reported that those with C. difficile disease had higher mortality rates, longer hospital stays, higher costs, more colectomies, and higher rates of complications associated with the transplanted organ compared with those patients without C. difficile disease.
Among pediatric kidney transplant recipients, young age (younger than 5 years), female gender, treatment with monoclonal antibodies, antibiotic use, and intraabdominal placement of the graft were associated with the development of C. difficile disease. Other risks factors for the development of C. difficile disease described for adult SOT recipients include recent hospitalization, augmentation of steroid dose, use of steroids before transplant, ganciclovir prophylaxis, the use of antithymocyte globulin, and transplant other that kidney alone.
C. difficile infection in pediatric HSCT recipients
The rates of C. difficile disease are similar in pediatric and adult HSCT patients, although some studies have found even higher rates in children compared with adults. The rate of C. difficile disease among pediatric allogeneic HSCT recipients was 17% compared with 11% among adult patients in the same institution. Infection with C. difficile is more common in adult patients undergoing allogeneic (9% to 27%) than autologous (7% to 9%) transplants, respectively. Differing rates of posttransplant C. difficile disease has been noted in several observational studies. Recipients of allogenic HSCTs are subject to prolonged use of broad-spectrum antibiotics, a higher immunosuppressive state, and an increased risk for graft-versus-host disease (GVHD), all of which are risk factors for C. difficile disease. Another contributing factor may be higher colonization rates with C. difficile before allogenic transplantation, a factor that has been reported among adult and pediatric allogenic HSCT recipients.
A nested case-control study with a multivariate analysis comparing 62 adult allogeneic HSCT recipients with 123 controls matched by graft type demonstrated that receipt of chemotherapy before conditioning, exposure to broad-spectrum antibiotics after transplant, and vancomycin-resistant enterococci colonization were associated with the development of C. difficile disease. Cord blood as the source of the stem cells, acute GVHD, and total body irradiation were associated with C. difficile disease among adult allogenic HSCT recipients.
C. difficile disease in pediatric oncologic patients
One-third of children younger than 3 years and one-fifth who are 3 years and older are colonized with C. difficile at the time of their first admission to the pediatric oncology ward. After 2 weeks of inpatient hospitalization, colonization rates increase to 90% for children younger than 3 years and 50% for those older than 3 years. Another surveillance study that used polymerase chain reaction (PCR) testing and culture, reported that 29% of asymptomatic pediatric oncology patients were colonized with C. difficile at the time of admission to an inpatient unit. Slightly more than half (55%) of those who had a history of C. difficile disease had positive results for stool sampling by molecular testing or culture noted intermittently for over 20 weeks, sometimes with different C. difficile strains. The high prevalence of colonization, gut dysbiosis, gastrointestinal effects of chemotherapy regimens, antibiotic exposure, other infections, and underlying conditions make the diagnosis of C. difficile disease challenging in pediatric oncologic patients.
Cancer has been reported as the most common comorbidity in pediatric C. difficile disease, with 25% of infections reported among children whose data was collected in administrative databases. , According to these databases, the rate of C. difficile disease is 10 times higher in pediatric patients with cancer compared with children without cancer. A multicenter retrospective cohort study evaluating children with acute myeloid leukemia reported that 37 (11%) developed diarrhea and had positive test results for C. difficile toxin while receiving chemotherapy.
A study that evaluated the risk factors associated with C. difficile disease among children with cancer reported that exposure to aminoglycosides, third- and fourth-generation cephalosporins, and proton pump inhibitors in the week before admission, and chemotherapy in the 8 to 14 days before admission were associated with the development of C. difficile disease. Another retrospective cohort study in children with acute myeloid leukemia reported that the duration of broad-spectrum antibiotics and infection of a sterile site were independently associated with C. difficile disease.
Clinical manifestations
Toxin detection and clinicopathologic correlation
Diarrhea or other gastrointestinal symptoms frequently develop in pediatric immunocompromised patients as the result of infections, as an adverse effect of antibiotics or chemotherapeutic agents, or as the result of an underlying disease. The detection of C. difficile toxin in stool samples does not necessarily correlate with organism activity. One of 11 children from whom C. difficile toxin was detected in their stools had postmortem histologic evidence of clostridial infection and abundant pseudomembranes. The other 10 children had negative clostridial immunohistochemistry and PCR results, and the macroscopic findings varied significantly, from normal to the presence of pseudomembranes.
C. difficile disease
C. difficile disease may be considered in children with new-onset diarrhea, with three or more episodes of unformed stools in less than 24 hours. The median time for development of C. difficile disease after SOT was 57 days (interquartile range [IQR] 14 to 227 days) varying by transplanted organ. In pediatric kidney transplant recipients C. difficile disease developed significantly earlier than in adults (mean time of onset of symptoms was 33 days for children, compared with 15 months in adults). C. difficile disease has been reported in four children who underwent a lung transplant for cystic fibrosis; two had disease within the first 4 months after transplant, whereas in the other two infection developed during the first 3 years of follow-up. The median time for detection of C. difficile disease was 204 days after the procedure (range 77 to 339 days) in small bowel transplant recipients.
Reports of the time of onset of gastrointestinal symptoms in patients with C. difficile disease after HSCT vary from center to center, likely related to differences in colonization rates, criteria for diagnostic testing, and differences in patient populations. The highest risk for C. difficile disease among HSCT recipients spans the duration of conditioning through 3 months post-transplant. C. difficile disease tends to develop earlier in recipients of autologous HSCT than in allogeneic HSCT recipients, with a median time of onset of 6.5 versus 33 days, respectively. When compared with adult allogeneic HSCT recipients, C. difficile disease developed in pediatric allogeneic HSCT recipients later, at a median of 51 days (IQR 5 to 72 days) compared with 16 days (IQR 5 to 49 days).
Severe C. difficile disease
The definition of severe C. difficile disease in immunocompromised hosts is challenging. Severe C. difficile disease with leukocytosis or fulminant colitis with pseudomembranes has been reported in pediatric SOT recipients. Nevertheless, most patients are neutropenic during and after chemotherapy or cytoreduction. Because the development of pseudomembranous colitis requires neutrophilic infiltration of the intestinal mucosa, this severity marker is not a common occurrence in patients with neutropenia.
Pseudomembranous colitis has been reported after both autologous and allogeneic bone marrow transplants, but patients were not neutropenic at the time of diagnosis of C. difficile disease. Another case series reported four allogeneic HSCT patients with C. difficile disease in whom nonspecific inflammatory changes developed within their colonic mucosa, without evidence of pseudomembranous colitis.
There are two sets of proposed criteria for the definition of severe C. difficile disease. The Society for Healthcare Epidemiology/Infectious Diseases Society of America (SHEA/IDSA) guidelines include the presence of white blood cell count higher that 15,000 cells/mL or a serum creatinine level greater than 1.5 mg/dL. The European Society of Clinical Microbiology and Infectious Diseases (ESCMID) guidelines require the presence of any of the following: fever; rigors; hemodynamic instability; signs or peritonitis or ileus; marked leukocytosis or marked left shift; elevated creatinine; reduced serum albumin; elevated lactate level; evidence of pseudomembranous colitis on endoscopy; distention of large bowel, colonic wall thickening, pericolonic fat stranding, or ascites on imaging. Given the differences between immunocompromised children and adults, it is reasonable to assume that classic clinical, pathologic, or diagnostic criteria for severe C. difficile disease may not be adequate for pediatric oncology patients and transplant recipients. When these criteria were applied to immunocompetent pediatric patients, half of the children with severe infections were not detected using the SHEA/IDSA criteria, whereas all children with severe infections were detected by the ESCMID criteria. Previous studies, one in adults and one in children, have used the presence of a complication of C. difficile disease or at least two abnormal laboratory findings (see Table 34.1 ) as indicators of severe C. difficile disease, , but this scoring system has not been validated.
Clinical outcomes
A large cohort study from Canada reported that 90% of children with C. difficile disease had resolution of symptoms by 30 days after onset, and 2% experienced severe outcomes (five required intensive care and one patient’s C. difficile disease was associated with mortality).
A large retrospective cohort study using secondary data from 41 free-standing children’s hospitals in the United States reported that children with a C. difficile diagnosis had a significantly higher mortality than those without a C. difficile diagnosis (1.4% vs. 0.7%). When subgroups were analyzed based on time of onset of disease, those who had C. difficile detected before the third day of hospitalization had mortality similar to C. difficile– negative children. Children with C. difficile detected after the third inpatient day had higher mortality rates than C. difficile– negative children, longer hospital stays, and higher costs. Other risk factors associated with higher mortality rates included older age (>13 years), underlying malignancy, cardiovascular disease, hematologic or immunologic conditions, gastric acid suppression, and the presence of more than one severity illness marker in the previous 48 hours (admission to intensive care unit, receipt of vasopressors, or need for respiratory support). A study of 200 children in Canada reported a C. difficile– associated mortality rate of 1%. The cohort included 38 children with underlying conditions that increased their risk of C. difficile disease. Both children who died were immunocompromised. A small cohort of pediatric lung transplant recipients in whom C. difficile disease developed had poor outcomes. Of four lung transplant recipients with C. difficile disease, one required an ileostomy, another died after developing renal insufficiency, and two had subsequent episodes of C. difficile disease that eventually resolved. Although it is challenging to separate C. difficile infection from disease and even more difficult to separate comorbidities of immunocompromised children from the clinical presentation of C. difficile disease, clinicians should be aware of the poorer clinical outcomes of children with C. difficile infection and disease that have been demonstrated both on the individual and population levels.
Recurrence of C. difficile disease
The rate of recurrence of C. difficile disease in children has been reported as between 20% and 30%. , In comparison, 10 (40%) of pediatric SOT recipients had a recurrent episode of C. difficile . Two (20%) of the SOT recipients had a recurrent C. difficile in the 8 weeks after the initial episode. Factors associated with recurrence included malignancy, recent surgery, and the number of antibiotic classes to which the patient was exposed. The mean time to recurrence was 34 days after the onset of the first episode. A case-control study comparing 48 children with severe C. difficile disease with 34 children with nonsevere C. difficile disease found no difference between these groups in terms of recurrence or treatment failure.
Children who received an HSCT had higher rates of recurrence C. difficile disease compared with those who did not receive a transplant, but the difference in rates was not statistically significant. , Patients in whom gastrointestinal GVHD (GI GVHD) developed had a 5-fold higher odds of recurrent C. difficile disease. GI GVHD remained significant after adjusting for recognized risk factors for C. difficile recurrence, including receipt of systemic steroids. In a case-control study, adults in whom difficile disease developed were more likely to have GI GVHD disease in the year after transplantation. In a subgroup of 12 (85%) patients, the diagnosis of C. difficile disease preceded the diagnosis of GI GVHD. The median time to development of GI GVHD was 21 days after the diagnosis of C. difficile disease. A retrospective cohort of adult and pediatric T-cell–depleted allogeneic HSCT recipients did not find an association between the development of GI GVHD and C. difficile disease.
Diagnosis
The diagnosis of C. difficile disease may be considered in high-risk patients with new onset of diarrhea. Current guidelines recommend consideration of C. difficile testing when a patient has more than three episodes of unformed stools in less than 24 hours. Clinicians should consider how they will interpret the results of diagnostic testing in the context of risk factors of the host. In immunocompromised pediatric hosts, the clinician should consider the likelihood of infection versus colonization, underlying conditions, recent antimicrobial, administration, radiation, and chemotherapy, as well as non– C. difficile etiologies for the symptoms.
Toxigenic culture and cell cytotoxicity neutralization assays in stools have been largely replaced by the detection of glutamate dehydrogenase (GDH, an enzyme present in all isolates of C. difficile ), and C. difficile toxins using enzyme immunoassays; and by nucleic acid amplification tests (NAAT) that can target C. difficile –specific genes, including tcdA, tcdB , and 16S ribosomal RNA.
Selection of a diagnostic assay should consider the sensitivity, specificity, and positive and negative predictive values. of the tests used. Enzyme immunoassay has a sensitivity of 35% compared with 95% with a PCR when pediatric samples are analyzed. Both tests have a specificity of 100%. PCR increases the yield of detection of C. difficile by toxin 2-fold compared with cytotoxin assays.
Diagnostic algorithms that include a multistep process (i.e., GDH plus toxin; GDH plus toxin, with NAAT; or NAAT plus toxin) to adjudicate inconclusive results have the optimal positive predictive value. The use of NAAT alone has a high sensitivity, which may lead to overdiagnosis of C. difficile . Currently NAAT alone or a multistep process is recommended as the preferred diagnostic method.
Clinicians caring for both immunocompetent and immunocompromised infants must carefully interpret of C. difficile diagnostic results, given the high rates of colonization and clinical insensitivity to C. difficile toxin in this age group. Although rare, C. difficile disease has been reported and the diagnosis should be considered, especially when an infant has manifestations of pseudomembranous colitis, toxic megacolon, or significant diarrhea when other etiologies have been excluded. Children between 1 and 2 years of age are also likely to have high C. difficile colonization rates, making diagnosis of active disease in a symptomatic host with clinical symptoms challenging. Children older than 2 years have similar colonization rates as older children and adults.
Increased reporting of severe gastrointestinal infections after transplantation, including C. difficile infection, have been affected by the use of molecular panels with increased sensitivity and multiple targets. The detection of co-infections with C. difficile , including rotavirus, norovirus, and sapovirus, has been noted. Comparisons of different commercial multiplex PCR platforms for enteropathogens have yielded equivalent results from stools of immunocompromised children.
Diagnostic tests of cure are not beneficial, as toxin may continue to be detected in up to 60% of asymptomatic children. Guidelines emphasize that repetitive diagnostics within 7 days of the initial test are not beneficial. Children with recurrence of symptoms after treatment and initial resolution may be evaluated with repeat diagnostics considering non– C. difficile etiologies.
The evidence to support the use of stool inflammatory markers to diagnose C. difficile disease is limited. A study evaluating C. difficile –positive and C. difficile –negative diarrheal stool samples found higher stool levels of lactoferrin, calprotectin, interleukin (IL)-8 and IL-23 in C. difficile –positive stool samples Other markers present included C5a, CD40L, granulocyte colony-stimulating factor, I-309, IL-13, IL-16, IL-27, monocyte chemoattractant protein-1, tumor necrosis factor alpha, and IL-8. The diagnostic utility of stool inflammatory markers in the management of children with diarrhea is undergoing assessment.
Treatment
The mainstay of treatment of C. difficile disease is to restore normal flora and reduce dysbiosis. Optimally this is accomplished by discontinuing antimicrobials, if this is feasible. The 2017 IDSA/SHEA C. difficile guidelines recommend that either metronidazole or vancomycin may be considered for the treatment of a nonsevere, first episode of C. difficile disease in children. An Emerging Infectious Disease Network survey of 285 pediatric infectious disease specialists noted that all respondents use oral metronidazole for a mild, first episode of C. difficile disease. In this survey, 41% to 79% of physicians would still use metronidazole for a mild, first episode of C. difficile disease when the patient had an underlying co-morbidity, including SOT.
In children with an initial, severe C. difficile disease episode, the treatment of choice is oral vancomycin. Either vancomycin or fidaxomicin is recommended for the treatment of an initial, severe episode of C. difficile disease in adults. These antibiotics have similar rates of cure, including resolution of symptoms, although adult fidaxomicin recipients had lower recurrence rates (71% vs. 57%) compared with oral vancomycin recipients. Fidaxomicin has been administered to adult oncology patients who have recurrent C. difficile disease, with resolution of clinical symptoms in 20 of 22 patients. Fidaxomicin has not been approved by the FDA for use in children younger than 18 years, although it has been tolerated in children with a pharmacokinetic profile similar to adults. A 10-year old boy, who was gastrostomy tube dependent, had five episodes of C. difficile disease after being treated for recurrent pneumonia. He received crushed fidaxomicin at a dose of 200 mg twice daily administered via his gastrostomy tube. His diarrhea resolved in less than 24 hours. C. difficile disease recurred after a subsequent course of antibiotics, and fidaxomicin receipt was accompanied by quick symptom resolution.
Oral vancomycin is the antimicrobial of choice for children with two or more episodes of recurrent C. difficile disease. If the antimicrobial used previously was vancomycin, an extended course of oral vancomycin (tapered and pulsed regimen) or oral vancomycin followed by rifaximin or fidaxomicin could be considered for treatment, extrapolated from adult studies. A survey of pediatric infectious diseases specialists reported that 23 (18%) would recommend fecal microbiota transplantation (FMT) and 20 (16%) would recommend off-label use of fidaxomicin for recurrent or severe C. difficile disease in children.
A review article summarized the experience of FMT in children with refractory C. difficile disease; 23 patients aged 16 months to 19 years (8 were immunocompromised) had cure rates between 50% and 100%. In a case series of 10 children with recurrent C. difficile disease, four immunocompromised (three with inflammatory bowel disease, one received chemotherapy for Wilms tumor), concluded that FMT administered via nasogastric tube was safe and effective. Nine (90%) had symptom resolution. One child had recurrence of symptoms 2 months after FMT while he was receiving chemotherapy, and C. difficile recurred after a second FMT ( Table 34.2 ).