78.1
Introduction
Testosterone is the principal circulating androgen in men, secreted almost entirely by the testes. The effects of testosterone on bone in men are therefore best observed when they are deficient in testosterone and then replaced with testosterone. The effects of physiologic concentrations of testosterone in men, as observed in these situations, are substantial.
Testosterone is also the principal circulating androgen, in potency, in women. It is secreted by the ovaries and the adrenal glands and derived also by peripheral conversion of the weak adrenal androgens, androstenedione, and dehydroepiandrosterone (DHEA). The normal serum concentration of testosterone in women, however, is only approximately 10% of that in men, and the effect of testosterone on bone in women is therefore much less clear than in men.
78.2
Endogenous androgens
Testosterone is the principal secreted and circulating androgen in both men and women. In men the serum concentration is 300–800 ng/dL, and in women, the serum concentration is 30–50 ng/dL. In both men and women the adrenal glands secrete DHEA, which is significant as an androgen only as a precursor of testosterone, and androstenedione, which is a weak androgen.
78.2.1
Androgens available for treatment
When testosterone is ingested, it is absorbed well, but because it is absorbed into the hepatic circulation, the liver catabolizes it so rapidly that testosterone cannot be administered in sufficient quantity orally to maintain a normal serum testosterone concentration in a hypogonadal man. The development of testosterone preparations for therapeutic use during the last half-century, therefore, has involved attempts to bypass hepatic catabolism.
Many early attempts to bypass hepatic catabolism involved the development of various 17-alkylated androgens, which were claimed to have greater anabolic than androgenic properties. Many of these, such as methyltestosterone, fluoxymesterone, etc., are still commercially available, and they are orally active, but it is not certain they have greater anabolic than androgenic properties, and they may cause cholestatic jaundice occasionally and peliosis hepatica rarely . These are the androgens often abused by athletes.
Other early attempts to bypass hepatic catabolism involved esterification of the 17β hydroxyl group of testosterone with a long-chain fatty acid to produce an ester that is even more lipophilic than testosterone itself and, when injected intramuscularly an oily solution, is hydrolyzed and released relatively slowly. These esters, testosterone enanthate and testosterone cypionate, are also still available for clinical use. When given in doses of 50–100 mg every week or 100–200 mg every 2 weeks, they result in serum testosterone concentrations that, on average, are normal but fluctuate widely from supranormal in the first day or two after an injection to low-normal to low just prior to the next . They result in normal virilization, but some men recognize the fluctuating serum testosterone concentrations by fluctuations in energy, mood, and libido. Erythrocytosis is common with these preparations. Testosterone enanthate is also now available as a subcutaneous injection .
Testosterone undecanoate is a testosterone ester that is even longer acting when given intramuscularly . When 750–1000 mg is administered every 10–14 weeks, serum levels peak at 1 week and decline thereafter. It is also available outside the United States as an oral preparation.
Currently, transdermal testosterone preparations are those most commonly used to replace testosterone in hypogonadal men. Two patches and several gels and a solution are now marketed. All contain excipients, compounds that facilitate the absorption of testosterone across the skin.
The patch causes severe skin rash in about one-third of users, but the gels rarely do. All are applied once a day and, after adjustment of the dose, result in relatively steady serum testosterone concentrations within the normal range in most hypogonadal men.
No testosterone-only preparation is currently approved by regulatory agencies for use in women.
78.3
Effects of testosterone on bone in men
The effects of testosterone on bone in men can be observed clearly in two situations: the deterioration of bone when men are hypogonadal (i.e., deficient in testosterone) and the improvement of bone when hypogonadal men are replaced with testosterone. In a third situation the deterioration of bone as men age, the effects of testosterone are now established but less pronounced.
78.3.1
Effect of hypogonadism on bone in men
78.3.1.1
Cross-sectional studies in hypogonadal men
Most studies of bone in hypogonadal men are cross-sectional, comparing bone in hypogonadal men to that in eugonadal men, since observing men longitudinally as they become hypogonadal is usually inappropriate. An early cross-sectional study examined 23 men with idiopathic hypogonadotropic hypogonadism, all severely hypogonadal . Bone density of the distal radius by single-photon absorptiometry and spine by computerized tomography (CT) was both more than two standard deviations (SD) below the means for age.
Another cross-sectional study examined 18 men who had acquired, yet unequivocal, hypogonadism due to hyperprolactinemia . Bone density of the wrist by single-photon absorptiometry and the spine by CT was both significantly less than those of age-matched eugonadal men. Together, these studies suggest that men who are hypogonadal have less bone, both in regions that are richer in cortical bone, such as the radius, and in trabecular bone, such as the spine.
78.3.1.2
Longitudinal studies in hypogonadal men
Longitudinal studies of the effect of hypogonadism on bone in men have been performed principally in men who have been made hypogonadal by the administration of a gonadotropin-releasing hormone (GnRH) analog as treatment for metastatic prostate cancer. The effects of the hypogonadism on their bone mineral density (BMD), bone strength, bone turnover, and fracture incidence have been studied.
78.3.1.2.1
Areal bone mineral density
In a study of 15 men, mean age 75 years, with prostate cancer, no bone metastases had areal BMD (aBMD) determinations before and after 1 year of treatment with a long-acting superactive GnRH analog that reduced their serum testosterone concentration from a mean of 467 ng/dL to 28 ng/mL . By comparison, 13 men, mean age 70 years, who did not have prostate cancer also had aBMD determinations at baseline and 1 year later. aBMD in the total hip decreased by 3.3% and in the ultradistal radius by 5.3% after 1 year, changes that were statistically significant compared to the changes in the eugonadal men, but the decreases in the spine (2.8%) and femoral neck (2.7%) were not statistically significant compared to eugonadal men. Urine N -telopeptide increased during treatment in the GnRH agonist-treated patients but not in the eugonadal men.
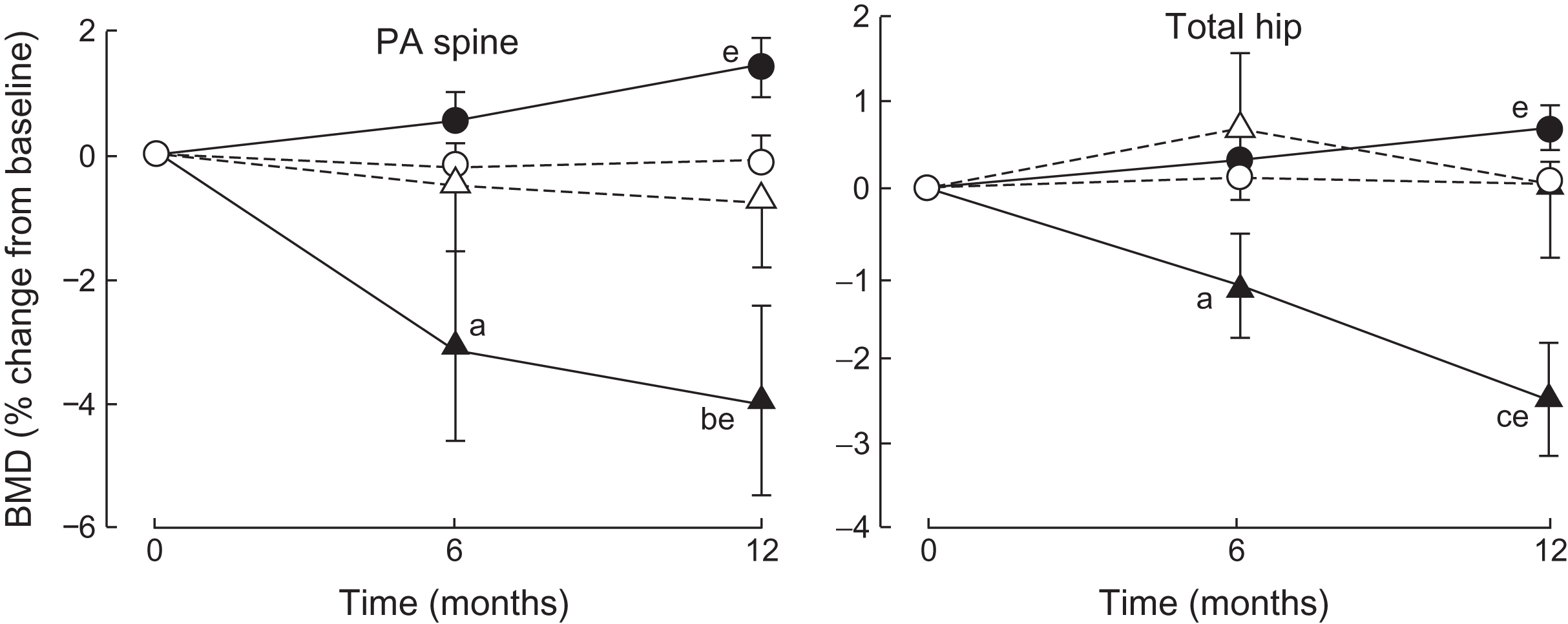
In a study, 152 men with prostate cancer—30 who had been kept hypogonadal for less than 6 months, 50 who had been kept hypogonadal for 6 months or longer, and 72 who had prostate cancer but were eugonadal, as well as men who did not have prostate cancer—had aBMD determinations on two occasions 1 year apart . During the 1-year period, the men who had been kept hypogonadal for less than 6 months experienced a 4.0% decrease in BMD of the spine and 2.5% of the total hip, but the men who had been hypogonadal for 6 months or longer and the eugonadal men did not experience a decrease ( Fig. 78.1 ). Urine N -telopeptide and bone-specific alkaline phosphatase were higher in the men receiving GnRH analog less than 6 months than in the other men. These results suggest that the decrease in BMD in response to testosterone deficiency occurs mainly in the first 6 months of deficiency.
In a study of men with prostate cancer who were treated with a GnRH agonist and followed for 6 years, men who had normal BMD ( n =124) or osteopenia ( n =242) at the outset experienced decreases in nondominant ultradistal forearm BMD of 12.7% and 14.1%, respectively .
78.3.1.2.2
Volumetric bone mineral density and microarchitecture
The effects of hypogonadism on volumetric BMD (vBMD) and bone microarchitecture were evaluated prospectively by high-resolution peripheral quantitative computed tomography (HR-pQCT) in 26 men with prostate cancer before and after 12 months of treatment with a GnRH agonist . After treatment, total vBMD at the distal radius (−5.2±5.4%, P <.001) and distal tibia (−4.2±2.7%, P <.001) both decreased; the decreases in cortical vBMD (distal radius −11.3±8.6%, P <.001; distal tibia −6.0±4.2%, P <.001) were greater than those in trabecular vBMD (distal radius −3.5±6.0%, P <.01; distal tibia −1.5±2.3%, P <.01). Hypogonadism was associated with significant decreases in trabecular number in both the distal radius ( P =.014) and distal tibia ( P =.045) but not with trabecular thickness.
78.3.1.2.3
Risk of fracture
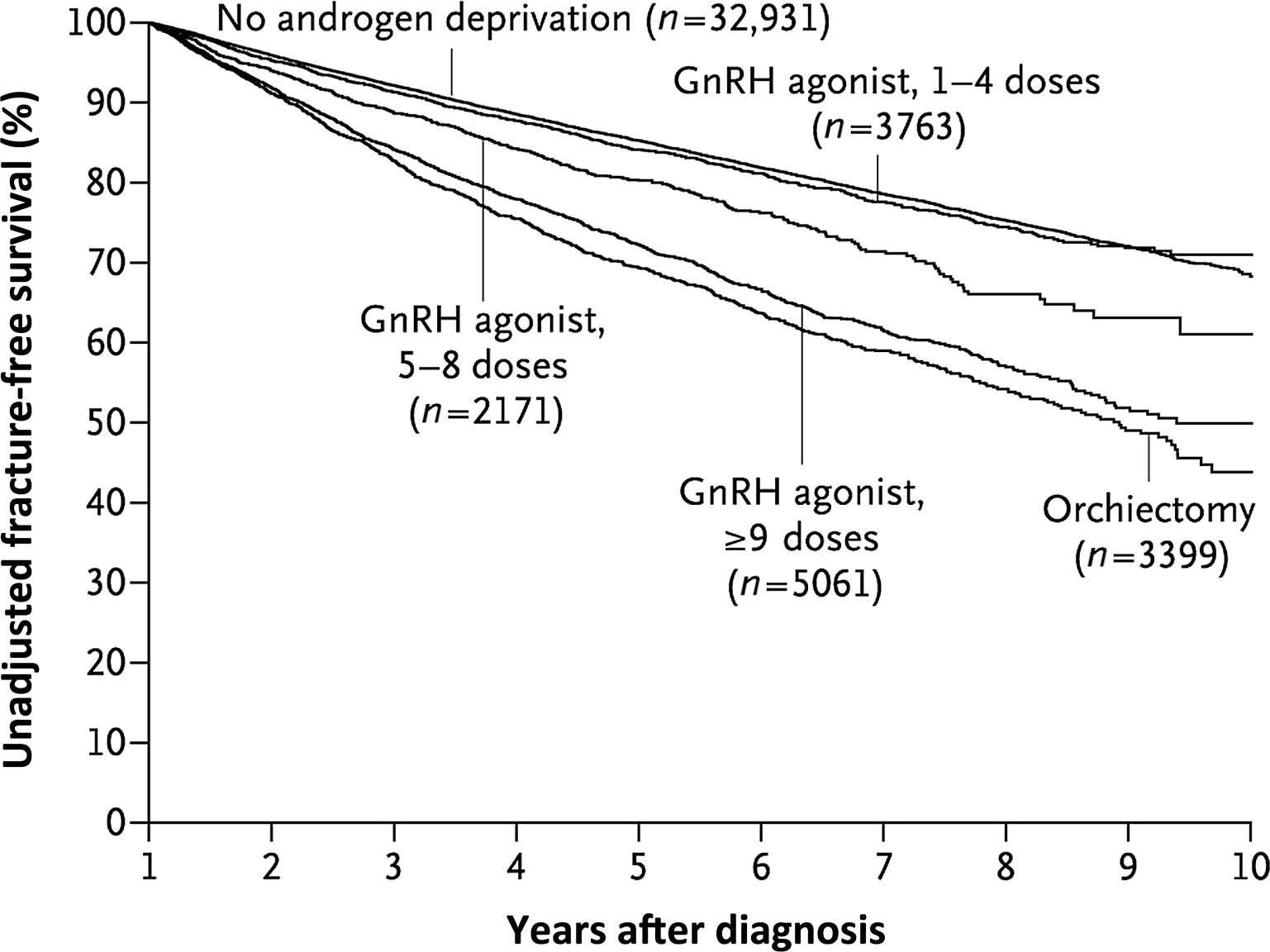
The risk of fracture in men with prostate cancer who have been made hypogonadal has been evaluated by cohort studies based on medical claims data. In a study using data from the Surveillance, Epidemiology, and End Results program and the Medicare database, the prevalence of fractures was assessed in 50,613 men aging over 66 years with prostate cancer who had been made hypogonadal by a GnRH agonist or bilateral orchiectomy. Men who had been made hypogonadal had a significantly higher prevalence of fracture (19.4%) than men not made hypogonadal (12.6%, P <.001). There was a significant relation between the number of doses of GnRH agonists and the risk of fracture ( Fig. 78.2 ) .
In another cohort study, a 5% random sample of Medicare beneficiaries comprising 3887 men with prostate cancer treated with a GnRH agonist was compared to 7774 men with prostate cancer who were not treated with the agonist . The men treated with the agonist had an increased risk of any clinical (vertebral and nonvertebral) fracture [relative risk (RR) 1.21, 95% confidence interval (CI) 1.14–1.29; P <.001]. The longer the duration of treatment, the greater the risk will be. Similar findings were reported in a third cohort study using a medical claims database from 16 large American companies . The rate of fracture was 7.91% in men who were treated for prostate cancer with a GnRH agonist compared to 6.55% in men who were not treated (RR 1.21, 95% CI 1.09–1.34; P <.001). The RR of hip fracture was greater than that of vertebral fracture.
These studies demonstrate that the development of severe hypogonadism leads to decreased bone density, increased bone turnover, deterioration of bone architecture, and increased risk of fracture.
78.3.1.3
Cross-sectional and longitudinal studies in elderly men
Serum testosterone concentrations in men decrease with age, as does BMD, and the incidence of bone fractures increases, leading to the hypothesis that the decrease in testosterone contributes to the decreased bone density and increased risk of fracture. To evaluate this possibility, several groups of investigators have performed epidemiological studies in elderly men to examine the association between testosterone concentrations and bone density and fractures.
The association of BMD with testosterone and other sex steroids was determined in elderly men in the US and Swedish Osteoporotic Fractures in Men Study (MrOS) studies. In the US study in 1238 men over 65 years, aBMD of the hip by dual-energy X-ray absorptiometry (DXA) was not correlated with total or bioavailable testosterone but was correlated with bioavailable estradiol . Similarly, hip aBMD decreased faster in men with the lowest bioavailable estradiol. In the Swedish study of almost 3000 men of average age 75 years, hip aBMD was correlated with both free testosterone and free estradiol . In the US study, trabecular vBMD of the spine was correlated with both bioavailable testosterone and bioavailable estradiol .
Bone fractures have also been found to be associated with testosterone and/or estradiol, usually free or bioavailable, in elderly men. In 793 men in the Framingham study followed for over 15 years, low estradiol, but not low testosterone, was associated with an increased incidence of hip fracture . In more than 2500 men in the Swedish MrOS study, free testosterone concentration below the median was associated with prevalent osteoporotic and vertebral fractures [odds ratio (OR) 1.56, 95% CI 1.14–2.14] , but only free estradiol was associated with incident fractures . In 623 men in the Longitudinal Ageing Study Amsterdam, bioavailable estradiol values below the median were associated with increased fracture risk . In 609 men over 60 years in the Dubbo study, low total testosterone was associated with an increased risk of overall incident osteoporotic fracture [hazard ratio 1.33, 95% CI 1.09–1.62] .
In a longitudinal study of 5487 men participating in MrOS study in the United States, Sweden, and Hong Kong for approximately 10 years, baseline testosterone, estradiol, and sex hormone–binding globin did not improve the fracture risk discrimination model . In another MrOS study of 1463 men followed for 4.6 years, higher baseline sex hormone binding globulin (SHBG), but not testosterone or estradiol, was associated with greater likelihood of prevalent vertebral fractures [OR 1.38 (CI 1.11–1.72) per SD increase in SHBG] and incident vertebral fractures (OR 1.42 (CI 1.03–1.95) per SD increase in SHBG) . Another case-cohort study from the MrOS that included 1978 men, 342 of which had an incident fracture, demonstrated that men in the lowest quartile of baseline bioavailable estradiol (11.4 pg/mL) and highest quartile of SHBG (>59.1 nM) had greater risk of nonvertebral fracture than those in the other three quartiles combined . Higher baseline testosterone levels in men in the MrOS study were associated with less of a decline in lean body mass , which might decrease fracture risk.
Although testosterone or estradiol, more often free or bioavailable, is associated with an increased risk of osteoporotic fracture, the lack of consistency as to which steroid is independently related suggests that the association is not strong. The relatively weak association is also illustrated by the observation that low testosterone in the Dubbo study increased incident osteoporotic fracture risk by only 33%, but age and history of prior fracture increased the risk by 76% and 83%, respectively . The relatively weak association may be related to the moderate degree of the decrease in serum testosterone with increasing age.
Observational studies, in short, show that the relatively small decrease in testosterone with aging in men results in a relatively small decrease in bone density and relatively small increase fracture risk.
78.3.2
Effect of replacing testosterone in hypogonadal men
78.3.2.1
Effect on bone mineral density
Several studies have shown that when men who are unequivocally hypogonadal due to pituitary or testicular disease are replaced with testosterone, their BMD increases substantially. In a study of 29 men with acquired secondary or primary hypogonadism who had not previously been treated and were then treated with testosterone enanthate, 100 mg once a week for 18 months, spine aBMD by DXA increased 5% and vBMD by quantitative CT 14% .
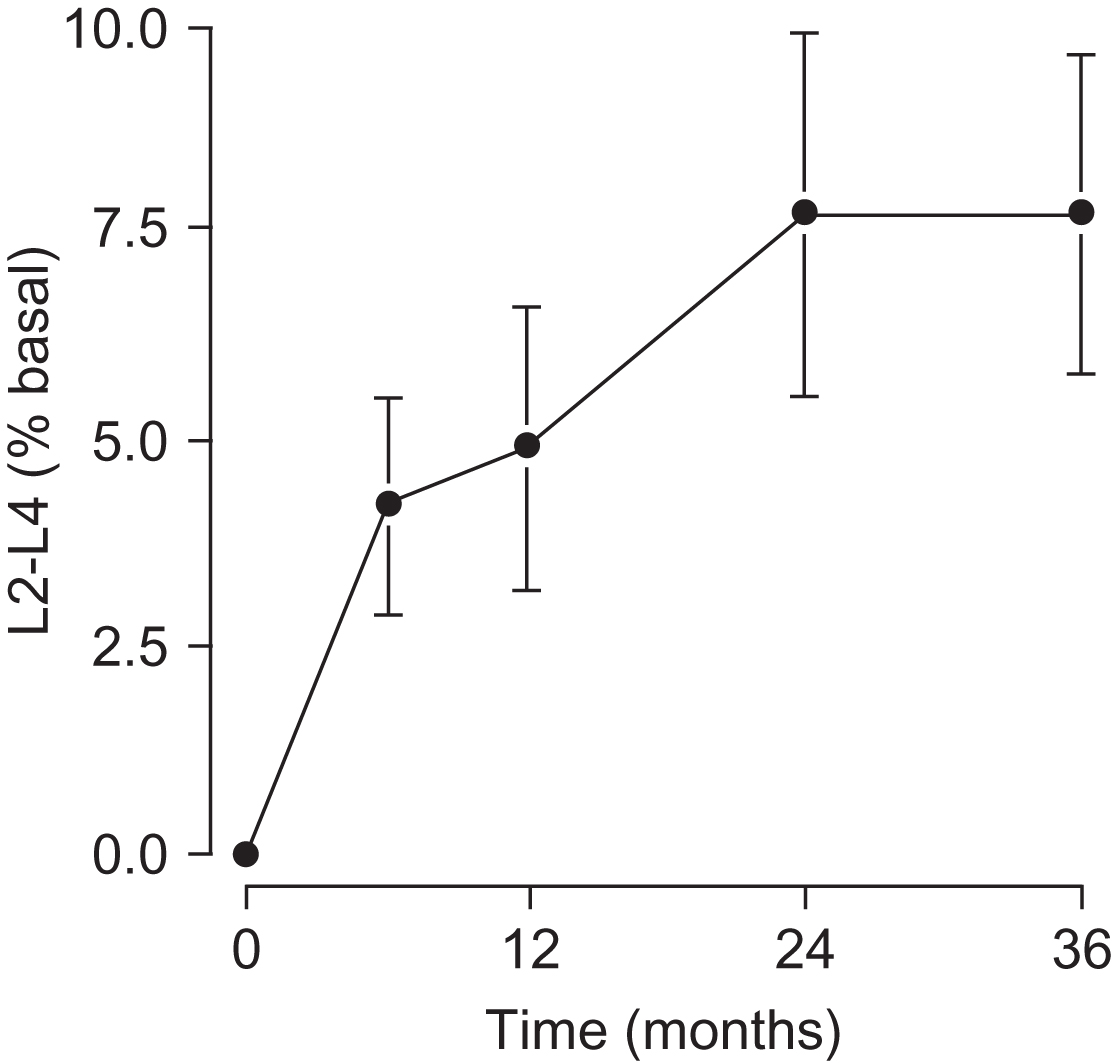
In another study, of 16 men with secondary or primary hypogonadism who had not previously been treated and were then treated with testosterone transdermally for 36 months, aBMD by DXA of the lumbar spine increased 7.7% ( Fig. 78.3 ) and of femoral trochanter 4.0% . Most of this increase occurred by 24 months.
In a third study, of 72 men with secondary and primary hypogonadism, 32 were studied before initiation of testosterone treatment, vBMD of the lumbar spine by quantitative CT scan increased 26% in the first year of treatment and another 5% in the next 2.7 years of treatment . After a mean of 3.2 years of testosterone replacement, trabecular vBMD of the lumbar spine increased 30% ( P <.001) and cortical vBMD 9% ( P <.001) .
The substantial magnitude of the increases in BMD following initiation of testosterone in previously untreated hypogonadal men in these three studies demonstrates that testosterone can restore bone, in some way, that has deteriorated as a consequence of long-standing testosterone deficiency. None of these studies employed a placebo-treated group because the men were too hypogonadal to justify withholding testosterone treatment, but the magnitude of the improvement in BMD cannot readily be explained except by an effect of testosterone.
78.3.2.2
Effect on bone turnover
Loss of testosterone appears to increase bone turnover, and replacement appears to decrease it, as judged by the measurement of biochemical markers of bone formation and resorption. When 27 consecutive men with prostate cancer but no documented bone metastases were treated with a GnRH antagonist to severely lower their testosterone levels for 2 years, a serum marker of bone formation—C-terminal propeptide of human type I procollagen (PICP)—and a serum marker of bone resorption—pyridinoline cross-linked C-terminal telopeptide of type I collagen (ICTP)—both increased significantly after 1 year of treatment but fell and were no longer significantly different from pretreatment by 2 years .
In three studies in which testosterone was replaced in untreated hypogonadal men, testosterone treatment usually reduced markers of bone formation and resorption. In the study of 29 men who were treated with 100 mg of testosterone enanthate for 18 months, serum bone–specific alkaline phosphatase and urine N -telopeptide were both significantly lower at 18 months than prior to treatment . In the study of 18 men treated with testosterone transdermally for 36 months, bone-specific alkaline phosphatase and urine N -telopeptide were both lower at the end of treatment than before . In a study of 10 men treated for 24 months, serum PINP (N-terminal propeptide of human type I procollagen) increased from 0 to 3 months but decreased from 3 to 6 months, and bone-specific alkaline phosphatase and urine N -telopeptide did not change .
The mechanism by which testosterone reduces bone turnover was explored in a group of 59 normal elderly men who were made hypogonadal by the administration of the GnRH analog leuprolide . Conversion of testosterone to estradiol in these men was blocked by administration of the aromatase inhibitor letrozole. Testosterone and estradiol were replaced transdermally. When testosterone was lowered by leuprolide administration, bone formation—as judged by osteocalcin and PINP concentrations—decreased markedly, and bone resorption, as judged by urine N -telopeptide and total deoxypyridinoline, increased markedly ( Fig. 78.4 ). Administration of estradiol restored osteocalcin and PINP to baseline values, and administration of testosterone also restored osteocalcin, even though its conversion to estradiol was blocked. Administration of estradiol also restored, at least partially, the markers of bone resorption, but testosterone, its conversion to estradiol blocked, did not ( Fig. 78.4 ). The results suggest that testosterone can stimulate bone formation either directly or via conversion to estradiol but can inhibit bone resorption only via conversion to estradiol.
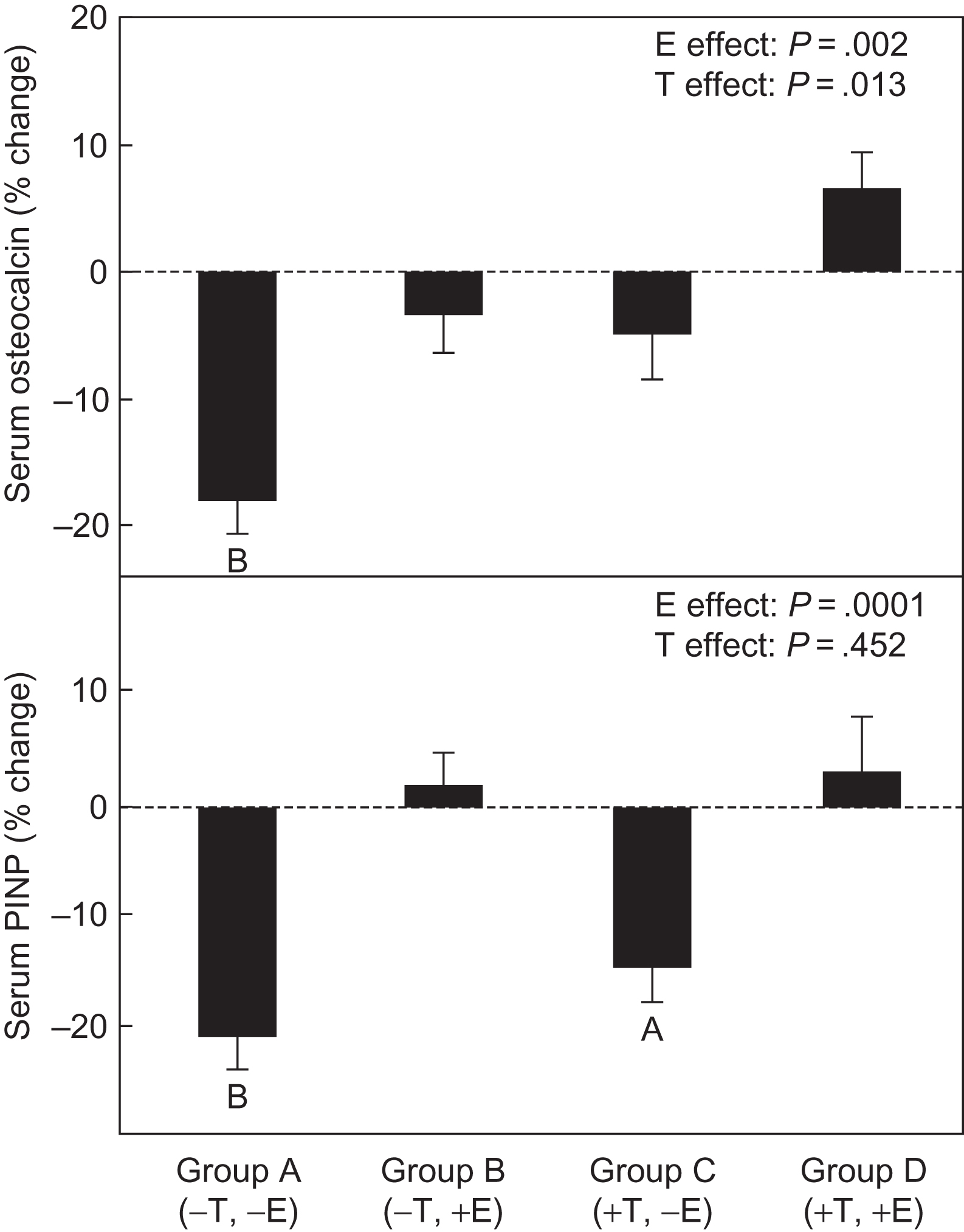
In a similar study, 192 men ages 20–50 years were made hypogonadal by the GnRH analog goserelin and then were randomized to receive graded doses of testosterone for 16 weeks. Another 202 men received the aromatase inhibitor anastrozole in addition to goserelin and graded doses of testosterone . This design allowed determining the effect of graded levels of testosterone and also the degree to which the effect of testosterone on bone is mediated via its conversion to estradiol.
In the men treated with goserelin and testosterone, serum testosterone concentrations <200 ng/dL resulted in significantly higher levels of C-telopeptide (CTx), a marker of bone resorption. In these men, only serum testosterone concentrations <100 ng/dL resulted in increased levels of PINP, a marker of bone formation. The results suggest that testosterone deficiency increases both bone resorption and formation, but only severe deficiency increases bone formation.
In the men treated with anastrozole as well as with goserelin and testosterone, CTx increased 50%–100% more than in those treated with just goserelin and testosterone, indicating that testosterone’s inhibition of bone resorption is mediated largely via its conversion to estradiol. However, the increase in CTx was less in those receiving higher doses of testosterone replacement, suggesting that testosterone has some direct effect in lowering bone resorption, independent of estradiol. Estradiol levels above 10 pg/mL and testosterone levels above 200 ng/dL were sufficient to prevent an increase in bone resorption.
78.3.2.3
Effect on trabecular architecture and strength
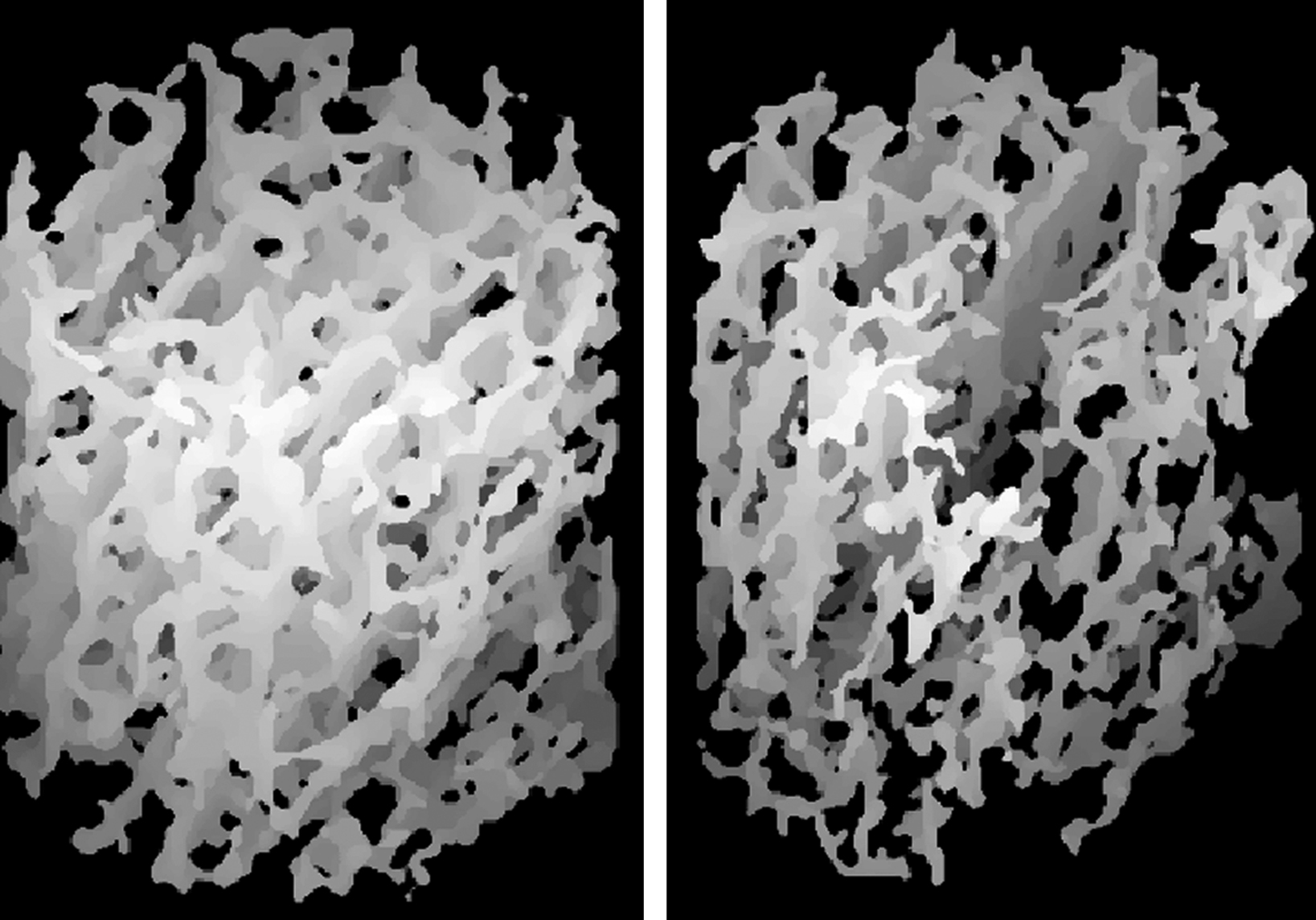
Hypogonadal men not only have lower BMD than eugonadal men but also, in one study, exhibited trabecular architecture that was deteriorated compared to that of eugonadal men ( Fig. 78.5 ) ; testosterone replacement partially restored the connectivity ( Fig. 78.6 ) . In this study, trabecular architecture of the distal tibia was assessed by micro–magnetic resonance imaging (micro-MRI), a noninvasive technique with sufficient resolution to discern individual trabeculae. When 10 men who were severely hypogonadal but were not being treated with testosterone were compared to 10 eugonadal men matched for race and age, the hypogonadal men exhibited parameters of trabecular architecture reflecting connectivity 33% worse than that of the eugonadal men, although their BMD was only 16% worse ( Table 78.1 , Fig. 78.5 ). When the 10 hypogonadal men were replaced with testosterone for 2 years, these architectural parameters improved by a mean of 11% ( Figs. 78.6 and 78.7 ), while the eugonadal men, who were not treated but followed for 2 years, showed no change in these parameters. This improvement in connectivity suggests the possibility that testosterone exerts an anabolic effect on bone.