Summary of Key Points
- •
Since the initial identification of radiofrequency ablation (RFA) as the prototypical thermal ablation technique, it has been joined by microwave ablation, cryoablation, and, more recently, irreversible electroporation as potential options for tumor ablation.
- •
Factors that influence size of the ablation zone can be divided into probe and tissue characteristics. Probe characteristics can vary by the number of probes used, the use of internal cooling, and their configuration (linear or curved array). Tissue characteristics greatly influence the ablation zone size; lung tissue is prone to tissue dehydration when heat is applied. RFA in the lung can be impeded by tissue dehydration with resultant decreased electrical conductivity. Microwave energy, by contrast, can penetrate charred tissues, thus allowing continuous power application for the duration of the treatment and generation of very high temperatures in the lung.
- •
Thermal ablation can be offered to patients with medically inoperable stage I nonsmall cell lung cancer (NSCLC). Patients should be selected by an interdisciplinary team, and the maximum tumor diameter should probably not exceed 3 cm to 3.5 cm.
- •
Aside from its use in stage I lung cancer, RFA is useful for the treatment for patients with a solitary pulmonary nodule remaining after standard therapy of a stage IIIa or IV NSCLC; for salvage therapy of residual or recurrent disease after resection, chemotherapy, and/or radiation; and for pulmonary metastases where the primary disease is controlled in a patient who is a poor surgical candidate.
- •
Major complications from RFA are rare. The reported rate of major complications is 9.8% and comprises pleuritis, pneumonia, lung abscess, hemorrhage, and pneumothorax requiring pleurodesis.
- •
Reported 3-year and 5-year survival rates after RFA range from 36% to 88% and from 19% to 27%, respectively. Estimated 3-year cancer-specific survival ranges from 59% to 88%.
- •
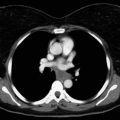
The expected postablation findings on computed tomography include a residual nodule, fibrosis, atelectasis, and cavitation.
Radiofrequency ablation (RFA) was first described with use of a modified Bovie knife in the liver in animal studies published in 1990.1,2 Subsequent descriptions of successful ablation of liver tumors in the mid-1990s elicited interest in using the technique in other organs.3,4 RFA in the lung was first found to be safe and efficacious in animal studies in both healthy lung and VX2 sarcomas in the lungs of rabbits.5,6 Successful use of RFA for lung tumors in humans was described in 2000 in a study of patients with inoperable NSCLC.7 RFA has since been adopted as a treatment alternative for patients with early stage NSCLC who are unable to undergo surgical resection. Since the initial identification of RFA as the prototypical thermal ablation technique, it has been joined by microwave ablation, cryoablation, and, more recently, irreversible electroporation as potential options for tumor ablation.
Mechanism of Action of Ablation Techniques
Radiofrequency Ablation
Radiofrequency refers to the portion of the electromagnetic spectrum from 3 Hz to 300 GHz. Thermal ablation using RFA occurs as a result of delivery of an electrical current to tumor cells surrounding the RFA probe tip. Molecules adjacent to the tip are forced to vibrate rapidly, thus creating frictional energy loss between adjacent molecules ( Fig. 38.1 ). These energy losses are manifested as a rise in tissue temperature, known as the Joule effect. Tissues nearest to the electrode are heated most effectively, whereas more peripheral areas are heated by thermal conduction.
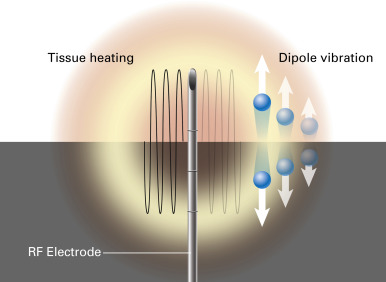
Thermal ablation with RFA results in coagulative necrosis. Once cytotoxic temperatures are achieved in the ablation zone, denaturation of intracellular proteins and destruction of the cell-membrane lipid bilayer result in irreversible cell death. Heat is transferred from cells immediately adjacent to the electrode tip away from the electrode by thermal conduction. A temperature at the electrode tip above 60°C is needed to achieve cell death. Tissue conductivity can, however, be impaired at temperatures above 95°C. Such overheating leads to boiling of the water-predominant tissues, causing steam formation, tissue charring, and a sharp rise in tissue impedance, thereby limiting the effectiveness of RFA. Therefore the aim of thermal tumor ablation is to achieve a temperature range of 50°C to 100°C throughout the entire target volume for 4 minutes to 6 minutes without charring or vaporizing tissues. Multiapplicator ablation is possible by rapidly switching from one electrode to another during electrode activation. The radiofrequency circuit requires a return path from the ablation probe tip. This return path consists of two to four grounding pads applied to the patient’s skin. The grounding pads disperse current over a much wider surface area than the probe tip, which is therefore the only site of tissue damage.
Microwave Ablation
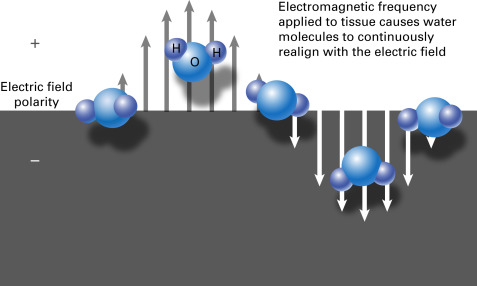
When an electromagnetic frequency of either 915 MHz or 2.45 GHz is applied to tissue, some of the energy forces molecules with an intrinsic dipole moment, such as water molecules, to continuously realign with the applied field. This rotation of molecules increases kinetic energy and local tissue temperatures in a process known as dielectric hysteresis ( Fig. 38.2 ). Tissue destruction occurs when tissues are heated to lethal temperatures, which can reach up to 150°C. Microwave power does not rely on electrical conductivity and can therefore penetrate tissues of low electric conductivity, such as lung and desiccated or charred tissue. The high temperatures achievable at the probe tip improve ablation efficacy by increasing thermal conduction into the surrounding tissues. Because it is not part of an electrical circuit, microwave ablation does not require grounding pads. Multiapplicator ablation is possible with microwave energy, and, unlike with RFA, this can be powered continuously without switching from one electrode to another during electrode activation. Also unique to microwave ablation is the ability to use multiple antennas, which are positioned and phased to exploit overlap of the electromagnetic field.
Cryoablation
Cryoablation involves rapid cooling of the tissues by means of the Joule–Thompson effect, whereby rapid expansion of a high-pressure gas results in a change in the temperature of the gas. When the gas, typically argon, reaches the distal tip of the cryoablation probe, it is forced through a narrow opening and rapidly expands at atmospheric pressure, leading to rapid cooling. This process occurs inside the needle so that the patient is not directly exposed to the emitted gas. The probe is then sequentially warmed and cooled again, to augment cellular damage. Warming is performed by the release of high-pressure helium through the probe tip, which increases in temperature when released into the atmosphere.
During rapid tissue cooling, water is trapped within the cellular membrane, resulting in intracellular ice formation. When the temperature is maintained below the freezing point of water, intracellular ice formation can cause recrystallization and extension of the ice within the intracellular matrix. Alternatively, if gradual cooling occurs, extracellular ice crystals form, which sequester extracellular water. During the thawing cycle, water returns to the intracellular space and causes cellular lysis and enzymatic and membrane dysfunction ( Fig. 38.3 ). As a secondary effect in the adjacent tissues, intracellular ice crystal formation in blood vessels causes damage to the vascular endothelial cells. Reperfusion in the post-thaw period recruits platelets, which contact the damaged endothelium, resulting in thrombosis and ischemia. With each successive freeze–thaw cycle, tissue cooling is faster, and the volume of frozen tissue and extent of tissue destruction are enlarged. The optimal temperature to ensure tumor death is around –50°C. Cryoablation has an advantage over other thermal ablation modalities in that the ice ball created during cryoablation is visible on computed tomography (CT) images, allowing the operator to monitor the extent of ablation.

Irreversible Electroporation
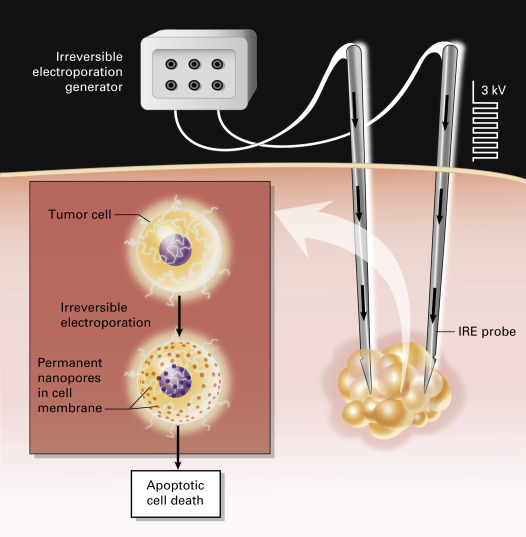
Irreversible electroporation is a nonthermal technique for tumor ablation that causes irreversible damage to the cell membrane by applying pulsed electric fields of up to 3 kV/cm to the ablation target, thereby inducing cell death. The cellular lipid bilayer is disrupted by these high-voltage electrical currents because of the formation of permanent nanopores, which in turn disrupt cellular homeostasis ( Fig. 38.4 ). Irreversible electroporation has two theoretical advantages over thermal ablation: (1) because it is nonthermal, the heat-sink phenomenon is not observed; and (2) irreversible electroporation theoretically preserves tissue interfaces and therefore is thought to spare sensitive structures such as the airways and nerve sheaths. Its use in the lung has not been fully studied in humans, but its potential for preserving airways and mediastinal vessels makes it an attractive future option for the treatment of unresectable lung tumors in anatomically sensitive locations. Irreversible electroporation must be performed using general anesthesia with complete neuromuscular blockade, to avoid generalized muscle contractions. High-voltage pulses are delivered with electrocardiographic gating to minimize the risk of cardiac arrhythmias. Further research is needed before the clinical application of this interesting technology can be optimized.
Technical Factors Influencing Size of the Ablation Zone
Factors that influence size of the ablation zone can be divided into probe and tissue characteristics.
Probe Characteristics
Probe characteristics include electrode exposure length, number of probes, treatment duration, maximum temperature reached, type of energy used, and energy pulsing and cooling (in the case of RFA). Length of the RFA zone has been shown to increase proportionally using exposure lengths of up to 3 cm in the liver. Above this tip size, however, a cylindric ablation zone is not achieved and heterogeneity is more often observed, leading to a dumbbell-shaped ablation zone. To offset this limitation, more than one probe can be used to produce overlapping ablation zones in larger or nonspherical lesions. Umbrella-configuration RFA probes with multiple hooked arrays have also allowed the creation of larger ablation zones. In addition, the size of the ablation zone increases linearly in proportion to the probe gauge size from 24G to 18G. Treatment duration is proportional to ablation zone volume up to a maximum of 6 minutes using RFA in the liver, but no additional benefit is achieved by using ablation times beyond 6 minutes.
To avoid the deleterious effects of tissue vaporization and charring and to increase the size of the ablation zone, internally cooled probe tips have been devised, whereby chilled saline is pumped through the shaft of the needle. Saline infusion of microwave ablation probes has also been shown to produce larger ablation zones in the lung and liver. However, the viscosity of water can limit flow and cooling capacity in small-diameter microwave antennas, and therefore compressed gas may be used to cool the microwave probe. Similar advantages have been described using a pulsed RFA system, whereby cyclic periods of low-current deposition are alternated with higher peak currents; this strategy allows the tissues to rehydrate during the ablation, thus reducing tissue impedance and charring. High levels of impedance at the ablation probe tip (e.g., >1000 ohm) can also be fed back to the radiofrequency generator and inhibit RFA impulse generation. As mentioned earlier, radiofrequency technology allows the combination of multiple probes to achieve a larger, more confluent ablation zone over a shorter treatment duration than with sequential ablation.
The type of energy applied influences the size of the ablation zone and the time required to reach an ablative temperature. Radiofrequency relies on both electrical and thermal conductivity. Heating a tissue to temperatures near or above 100°C causes water to boil and evaporate, leading to tissue dehydration and altered electrical conductivity (e.g., charred tissue), which inhibits the flow of electrical current. Microwave energy, by contrast, can penetrate charred or desiccated tissues, thus allowing continuous power application for the duration of the treatment, generation of very high temperatures, and less susceptibility to heat-sink effects than radiofrequency energy. These high temperatures can be reflected along the probe shaft, raising concerns about fistula formation and skin burns at the needle entry site; these complications can be avoided by using shorter ablation sessions and cooled antenna devices.
Tissue Characteristics
The electrical, thermal, and mechanical properties of the ablated tissue are largely dependent on water content and cellular makeup. Normal lung tissue, when compared with the liver, has lower electrical and thermal conductivity, relative permittivity, and effective conductivity. A low level of electrical and thermal conductivity limits the flow of radiofrequency energy. Lung tumors have relatively densely packed cells and therefore behave similar to solid organs, with higher conductivity than adjacent lung tissue. Enhanced heating has been observed in experimental models at the interface between these tissues (e.g., tumor and aerated lung), but the clinical application of this feature has not been elucidated. Microwave ablation takes advantage of the low relative permittivity and effective conductivity of the lung and as a result leads to deeper penetration than in solid organs. Cryoablation relies on thermal conductivity and ice-ball formation, and because most tissues have a high water content, thermal conductivity is usually good. However, cryoablation in the lung is limited by its inherently low thermal conductivity. Nevertheless, this drawback is progressively overcome as ice-ball formation progresses, which increases the thermal conductivity of the ablation target. Cryoablation in the lung produces an ablation zone slightly smaller than that produced in the kidney but larger than that produced in the liver.
Heat Sink
Blood vessels within the target tissue provide a source of convective tissue cooling (or heating in the case of cryoablation) known as heat sink. Heat sink theoretically applies to all methods of thermal ablation, but is clinically observed in varying degrees. Vessel size within the target tissue is a major determinant of this effect. The presence of vessels larger than 3 mm in diameter in direct contact with the ablation target was reported to be associated with decreased coagulation necrosis in RFA and an increased rate of local recurrence in the liver and lung, respectively. Microwave ablation appears to be less sensitive to the heat-sink effect because of a greater magnitude of heating and better tissue penetration than with RFA. Cryoablation, which relies on thermal conductivity, has not shown a measurable heat-sink effect in the lung or liver. ,
Indications for Thermal Ablation
The standard of care for stage I NSCLC is surgical resection. However, only one-third of patients are eligible for surgical intervention. Thermal ablation can be offered to patients with medically inoperable stage I NSCLC using standards described by the Society of Interventional Radiology. Patients should be selected by an interdisciplinary team, and the maximum tumor diameter should probably not exceed 3 cm to 3.5 cm.
Because the duration of clinical experience and the volume of published data are greater for RFA than for other ablation modalities, RFA has received endorsement by means of clinical guidelines. However, microwave ablation is likely to be increasingly used for NSCLC because of its theoretical advantages over RFA, including a less severe heat-sink effect and faster, greater heating. Cryoablation and irreversible electroporation have not yet been formally recommended for use in lung tumor ablation outside of a research setting.
Aside from its use in stage I lung cancer, RFA has been identified as useful for the following: treatment for patients with a solitary pulmonary nodule remaining after standard therapy of a stage IIIa or IV NSCLC; salvage therapy of residual or recurrent disease after resection, chemotherapy, and/or radiation; and pulmonary metastases where the primary disease is controlled in a patient who is a poor surgical candidate. Combined RFA therapy and inhibition of the epidermal growth factor receptor (EGFR) receptor may also confer a benefit in the treatment of patients who have NSCLC with an EGFR mutation and acquired resistance to EGFR tyrosine kinase inhibitors in one or more metastases.
Complications of Thermal Ablation
Pneumothorax is the most commonly reported complication for all thermal modalities. Self-limited pneumothorax had a reported incidence of 22.4% after RFA, and pneumothorax requiring chest tube insertion (but not pleurodesis) had a similar incidence (22.1%) in a retrospective study of 1000 patients. Major complications from RFA are rare. The reported rate of major complications is 9.8% and comprises aseptic pleuritis, pneumonia, lung abscess, hemorrhage, and pneumothorax requiring pleurodesis. Rarer major complications include bronchopleural fistula, tumor seeding, and nerve or diaphragmatic injury (<0.5% incidence). The reported mortality rate after RFA for lung tumors is 0.4%. Nerve injury has an incidence of 0.2% to 0.3%, affecting the phrenic, brachial, left recurrent laryngeal, and intercostal nerves and the stellate ganglion.
The literature on microwave ablation includes reports of similar complication rates, including self-limited pneumothorax (27%), pneumothorax requiring chest tube insertion (12%), and skin burns (3%). Although not commonly reported in the literature on RFA, skin burns have been described in two patients who had microwave ablation, one of whom required debridement and chest wall reconstruction, presumably due to shaft heating. Intraprocedural or periprocedural death has not been described after microwave ablation. One death was reported from a delayed infectious complication at 6 months after microwave ablation. Bronchopleural fistula has also been reported as a rare complication of microwave ablation, although endobronchial valve insertion is reported to be a potential therapy for fistulas.
Complications from cryotherapy of lung tumors are similar to those of previously described ablation modalities. The risk of hemorrhage with cryotherapy is theoretically greater because this procedure lacks the cautery effect of heat-based ablation. Two studies of cryoablation-related complications reported hemoptysis in 36.8% to 55.4% and massive hemoptysis in 0% to 0.6%. Pneumothorax and pleural effusion are the most common complications, with incidence rates of 61.7% and 70.5%, respectively. Less common complications include phrenic nerve palsy, frostbite, and empyema (0.5% each), and tumor seeding (0.2%).
Pulmonary function testing after RFA has been described in the literature. One study reported impairment of vital capacity and forced expiratory volume at 1 second at both 1 month and 3 months after ablation. In this study, severe pleuritis and ablation of a large volume of adjacent parenchyma were shown to be independently associated with a decline in pulmonary function. However, two other studies reported no deterioration in pulmonary function at 3 months, 6 months, or 24 months, which suggests that the functional impact of RFA may be transient.
Careful patient selection may minimize the risk of complications. For example, in the largest case series that included a report of complications after RFA, previous systemic chemotherapy was a significant risk factor for aseptic pleuritis, and prior external-beam radiotherapy and advanced age were significant risk factors for pneumonia. Patients with emphysema had a greater predilection for lung abscess and pneumothorax requiring pleurodesis. Finally, serum platelet count (≤180,000 cells/μL) and tumor size (>3 cm) are significant predictors of hemorrhage ( p < 0.002 and 0.02, respectively).
Reported Outcomes of Thermal Ablation
Survival and Recurrence
The literature analyzing outcomes after RFA as a therapy for stage I primary NSCLC is limited by study size and population heterogeneity in terms of concomitant treatments administered (e.g., chemotherapy and in-field radiation) and selection bias. The latter relates to the fact that long-term survival data are strongly influenced by medical comorbidity. For example, the all-cause mortality rate for patients with inoperable early stage lung carcinoma (19.1%) is substantially higher than that of operable patients (3.4%). Therefore, lung cancer–specific survival and disease-free intervals may be better measures of treatment outcome. With allowance for these limitations, recurrence rates after RFA of stage I NSCLC were reported in 11 studies; the aggregate rate of local progression was 24% (95 of 403 patients) and the rate of metastasis at any site was 31% (42 of 137; Table 38.1 ). Reported 3-year and 5-year survival rates ranged from 36% to 88% and from 19% to 27%, respectively. Estimated 3-year cancer-specific survival ranged from 59% to 88%. Patients with synchronous and metachronous primary NSCLCs were also reported to have similar local control rates and survival outcomes. The cellular subtype previously called bronchioloalveolar carcinoma (now referred to as adenocarcinoma in situ or minimally invasive adenocarcinoma) may have a better prognosis after RFA. Lanuti et al. described a 90% local control rate of bronchioloalveolar carcinoma after RFA, compared with 68.5% for all cell types.
| Author | Year of Publication | Modality | No. of Tumors | Median Size (range), mm | 2-Y Survival, % | 3-Y Survival, % | 5-Y Survival, % | 3-Y CSS, % | No. of Tumors With Local Progression, % | No. of Tumors With Any Metastasis, % |
|---|---|---|---|---|---|---|---|---|---|---|
| Pennathur et al. | 2007 | RFA | 19 | 26 a (16–38) | 68 | NR | NR | NR | 3 (16) | NR |
| Hiraki et al. | 2007 | RFA | 20 | 20 (13–60) | 84 | 83 | NR | 83 | 7 (35) | 4 (20) |
| Simon et al. | 2007 | RFA | 80 | 30 a (10–75) | 57 | 36 | 27 | NR | NR | NR |
| Hsie et al. | 2009 | RFA | 12 | NR | NR | NR | NR | NR | 1 (8) | 4 (33) |
| Lanuti et al. | 2009 | RFA | 34 a | 20 a (8–44) | 78 | 47 | NR | NR | 12 (35) | 15 (44) |
| Zemlyak et al. | 2010 | RFA | 12 | NR | NR | 87.5 | 19 | 87.5 | 4 (33) | 3 (25) |
| Hiraki et al. | 2011 | RFA | 52 | 21 a (7–60) | 86 | 74 | NR | 80 | 16 (31) | NR |
| Ambrogi et al. | 2011 | RFA | 59 a | 26 (11–50) | NR | NR | 25 | 59 | 13 (22) | 16 (27) |
| Dupuy et al. | 2013 | RFA | 52 | NR | 70 | NR | NR | NR | 19 (37) | NR |
| Liu et al. | 2013 | MWA | 15 | 24 (8–40) | NR | NR | NR | NR | 5 (33) | NR |
| Zemlyak et al. | 2010 | Cryoablation | 27 | NR | NR | 77 | 77 | 90.2 | 3 (11) | 2 (7) |
| Yamauchi et al. | 2011 | Cryoablation | 34 | 14 (5–30) | 88 | 88 | NR | NR | 1 (3) | 6 (18) |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree